Methods Type:
Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2011.29
| Attachment | Size |
|---|---|
| 221.93 KB |
The endoplasmic reticulum (ER) is an intracellular organelle that serves many functions including protein synthesis, processing (folding, glycosylation, quality control) and trafficking, lipid and xenobiotic metabolism, calcium homeostasis and intracellular signaling (4, 12, 14). Morphologically the ER is composed of a continuous network of flattened saccules or cisternae, tubules and the nuclear envelope which together extend throughout the cell (2, 20). ER is usually divided into rough (RER) and smooth (SER) by the presence or absence of membrane bound ribosomes on the external surface that are engaged in protein synthesis.
RER is responsible for processing about 1/3 of cellular proteins in most cells including secretory proteins and membrane proteins such as receptors, transporters and ion channels. Because of its role in synthesizing and secreting digestive enzymes the pancreatic acinar cell possesses abundant RER and is responsible for the synthesis of over 90% of cellular protein. Beta cells and activated pancreatic stellate cells also possess abundant RER while pancreatic duct cells have much less.
Somewhat surprisingly the RER in acinar cells also plays a major role as the storage site for sequestered Ca2+ which can be released upon cellular stimulation by acetylcholine or CCK. Whether SER plays any role in Ca2+ release is unclear.
Smooth ER is much less prominent in pancreas than it is in hepatocytes where it is involved in drug and xenobiotic metabolism. Although not well studied, smooth ER in the pancreas is almost certainly involved in lipid synthesis for maintaining cellular membranes.
To study the composition and functions of the ER, various methods have been described to isolate enriched or purified ER. This entry will present the methods we used to purify ER and assess its protein composition by Western blotting and mass spectrometry. Isolated pancreatic ER fractions have been used by us and others to study protein translation, Ca2+transport and release, cargo sorting and export and identification of resident proteins. Recent interest in the ER has focused on ER stress and its potential role in pancreatic disease. For imaging the ER in intact cells see the Pancreapedia entry by Dingsdale et al (11).
1. Materials for Isolation of ER
1.1 Rat or mouse pancreas as desired from control or treated animals.
1.2 Motor driven Potter – Elevjhem homogenizer. The glass Teflon homogenizer can be purchased from Kontes, Wheaton or Charles Thomas. We use a Charles Thomas Size B or Kontes size 22 for rat or mouse pancreas and a Kontes size 20 for isolated acini. For homogenizing intact pancreas first mince the tissue with scissors to enhance tissue breakdown. We then used a serrated pestle to prepare the homogenate. The most important aspect is the clearance which generally increases with the size of the chamber volume. The Kontes homogenizers generally have a smaller clearance. A Polytron type homogenizer should not be used because it will disrupt cellular organelles and fragment all cellular membranes.
1.3 A high speed refrigerated centrifuge and an ultracentrifuge. We use Beckman ultracentrifuges with a Ti70 angle head and a SW-41 swinging bucket rotors with appropriate tubes.
1.4 A 2M sucrose stock for preparing discontinuous sucrose gradients.
1.5 A homogenizing solution of 0.3 M sucrose, 10 mM Hepes-NaOH (pH 7.0 at 40C) with 2mM dithiothreitol and protease inhibitors usually 1mM benzamidine, 0.5 mM PMSF (freshly added from a 100 mM stock in ethanol) and 150-200 units/ml of aprotinin, a kallikrein inhibitor. For studies of protein phosphorylation, kinase and phosphatase inhibitors can be added.
1.6 A solution of 1 M KCl and 1 mM puromycin for removing ribosomes.
2. Methods for Isolation of ER
2.1 Rat or mouse pancreas is homogenized in 5-10 volumes of ice-cold homogenizing solution by use of a motor driven glass Teflon homogenizer at medium speed. For pancreas 7-10 strokes over 1 min at about 1000 rpm is usually sufficient. For isolated acini, the acinar suspension is centrifuged at low speed (50-100 g) and washed in either calcium free KHB or PBS and resuspended in 10 volumes homogenizing solution. Homogenization requires 15- 25 strokes of the motorized pestle.
2.2 The homogenate is first centrifuged at 500-1000 g for 10 min and the supernatant then centrifuged at 12,000 g for 10-20 min to remove all debris, nuclei, zymogen granules and mitochondria. If you set the final spin at the high end of the g x time range ( 12,000 g for 15 min) you will remove mitochondria more completely, but will lose some RER.
2.3 To collect vesiculated ER also referred to as “microsomes”, one of the following procedures is used.
a. Centrifugation at 150,000 g for 30 min or 100,000 g for 60 min in the Ti 70 rotor or equivalent to obtain total microsomes. (Note 1)
b. Centrifugation at 27,000 g for 10-20 min to obtain a heavy microsomal fraction which contains primarily RER (17). This method has the advantage of speed but is less pure than method c.
c. Centrifugation at 100,000 g for 60 min in a SW 27 or SW 41 rotor of a gradient prepared by layering 4 ml of the postmitochondrial supernatant on top of a 2ml cushion of 1.3 M sucrose buffered with 10 mM Hepes (pH 7.0) and supplemented with 1 mM MgCl2, 2 mM DTT and protease inhibitors. Purified rough microsomes are collected at the bottom of the tube (22).
2.4 Purification of both RER and SER is by the workflow shown in Fig 1. The total microsomal pellet prepared as in 2.3a is resuspended in 1.25 M sucrose, 20 mM Hepes with benzamidine, DTT and PMSF homogenizing solution (see point 1.5) by three strokes of a motor driven homogenizer. This suspension is placed in the middle of a discontinuous sucrose gradient whose volumes are adapted to fill the centrifuge tube. For SW 41 tubes we use (from top to bottom); 6 ml of 0.3 M sucrose, 1.5 ml of 1.15 M sucrose, 0.5 ml of 1.28 M sucrose containing the total microsomes, 1.5ml of 1.35 M sucrose and 2 ml of 2 M sucrose ( 7, 16). The tubes are centrifuged for 90 min at 180,000 g and 4o C in a SW 41 rotor with 2 to 6 tubes used depending on the scale of the preparation. The smooth microsomes can be collected with a pipette tip at the 0.3 - 1.15 M sucrose interface and the rough microsomes at the 1.35 – 2.0 M interface. The material at each interface is diluted to 0.3 M sucrose and collected by centrifugation at 100,000 g for 60 min.
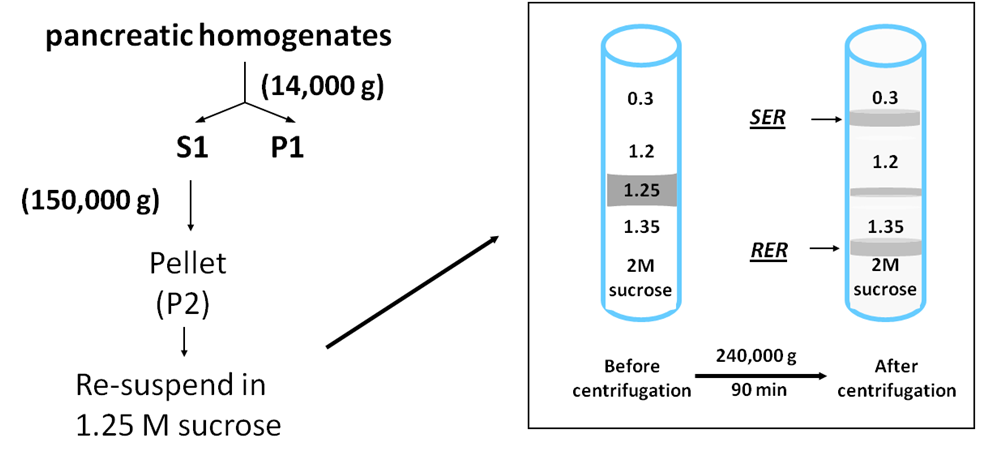
Fig 1. Workflow for purification of pancreatic RER and SER. Rat pancreas was homogenized and after two centrifugations the P2 pellet was suspended in 1.25 M sucrose and subjected to discontinuous gradient fractionation. After centrifugation the RER and SER are collected at interfaces. Modified from Ref (7); for Ems of the RER and SER fractions see Ref (16).
2.5 If it is desirable to remove the ribosomes (usually it is not complete) in a non destructive manner, the RER pellet is resuspended in 1.0 M KCl with 1 mM puromycin, incubated on ice for 30 min and collected by centrifugation at 48,000 g for 30 min (1). This treatment will also remove most secretory proteins adherent or in the lumen. More complete removal can be obtained by alkaline wash with NaHCO3 at pH 7.8. (19).
2.6 The purity of the cell fractions can be assessed by enzyme assays (rarely done today), electron microscopy or immunoblotting using markers for ER such as calreticulin, BIP and calnexin along with markers for contaminating organelles such as cyclophillin A (cytoplasm) or VDAC1 (mitochondria) (7, 16, 19) (Note 2). Immunoblotting for a ribosomal protein such as S6 can assess efficiency of ribosomal stripping. Fig 2 shows an assessment of a preparation of RER by immunoblotting.

Fig 2. Following the purification of rat pancreatic RER by immunoblotting of known protein. Western blots of selected proteins in pancreatic lysate (total) a 14,000 g pellet (P1) a subsequent 150,000 g pellet (P2), RER purified on the sucrose gradient described in Fig 1, and RER stripped with puromycin and high salt. Calreticulin and BiP are content markers for the RER, S6 a ribosomal protein, Cyclophillin A a cytosolic marker and VDAC1 a mitochondrial marker. Modified from Ref (7).
2.7 Alternative methods of ER fractionation that have been applied to the rodent pancreas include continuous manitol density gradients (3) and Percoll gradients (9). For integration into an earlier scheme of cell fractionation used for guinea pig pancreas see Tartakoff and Jamieson (19). Purification of cat pancreatic microsomes and characterization by enzymatic markers is described by Milutinovic et al (15) and Kribben et al (13). Preparation of canine microsomes and their use for protein translation studies is detailed in Walter and Blobel (21).
3. Analysis of ER Proteins
Analysis of the protein composition of the pancreatic ER has been primarily carried out by N-terminal Edmean sequencing (5), immunoblotting and immunohistochemistry (6) of individual proteins and by high throughput proteomic analysis. Both immunoblotting and mass spectrometry are dependent on the purity of the preparation, although immunohistochemistry has the ability to localize without purification and is therefore an excellent tool for confirmation when antibodies exist. It also allows determination of the cell type containing the protein in the pancreas. One area that has been studied extensively using canine pancreatic ER is the protein translocation and biosynthesis machinery (10, 22) which has led to the identification of the individual proteins involved.
Recently, proteomic techniques have been applied to the pancreatic ER. Two large datasets as well as several more targeted studies have been carried out and are reviewed in (8). Zahedi et al (23) studied carbonate extracted stripped canine RER to identify integral membrane proteins. They identified 258 proteins most of which had a predicted transmembrane domain although some were ribosomal or soluble secretory proteins. In a smaller study, canine stripped RER was extracted with detergent and 32 integral membrane proteins were identified (18). In a study of rat RER and changes in protein content with pancreatitis Chen et al (7) identified 469 unique proteins including ones from all major functional categories of RER. In this study relative quantitation between normal and pancreatitis was evaluated with iTRAQ tagging and 37 proteins identified whose relative amount changed in models of pancreatitis. To date there have not been studies of mouse or human isolated ER. The data compiled from the two large studies is compared to liver ER in (Chen et al Proteomics, 2010) and the pancreatic data set is listed elsewhere in The Pancreapedia.
4. Notes
1. To preserve structure and function of microsomes it helps to centrifuge onto a 2M sucrose cushion. This is especially important for ER budding assays where suspending directly from a pellet can produce fragmented and non-functional ER (Personal Communication, Fred Gorelick).
2. It is difficult to use protein markers to separate ER from other parts of the secretory pathway such as Golgi complex or zymogen Granules as their content is synthesized in the RER. For these organelles morphological evaluation is preferable.
5. References
- Adelman MR, Sabatini D, and Blobel G. Ribosome –membrane interaction: Nondestructive disassembly of rat liver rough microsomes into ribosomal and membranous components. J. Cell Biology 56:206-229, 1973. PMID: 4345164
- Baumann O and Walz B. Endoplasmic reticulum of animal cells and its organization into structural and functional domains. Int Rev Cytol. 205:149-214, 2001. PMID: 11336391
- Bayerdorffer E, Streb H, Eckhardt W, Haase W and Schulz I. Characterization of calcium uptake into rough endoplasmic reticulum of rat pancreas. J Membrane Biol. 81:69-82, 1984. PMID: 6208363
- Berridge MJ. The endoplasmic reticulum: a multifunctional signaling organelle. Cell Calcium 32:235-249, 2002. PMID: 12543086
- Blum R, Feick P, Puype M, Vandekerckhove, Klengel W, Nastainczyk W and Schulz I. Tmp21 and p24A, two type I proteins enriched in pancreatic microsomal membranes, are members of a protein family involved in vesicular trafficking. J Biol Chem. 271:17183-17189, 1996. PMID: 8663407
- Bole DG, Dowin R, Doriaux M, and Jamieson JD. Immunocytochemical localization of BiP to the rough endoplasmic reticulum: evidence for protein sorting by selective retention. J Histochem Cytochem. 37:1817-1823, 1989. PMID: 2685110
- Chen X, Sans MD, Strahler JR, Karnovsky A, Ernst SA, Michailidis G, Andrews PC and Williams JA. Quantitative organellar proteomics analysis of rough endoplasmic reticulum from normal and acute pancreatitis rat pancreas. J Proteome Res. 9:885-896, 2010. PMID: 19954227
- Chen X, Karnovsky A, Sans MD, Andrews PC and Williams JA. Molecular characterization of the endoplasmic reticulum: insights from proteomic studies. Proteomics 10:4040-4052, 2010. PMID: 21046616
- Dehlinger-Kremer M, Zeuzem S and Schulz I. Interaction of caffeine-, IP3- and vanadate-sensitive Ca2+ pools in acinar cells of the exocrine pancreas. J Membrane Biol. 119:85-100, 1991. PMID: 2008014
- Dierks T, Volkmer J, Schlenstedt G, Jung C, Sandholzer U, Zachmann K, Schotterhose P, Neifer K, Schmidt B, and Zimmermann R. A microsomal ATP-binding protein involved in efficient protein transport into the mammalian endoplasmic reticulum. EMBO J 15:6931-6942, 1996. PMID: 9003769
- Dingsdale, H, Haynes LP, Tepikin AV, and Lur G. Visualizing the endoplasmic reticulum and its contacts with other organelles in live acinar cells. The Pancreapedia: Exocrine Pancreas Knowledge Base, 2011. DOI: 10.3998/panc.2011.6
- Groenendyk J and Michalak M. The Endoplasmic Reticulum. in Cellular Domains, Ed IR Nabi, John Wiley and Sons, 2011, pp. 113- 131.
- Kribben A, Tyrakowski T and Schulz I. Characterization of Mg-ATP- dependent Ca2+ transport in cat pancreatic microsomes. Am J Physiol Gastrointest Liver Physiol. 244:G480-G490, 1983. PMID: 6133452
- Lavoie C and Paiement J. Topology of molecular machines of the endoplasmic reticulum: a compilation of proteomics and cytological data. Histochem Cell Biol. 129:117-128, 2008. PMID: 18172663
- Milutinovic S, Sachs G, Haase, and Schulz I. Studies on isolated subcellular components of cat pancreas: I. Isolation and enzymatic characterization. J Membrane Biol. 36:253-279, 1977. PMID: 143536
- Preissler M and Williams JA. Localization of ATP-dependent calcium transport activity in mouse pancreatic microsomes. J. Membrane Biol. 73:137-144, 1983. PMID: 6864771
- Ponnappa BC, Dormer RL and Williams JA. Characterization of an ATP-dependent Ca2+ uptake system in mouse pancreatic microsomes. Am J Physiol Gastrointest Liver Physiol. 240:G122-G129, 1981. PMID: 6258447
- Sakai K, Hamanaka R, Yuki H, and Watanabe M. A novel fractionation method of the rough ER integral membrane proteins; resident proteins versus exported proteins. Proteomics 9:3036-3046, 2009. PMID: 19526556
- Tartakoff AM and Jamieson JD. Subcellular fractionation of the pancreas. Methods in Enzymology 31:41-59, 1974. PMID: 4419641
- Voeltz GK, Rolls MM and Rapoport TA. Structural organization of the endoplasmic reticulum. EMBO Reports. 3:944-950, 2002. PMID: 12370207
- Walter P and Blobel G. Preparation of microsomal membranes for cotranslational protein translocation. Methods in Enzymology 96: 84-93, 1983. PMID: 6656655
- Walter P and Lingappa VR. Mechanism of protein translocation across the endoplasmic reticulum membrane. Ann Rev Cell Biol. 2:499-516, 1986. PMID: 3030381
- Zahedi RP, Völzing C, Schmitt A, Frien M, Jung M, Dudek J, Wortelkamp S, Sickmann A, Zimmermann R. Analysis of the membrane proteome of canine pancreatic rough microsomes identifies a novel Hsp40, termed ERj7. Proteomics. 9:3463-3473, 2009. PMID: 19579229

