Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2023.01
| Attachment | Size |
|---|---|
| 245.67 KB | |
| 324.62 KB |
Gene symbol: GRP
1. General Information
Peptides of the bombesin family were first isolated from frog skin in the early 1970s. Bombesin, a 14 amino acid peptide was isolated from the skin of the amphibian Bombina bombina (Figure 1) by Vittorio Erspamer and colleagues, in Parma, Italy.
Figure 1. Bombina bombina also known as the European fire-bellied toad. Image from en.wikipedia.org.
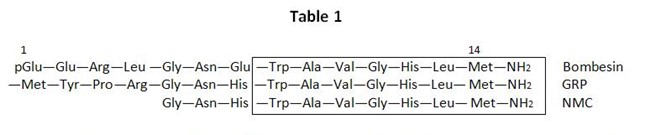
Its amino acid sequence was determined and it was shown to contain blocked amino and carboxyl terminals (1). Related peptides termed alytensin, ranatensin and litorin were isolated from other frogs (18). In the 1980s bombesin like peptides were identified in mammals. The first mammalian bombesin like peptide was isolated from pig gastric tissue and named gastrin-releasing peptide (GRP) because it had a potent action to release gastrin (53). GRP is a 27 amino acid peptide and results from the processing of a 148 amino acid precursor (85). Other subsequently identified related mammalian molecules include Neuromedin B (NMB) isolated from porcine spinal cord (54, 62) and Neuromedin C (NMC) also isolated from pig spinal cord (55) but which is now known to be GRP 18-27 (37). Bombesin, GRP, and NMC share a common carboxyl terminal sequence as shown in Table 1. NMB has a different precursor and one amino acid difference in the consensus heptapeptide. Using cDNA cloning the frog bombesin precursor was shown to contain 107 amino acids (76). The precursor contained a signal peptide sequence and one copy of bombesin followed by a typical processing site. The mammalian peptides GRP and NMB also have a similarly structured prohormone.
Table 1. The entire structure is shown except for GRP which has 27 amino acids (the first 13 amino acids are not shown). The GRP sequence shown is for human, pig, and dog. The other bombesin related peptides have similarity but not identity to the carboxy-terminal sequence.
Bombesin family members are neuropeptides that are broadly located in the CNS and peripheral nervous system especially the parasympathetic nerves. They have also been shown to be present in organs such as the pancreas, urinary bladder, and uterus and in the enteric nervous system where their cell bodies are present in the myenteric plexus. Bombesin and bombesin like peptides (BLP) in the GI tract regulate gastric acid and pancreatic secretion. In the stomach, GRP is the most potent known stimulator of gastrin release in dogs and humans and thereby stimulates gastric acid secretion (6). Bombesin also stimulates CCK release (9, 19) as well as directly regulating pancreas cells as described later. BLPs also stimulate smooth muscle contraction in the intestine, stomach, and gall bladder. They can also regulate uterine and ureter contraction. In all these actions BLPs act in concert with other regulatory systems and are in some cases species dependent.
In the CNS, BLPs are located in specific neurons. BLPs play roles in satiety, thermoregulation, energy balance, circadian rhythm, and behavior (26, 75). A recent important discovery is the role of GRP neurons in the dorsal horn of the spinal cord in relaying the itch response to the brain (67, 73). Another interesting site is the participation of NMB in the sighing response of the respiratory system (46).
Bombesin Receptors
Mammals express three types of bombesin receptors all of which are 7 transmembrane cell surface receptors and signal through heterotrimeric G proteins of the Gq family. Properties of these receptors as determined by ligand binding and molecular cloning are only summarized here but have been reviewed extensively in several publications (26, 37, 42, 73, 75). The most common type is the GRP-R or BBR-2 which responds with high affinity to bombesin, GRP and NMC and was first characterized by the high affinity binding of radioiodinated bombesin to pancreatic acini and brain membranes (38, 57).The second is the NMB-R which responds preferentially to NMB and was originally described in the rat esophagus (92). The third type is an orphan receptor without a known natural ligand identified by homology and termed bombesin receptor subtype 3 (BRS-3) that is present in brain, muscle and islet beta cells and may play a role in glucose homeostasis (21). A fourth receptor has been described in amphibian brain (58). Agonists, antagonists, and radioligands have been identified for the three mammalian receptors and reviewed (37, 75). The receptors appear to internalize agonist peptides but not antagonists although the functional importance of this is unclear. Receptors can also be desensitized and internalized. Bombesin receptors are increased on many tumor cells, and this has been used for ligand imaging and to deliver chemotherapeutic agents particularly in prostate cancer (4, 70). Further properties of the GRP-R and its actions will be considered as studied using pancreatic acinar cells.
2. Bombesin and the Pancreas
In the pancreas, endogenous GRP, the mammalian equivalent of bombesin is primarily located in neurons. Immunofluorescence has localized it to preganglionic vagal fibers, pancreatic ganglia and in beaded neurons running between acini (13, 24, 40, 56, 83). The pig however, was the only species to have a high concentration of immunoreactive GRP in the pancreas (56). Using rat pancreatic lobules, GRP was shown to stimulate acetylcholine release and about half of the effect on amylase release was blocked by neuronal or ganglionic blockers (23). This suggests GRP may act both on neurons and acinar cells. Studies in the pig have shown that electrical stimulation of the vagus nerve releases GRP and its active fragment, GRP (18-27) both in vivo and in the perfused pancreas (40, 41). Furthermore, bombesin receptor antagonists inhibited secretion induced by vagal stimulation by 33% (33). Thus, in the pig, GRP plays a role in endogenous pancreatic secretion although the role in other species may be less.
Pancreatic Enzyme Secretion
Early studies indicated that bombesin administration could stimulate pancreatic secretion in a variety of species including humans, dogs, rats and guinea pigs (2, 5, 14, 34, 59, 66, 91) with the biggest effect being on digestive enzyme secretion. Subsequent studies showed a similar action induced by mammalian GRP and NMC. Moreover, a prominent role was found in the perfused pig pancreas (41). Bombesin peptides also stimulated gastrin, CCK, and acetylcholine release (5, 9, 19, 52). The secretion induced was enzyme rich and poor in bicarbonate. Thus, an initial question was whether bombesin stimulated the pancreas directly or through a neural or hormonal intermediate. However, studies using antrectomized dogs to remove gastrin, CCK antagonists, and atropine have shown in multiple species including humans that the effect of bombesin was primarily direct (2, 30, 31, 47). This was confirmed by the finding of specific bombesin receptors on human pancreatic membranes (82), isolated rodent pancreatic acini (38, 98) and pancreatic AR42J cells (48, 84). The major form in the pancreas is the GRP-R also known as the BB2 receptor (63). In its native state this receptor on mouse acini is an 80 kDa glycoprotein (36). A number of bombesin receptor antagonists have also been developed most of which are modified bombesin peptides some having a reduced peptide bond (12, 29, 93). One of the most potent is [D-Phe6]BN-(6-13) ethyl ester with a Ki of 5nM to inhibit pancreatic amylase secretion stimulated by bombesin. The use of a specific receptor antagonist has shown that bombesin stimulation through these receptors does not play a role in meal stimulated digestive enzyme secretion in vivo (91).
Actions of Bombesin on isolated pancreatic acini
Even after the discovery of mammalian GRP, bombesin continued in common use for the study of cellular mechanisms using isolated acini because of its ready availability. Bombesin and related peptides showed a slight biphasic dose response with reduced ability to stimulate in vitro amylase release from isolated guinea pig acinar cells at supramaximal concentrations; maximal secretion was observed at 100 nM (51, 88). Similar results but with maximal secretion at 100 pM were seen in mouse acini (35). By contrast in rat acini, bombesin induced a monophasic dose response with a maximal response at 1 nM (50, 60). Thus, there is a difference from CCK stimulation which induces a pronounced biphasic dose-response curve in all species studied. Stimulation of amylase release by bombesin and GRP has also been reported from human isolated acini (86).
Bombesin activated IP3 and diacylglycerol production and intracellular calcium release in rodent acini (15, 50, 72). Thus, bombesin activates signaling pathways through heterotrimeric Gq/11 protein similar to CCK although by different receptors. Other actions of bombesin shared with CCK have included activation of protein synthesis mechanisms, protein tyrosine kinases, Src family kinases, PKC, calcineurin, phospholipase A2, p125FAK, ERK, JNK, p70 S6K, eEF2K, PAK2 and PAK4, and downregulation of c-Met (7, 10, 32, 39, 60, 61, 74, 78-80). Other differences between bombesin and CCK on acini are that bombesin induces less damage and ER stress (43). In some studies, bombesin is used as an alternative agonist to show that an event is not initiated by a single receptor (8). Other studies have looked at the properties of bombesin receptors to internalize bombesin (98), their ability to induce residual stimulation (35), to desensitize (50), and to be regulated by CCK (97).
Bombesin and Experimental Pancreatitis
Bombesin by itself does not induce experimental pancreatitis in vivo or acinar cell damage in vitro in contrast to what is produced by caerulein (71, 96). Bombesin increases the activation of intracellular trypsin and the processing of procarboxypeptidase A1 in isolated acini (25). However, the activated enzyme was secreted from the cell following bombesin stimulation thus plausibly explaining the lack of cell damage (25). By contrast, when bombesin stimulation was combined with pancreatic duct obstruction, retention of active enzymes and pancreatitis resulted (65). Another difference from caerulein is that bombesin failed to activate NF-κB (28).
Pancreatic Ductal Secretion
There is some evidence that bombesin peptides can influence the ductal secretion of fluid and electrolytes. This is somewhat species dependent, and in most cases may not be of physiological importance. Most bombesin immunoreactive neurons in the pancreas are located near acinar cells with a lesser innervation of ducts (83). Injection of bombesin can stimulate fluid secretion especially in rats and guinea pigs (2, 66). Most directly, bombesin stimulates fluid secretion by isolated rat and guinea pig ducts and this effected is blocked by a GRP-R antagonist (3, 87). The maximum rate of secretion in response to bombesin was similar to the response to secretin or CCK. While the machinery is present to support secretion there is little evidence that this occurs physiologically. There is no published data that a GRP-R antagonist or gene deletion of the receptor reduces pancreatic juice secretion except for a partial effect in the pig.
Trophic Function of Bombesin Peptides
In addition to stimulating pancreatic enzyme and ductal secretion, bombesin injection stimulates the growth of the pancreas in both adult and newborn rodents (44, 69, 89, 90). Both hyperplasia (increased cell number) and hypertrophy (increased cell size) have been reported. Exogenous GRP also has effects similar to bombesin (11). Bombesin administration can release CCK but blocking CCK action does not reduce the bombesin-induced pancreatic growth (81). In addition, bombesin can preserve exocrine pancreatic function that is reduced in parenteral nutrition (68). Bombesin can also stimulate the mRNA and protein content of specific digestive enzymes (77) and increase polyamine synthesis (89).
Bombesin can also stimulate cell growth pathways in primary cultures of rodent acinar cells (45) and activate expression of early response genes in isolated rat acini (49). However, the importance of bombesin as a trophic factor for the exocrine pancreas is unclear, similar to effects on physiological secretion discussed earlier as bombesin stimulation activates growth pathways normally activated by CCK. Deletion of GRP or its receptor has not been reported to alter pancreatic size.
Bombesin like peptides can also affect the growth of pancreatic cancer cells. This has been studied the most in pancreatic cancer derived cell lines. Similar to effects in some lung cancer, bombesin can act as an autocrine stimulator in cell lines such as Mia PaCa-2 and HPAF (17, 95) although it inhibits growth of H2T cells (20). Pathways involved in growth stimulation include tyrosine kinases, tyrosine phosphatases and MAP kinases (16, 17). However, in a study localizing bombesin receptors in distinct tissue compartments, bombesin receptors were not found in the tumor tissue of pancreatic cancer although they were found in chronic pancreatitis (22). Thus, the role of bombesin in pancreatic cancer requires further study.
3. Tools to study bombesin and GRP
a. Synthetic Peptide
Bombesin can be obtained from multiple sources including Sigma-Aldrich, Research Plus, Bachem, Anaspec and Abbiotec.
b. Antibodies
Antibodies against bombesin are available from Genway and Phoenix Pharmaceuticals.
c. ELISA and RIA
An RIA kit for GRP is available from Phoenix Pharmaceuticals. An ELISA kit against Bombesin is available from My BioSource and against mouse GRP from Antibodies-online.
d. Antagonists
Peptide antagonists against the Bombesin receptor are available from Sigma-Aldrich and Tocris. One of the most potent against the GRP-R is [D-Phe6] BN-(6-13) ethyl ester.
e. Gene deleted mice
Preparation of GRP-R gene deleted mice have been reported (27, 94). Mice have also been prepared with the NMB-R gene deleted (64).
4. References
- Anastasi A, Erspamer V, and Bucci M. Isolation and amino acid sequences of alytesin and bombesin, two analogous active tetradecapeptides from the skin of European discoglossid frogs. Arch Biochem Biophys 148: 443-446, 1972.
- Anderson L, and Dockray GJ. The cholecystokinin antagonist L-364,718 inhibits the action of cholecystokinin but not bombesin on rat pancreatic secretion in vivo. Eur J Pharmacol 146: 307-311, 1988.
- Ashton N, Argent BE, and Green R. Effect of vasoactive intestinal peptide, bombesin and substance P on fluid secretion by isolated rat pancreatic ducts. J Physiol 427: 471-482, 1990.
- Baratto L, Jadvar, H., Iagaru, A. Prostate Cancer Theranostics Targeting Gastrin-Releasing Peptide Receptors. Mol Imaging and Biology 20: 501-509, 2017.
- Basso N, Giri S, Improta G, Lezoche E, Melchiorri P, Percoco M, and Speranza V. External pancreatic secretion after bombesin infusion in man. Gut 16: 994-998, 1975.
- Bertaccini G, Erspamer V, Melchiorri P, and Sopranzi N. Gastrin release by bombesin in the dog. Br J Pharmacol 52: 219-225, 1974.
- Bragado MJ, Groblewski GE, and Williams JA. p70s6k is activated by CCK in rat pancreatic acini. Am J Physiol 273: C101-109, 1997.
- Chen X, Edwards JA, Logsdon CD, Ernst SA, and Williams JA. Dominant negative Rab3D inhibits amylase release from mouse pancreatic acini. J Biol Chem 277: 18002-18009, 2002.
- Cuber JC, Vilas F, Charles N, Bernard C, and Chayvialle JA. Bombesin and nutrients stimulate release of CCK through distinct pathways in the rat. Am J Physiol 256: G989-996, 1989.
- Dabrowski A, Grady T, Logsdon CD, and Williams JA. Jun kinases are rapidly activated by cholecystokinin in rat pancreas both in vitro and in vivo. J Biol Chem 271: 5686-5690, 1996.
- Damge C, Hajri A, Lhoste E, and Aprahamian M. Comparative effect of chronic bombesin, gastrin-releasing peptide and caerulein on the rat pancreas. Regul Pept 20: 141-150, 1988.
- de Castiglione R, and Gozzini L. Bombesin receptor antagonists. Crit Rev Oncol Hematol 24: 117-151, 1996.
- De Giorgio R, Sternini C, Anderson K, Brecha NC, and Go VL. Tissue distribution and innervation pattern of peptide immunoreactivities in the rat pancreas. Peptides 13: 91-98, 1992.
- Delle Fave G, Annibale B, de Magistris L, Severi C, Bruzzone R, Puoti M, Melchiorri P, Torsoli A, and Erspamer V. Bombesin effects on human GI functions. Peptides 6 Suppl 3: 113-116, 1985.
- Deschodt-Lanckman M, Robberecht P, De Neef P, Lammens M, and Christophe J. In vitro action of bombesin and bombesin-like peptides on amylase secretion, calcium efflux, and adenylate cyclase activity in the rat pancreas: a comparison with other secretagogues. J Clin Invest 58: 891-898, 1976.
- Douziech N, Calvo E, Laine J, and Morisset J. Activation of MAP kinases in growth responsive pancreatic cancer cells. Cell Signal 11: 591-602, 1999.
- Douziech N, Lajas A, Coulombe Z, Calvo E, Laine J, and Morisset J. Growth effects of regulatory peptides and intracellular signaling routes in human pancreatic cancer cell lines. Endocrine 9: 171-183, 1998.
- Erspamer V. Discovery, isolation, and characterization of bombesin-like peptides. Ann N Y Acad Sci 547: 3-9, 1988.
- Erspamer V, Improta G, Melchiorri P, and Sopranzi N. Evidence of cholecystokinin release by bombesin in the dog. Br J Pharmacol 52: 227-232, 1974.
- Farre A, Ishizuka J, Gomez G, Evers BM, Saydjari R, Koo JY, Townsend CM, Jr., and Thompson JC. Bombesin inhibits growth of pancreatic ductal adenocarcinoma (H2T) in nude mice. Pancreas 9: 652-656, 1994.
- Feng Y, Guan XM, Li J, Metzger JM, Zhu Y, Juhl K, Zhang BB, Thornberry NA, Reitman ML, and Zhou YP. Bombesin receptor subtype-3 (BRS-3) regulates glucose-stimulated insulin secretion in pancreatic islets across multiple species. Endocrinology 152: 4106-4115, 2011.
- Fleischmann A, Laderach U, Friess H, Buechler MW, and Reubi JC. Bombesin receptors in distinct tissue compartments of human pancreatic diseases. Lab Invest 80: 1807-1817, 2000.
- Flowe KM, Welling TH, and Mulholland MW. Gastrin-releasing peptide stimulation of amylase release from rat pancreatic lobules involves intrapancreatic neurons. Pancreas 9: 513-517, 1994.
- Ghatei MA, George SK, Major JH, Carlei F, Polak JM, and Bloom SR. Bombesin-like immunoreactivity in the pancreas of man and other mammalian species. Experientia 40: 884-886, 1984.
- Grady T, Mah'Moud M, Otani T, Rhee S, Lerch MM, and Gorelick FS. Zymogen proteolysis within the pancreatic acinar cell is associated with cellular injury. Am J Physiol 275: G1010-1017, 1998.
- Guo M, Qu X, and Qin XQ. Bombesin-like peptides and their receptors: recent findings in pharmacology and physiology. Curr Opin Endocrinol Diabetes Obes 22: 3-8, 2015.
- Hampton LL, Ladenheim EE, Akeson M, Way JM, Weber HC, Sutliff VE, Jensen RT, Wine LJ, Arnheiter H, and Battey JF. Loss of bombesin-induced feeding suppression in gastrin-releasing peptide receptor-deficient mice. Proc Natl Acad Sci U S A 95: 3188-3192, 1998.
- Han B, and Logsdon CD. Cholecystokinin induction of mob-1 chemokine expression in pancreatic acinar cells requires NF-kappaB activation. Am J Physiol 277: C74-82, 1999.
- Heinz-Erian P, Coy DH, Tamura M, Jones SW, Gardner JD, and Jensen RT. [D-Phe12]bombesin analogues: a new class of bombesin receptor antagonists. Am J Physiol 252: G439-442, 1987.
- Herzig KH, Louie DS, and Owyang C. In vivo action of bombesin on exocrine pancreatic secretion in the rat: independent of cholecystokinin and cholinergic mediation. Pancreas 3: 292-296, 1988.
- Hildebrand P, Drewe J, Luo H, Ketterer S, Gyr K, and Beglinger C. Role of cholecystokinin in mediating GRP-stimulated gastric, biliary and pancreatic functions in man. Regul Pept 41: 119-129, 1992.
- Hoffmann KM, Tapia JA, Berna MJ, Thill M, Braunschweig T, Mantey SA, Moody TW, and Jensen RT. Gastrointestinal hormones cause rapid c-Met receptor down-regulation by a novel mechanism involving clathrin-mediated endocytosis and a lysosome-dependent mechanism. J Biol Chem 281: 37705-37719, 2006.
- Holst JJ, Knuhtsen S, and Nielsen OV. Role of gastrin-releasing peptide in neural control of pancreatic exocrine secretion. Pancreas 4: 581-586, 1989.
- Hosotani R, Inoue K, Fujii N, Yajima H, and Tobe T. Effect of synthetic neuromedin C, a decapeptide of gastrin-releasing peptide (GRP [18-27]), on blood flow and exocrine secretion of the pancreas in dogs. Life Sci 36: 2429-2434, 1985.
- Howard JM, Jensen RT, and Gardner JD. Bombesin-induced residual stimulation of amylase release from mouse pancreatic acini. Am J Physiol 248: G196-199, 1985.
- Huang SC, Yu DH, Wank SA, Gardner JD, and Jensen RT. Characterization of the bombesin receptor on mouse pancreatic acini by chemical cross-linking. Peptides 11: 1143-1150, 1990.
- Jensen RT, Battey JF, Spindel ER, and Benya RV. International Union of Pharmacology. LXVIII. Mammalian bombesin receptors: nomenclature, distribution, pharmacology, signaling, and functions in normal and disease states. Pharmacol Rev 60: 1-42, 2008.
- Jensen RT, Moody T, Pert C, Rivier JE, and Gardner JD. Interaction of bombesin and litorin with specific membrane receptors on pancreatic acinar cells. Proc Natl Acad Sci U S A 75: 6139-6143, 1978.
- Kiehne K, Herzig KH, and Folsch UR. Differential activation of p42ERK2 and p125FAK by cholecystokinin and bombesin in the secretion and proliferation of the pancreatic amphicrine cell line AR42J. Pancreatology 2: 46-53, 2002.
- Knuhtsen S, Holst JJ, Baldissera FG, Skak-Nielsen T, Poulsen SS, Jensen SL, and Nielsen OV. Gastrin-releasing peptide in the porcine pancreas. Gastroenterology 92: 1153-1158, 1987.
- Knuhtsen S, Holst JJ, Jensen SL, Knigge U, and Nielsen OV. Gastrin-releasing peptide: effect on exocrine secretion and release from isolated perfused porcine pancreas. Am J Physiol 248: G281-286, 1985.
- Kroog GS, Jensen RT, and Battey JF. Mammalian bombesin receptors. Med Res Rev 15: 389-417, 1995.
- Kubisch CH, and Logsdon CD. Secretagogues differentially activate endoplasmic reticulum stress responses in pancreatic acinar cells. Am J Physiol Gastrointest Liver Physiol 292: G1804-1812, 2007.
- Lehy T, Puccio F, Chariot J, and Labeille D. Stimulating effect of bombesin on the growth of gastrointestinal tract and pancreas in suckling rats. Gastroenterology 90: 1942-1949, 1986.
- Lhoste EF, Aprahamian M, Balboni G, and Damge C. Evidence for a direct trophic effect of bombesin on the mouse pancreas: in vivo and cell culture studies. Regul Pept 24: 45-54, 1989.
- Li P, Janczewski WA, Yackle K, Kam K, Pagliardini S, Krasnow MA, and Feldman JL. The peptidergic control circuit for sighing. Nature 530: 293-297, 2016.
- Liehr RM, Reidelberger RD, Varga G, and Solomon TE. Mechanism of bombesin-induced pancreatic secretion in unanesthetized rats. Peptides 14: 717-723, 1993.
- Logsdon CD, Zhang JC, Guthrie J, Vigna S, and Williams JA. Bombesin binding and biological effects on pancreatic acinar AR42J cells. Biochem Biophys Res Commun 144: 463-468, 1987.
- Lu L, and Logsdon CD. CCK, bombesin, and carbachol stimulate c-fos, c-jun, and c-myc oncogene expression in rat pancreatic acini. Am J Physiol 263: G327-332, 1992.
- Matozaki T, Zhu WY, Tsunoda Y, Goke B, and Williams JA. Intracellular mediators of bombesin action on rat pancreatic acinar cells. Am J Physiol 260: G858-864, 1991.
- May RJ, Conlon TP, Erspamer V, and Gardner JD. Actions of peptides isolated from amphibian skin on pancreatic acinar cells. Am J Physiol 235: E112-118, 1978.
- McDonald TJ, Ghatei MA, Bloom SR, Adrian TE, Mochizuki T, Yanaihara C, and Yanaihara N. Dose-response comparisons of canine plasma gastroenteropancreatic hormone responses to bombesin and the porcine gastrin-releasing peptide (GRP). Regul Pept 5: 125-137, 1983.
- McDonald TJ, Jornvall H, Nilsson G, Vagne M, Ghatei M, Bloom SR, and Mutt V. Characterization of a gastrin releasing peptide from porcine non-antral gastric tissue. Biochem Biophys Res Commun 90: 227-233, 1979.
- Minamino N, Kangawa K, and Matsuo H. Neuromedin B: a novel bombesin-like peptide identified in porcine spinal cord. Biochem Biophys Res Commun 114: 541-548, 1983.
- Minamino N, Kangawa K, and Matsuo H. Neuromedin C: a bombesin-like peptide identified in porcine spinal cord. Biochem Biophys Res Commun 119: 14-20, 1984.
- Moghimzadeh E, Ekman R, Hakanson R, Yanaihara N, and Sundler F. Neuronal gastrin-releasing peptide in the mammalian gut and pancreas. Neuroscience 10: 553-563, 1983.
- Moody TW, Pert CB, Rivier J, and Brown MR. Bomebesin: specific binding to rat brain membranes. Proc Natl Acad Sci U S A 75: 5372-5376, 1978.
- Nagalla SR, Barry BJ, Creswick KC, Eden P, Taylor JT, and Spindel ER. Cloning of a receptor for amphibian [Phe13]bombesin distinct from the receptor for gastrin-releasing peptide: identification of a fourth bombesin receptor subtype (BB4). Proc Natl Acad Sci U S A 92: 6205-6209, 1995.
- Namba M, Ghatei MA, Ghiglione M, and Bloom SR. Effects of decapeptide of mammalian bombesin and neuromedin B on pancreatic exocrine secretion in the rat. Digestion 34: 105-114, 1986.
- Nishino H, Tsunoda Y, and Owyang C. Mammalian bombesin receptors are coupled to multiple signal transduction pathways in pancreatic acini. Am J Physiol 274: G525-534, 1998.
- Nuche-Berenguer B, and Jensen RT. Gastrointestinal hormones/neurotransmitters and growth factors can activate P21 activated kinase 2 in pancreatic acinar cells by novel mechanisms. Biochim Biophys Acta 1853: 2371-2382, 2015.
- Ohki-Hamazaki H. Neuromedin B. Prog Neurobiol 62: 297-312, 2000.
- Ohki-Hamazaki H, Iwabuchi M, and Maekawa F. Development and function of bombesin-like peptides and their receptors. Int J Dev Biol 49: 293-300, 2005.
- Ohki-Hamazaki H, Sakai Y, Kamata K, Ogura H, Okuyama S, Watase K, Yamada K, and Wada K. Functional properties of two bombesin-like peptide receptors revealed by the analysis of mice lacking neuromedin B receptor. J Neurosci 19: 948-954, 1999.
- Otani T, Matsukura A, Takamoto T, Seyama Y, Shimizu Y, Shinomiya M, Usui H, Gorelick FS, and Makuuchi M. Effects of pancreatic duct ligation on pancreatic response to bombesin. Am J Physiol Gastrointest Liver Physiol 290: G633-639, 2006.
- Padfield PJ, Garner A, and Case RM. Patterns of pancreatic secretion in the anaesthetised guinea pig following stimulation with secretin, cholecystokinin octapeptide, or bombesin. Pancreas 4: 204-209, 1989.
- Pagani M, Albisetti GW, Sivakumar N, Wildner H, Santello M, Johannssen HC, and Zeilhofer HU. How Gastrin-Releasing Peptide Opens the Spinal Gate for Itch. Neuron 103: 102-117 e105, 2019.
- Pierre JF, Neuman JC, Brill AL, Brar HK, Thompson MF, Cadena MT, Connors KM, Busch RA, Heneghan AF, Cham CM, Jones EK, Kibbe CR, Davis DB, Groblewski GE, Kudsk KA, and Kimple ME. The gastrin-releasing peptide analog bombesin preserves exocrine and endocrine pancreas morphology and function during parenteral nutrition. Am J Physiol Gastrointest Liver Physiol 309: G431-442, 2015.
- Pollack PF. Age and enzyme specificity of the response of developing rat pancreas to the trophic effects of bombesin. Pancreas 4: 101-106, 1989.
- Pooja D, Gunukula A, Gupta N, Adams DJ, and Kulhari H. Bombesin receptors as potential targets for anticancer drug delivery and imaging. Int J Biochem Cell Biol 114: 105567, 2019.
- Powers RE, Grady T, Orchard JL, and Gilrane TB. Different effects of hyperstimulation by similar classes of secretagogues on the exocrine pancreas. Pancreas 8: 58-63, 1993.
- Pralong WF, Wollheim CB, and Bruzzone R. Measurement of cytosolic free Ca2+ in individual pancreatic acini. FEBS Lett 242: 79-84, 1988.
- Qin X, and Qu X. Recent advances in the biology of bombesin-like peptides and their receptors. Curr Opin Endocrinol Diabetes Obes 28: 232-237, 2021.
- Ramos-Alvarez I, and Jensen RT. P21-activated kinase 4 in pancreatic acinar cells is activated by numerous gastrointestinal hormones/neurotransmitters and growth factors by novel signaling, and its activation stimulates secretory/growth cascades. Am J Physiol Gastrointest Liver Physiol 315: G302-G317, 2018.
- Ramos-Alvarez I, Moreno P, Mantey SA, Nakamura T, Nuche-Berenguer B, Moody TW, Coy DH, and Jensen RT. Insights into bombesin receptors and ligands: Highlighting recent advances. Peptides 72: 128-144, 2015.
- Richter K, Egger R, and Kreil G. Molecular cloning of a cDNA encoding the bombesin precursor in skin of Bombina variegata. FEBS Lett 262: 353-355, 1990.
- Rosewicz S, Liehr RM, Solomon TE, Riecken EO, and Logsdon CD. Effects of bombesin on pancreatic digestive enzyme gene expression. Endocrinology 130: 1451-1458, 1992.
- Sancho V, Nuche-Berenguer B, and Jensen RT. The Src kinase Yes is activated in pancreatic acinar cells by gastrointestinal hormones/neurotransmitters, but not pancreatic growth factors, which stimulate its association with numerous other signaling molecules. Biochim Biophys Acta 1823: 1285-1294, 2012.
- Sans MD, and Williams JA. Calcineurin is required for translational control of protein synthesis in rat pancreatic acini. Am J Physiol Cell Physiol 287: C310-319, 2004.
- Sans MD, Xie Q, and Williams JA. Regulation of translation elongation and phosphorylation of eEF2 in rat pancreatic acini. Biochem Biophys Res Commun 319: 144-151, 2004.
- Scarpignato C, Varga G, Dobronyi I, and Papp M. Effect of a new potent CCK antagonist, lorglumide, on caerulein- and bombesin-induced pancreatic secretion and growth in the rat. Br J Pharmacol 96: 661-669, 1989.
- Scemama JL, Zahidi A, Fourmy D, Fagot-Revurat P, Vaysse N, Pradayrol L, and Ribet A. Interaction of [125I]-Tyr4-bombesin with specific receptors on normal human pancreatic membranes. Regul Pept 13: 125-132, 1986.
- Shimosegawa T, Asakura T, Kashimura J, Yoshida K, Meguro T, Koizumi M, Mochizuki T, Yanaihara N, and Toyota T. Neurons containing gastrin releasing peptide-like immunoreactivity in the human pancreas. Pancreas 8: 403-412, 1993.
- Singh P, Draviam E, Guo YS, and Kurosky A. Molecular characterization of bombesin receptors on rat pancreatic acinar AR42J cells. Am J Physiol 258: G803-809, 1990.
- Spindel ER, Chin WW, Price J, Rees LH, Besser GM, and Habener JF. Cloning and characterization of cDNAs encoding human gastrin-releasing peptide. Proc Natl Acad Sci U S A 81: 5699-5703, 1984.
- Susini C, Estival A, Scemama JL, Ruellan C, Vaysse N, Clemente F, Esteve JP, Fourmy D, and Ribet A. Studies on human pancreatic acini: action of secretagogues on amylase release and cellular cyclic AMP accumulation. Pancreas 1: 124-129, 1986.
- Szalmay G, Varga G, Kajiyama F, Yang XS, Lang TF, Case RM, and Steward MC. Bicarbonate and fluid secretion evoked by cholecystokinin, bombesin and acetylcholine in isolated guinea-pig pancreatic ducts. J Physiol 535: 795-807, 2001.
- Uhlemann ER, Rottman AJ, and Gardner JD. Actions of peptides isolated from amphibian skin on amylase release from dispersed pancreatic acini. Am J Physiol 236: E571-576, 1979.
- Upp JR, Jr., Poston GJ, MacLellan DG, Townsend CM, Jr., Barranco SC, and Thompson JC. Mechanisms of the trophic actions of bombesin on the pancreas. Pancreas 3: 193-198, 1988.
- Varga G, Papp M, Dobronyi I, and Scarpignato C. Effect of bombesin and its mammalian counterpart, GRP, on exocrine pancreas in the rat. Digestion 41: 229-236, 1988.
- Varga G, Reidelberger RD, Liehr RM, Bussjaeger LJ, Coy DH, and Solomon TE. Effects of potent bombesin antagonist on exocrine pancreatic secretion in rats. Peptides 12: 493-497, 1991.
- Von Schrenck T, Heinz-Erian P, Moran T, Mantey SA, Gardner JD, and Jensen RT. Neuromedin B receptor in esophagus: evidence for subtypes of bombesin receptors. Am J Physiol 256: G747-758, 1989.
- von Schrenck T, Wang LH, Coy DH, Villanueva ML, Mantey S, and Jensen RT. Potent bombesin receptor antagonists distinguish receptor subtypes. Am J Physiol 259: G468-473, 1990.
- Wada E, Watase K, Yamada K, Ogura H, Yamano M, Inomata Y, Eguchi J, Yamamoto K, Sunday ME, Maeno H, Mikoshiba K, Ohki-Hamazaki H, and Wada K. Generation and characterization of mice lacking gastrin-releasing peptide receptor. Biochem Biophys Res Commun 239: 28-33, 1997.
- Wang QJ, Knezetic JA, Schally AV, Pour PM, and Adrian TE. Bombesin may stimulate proliferation of human pancreatic cancer cells through an autocrine pathway. Int J Cancer 68: 528-534, 1996.
- Wisner JR, Jr., Ozawa S, and Renner IG. Pancreatic exocrine function in unconscious rats treated with submaximal, maximal, and supramaximal doses of bombesin tetradecapeptide. Pancreas 4: 83-89, 1989.
- Younes M, Wank SA, Vinayek R, Jensen RT, and Gardner JD. Regulation of bombesin receptors on pancreatic acini by cholecystokinin. Am J Physiol 256: G291-298, 1989.
- Zhu WY, Goke B, and Williams JA. Binding, internalization, and processing of bombesin by rat pancreatic acini. Am J Physiol 261: G57-64, 1991.