Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2014.13
Introduction
Intraductal papillary mucinous neoplasm (IPMN) may have distinct pancreatic ductal adenocarcinoma (PDAC) in the same pancreas and the incidence of this has been reported to range from 2.0 to 9.9% (7, 9, 11, 13, 16, 20-22, 24, 25); therefore, IPMN is expected to become a good predictor for the early detection of PDAC. However, most PDAC occurring concomitant with IPMNs have been still diagnosed as advanced stage cancer (7, 9, 11, 13, 16, 20-22, 24, 25). Thus, it is urgent to establish an effective screening system to detect early stage PDAC during the management of IPMNs. In this contribution, we describe the clinical characteristics of PDAC concomitant with IPMN along with the diagnostic tools and molecular aspects which predict the potential development of distinct PDAC, and then describe our recent approach to the early diagnosis of PDAC concomitant with IPMN.
Clinical characteristics of PDAC concomitant with IPMN
PDAC concomitant with IPMN is usually defined as occurring when the lesion is separated from the IPMN by an uninvolved segment of pancreatic duct, and there is no transition area from adenoma to carcinoma in the distinct PDAC (5, 8). However, it is often difficult to distinguish PDAC concomitant with IPMN from invasive carcinoma derived IPMN (25).
IPMNs are morphologically classified into two types, main duct IPMN (MD-IPMN) and branched duct IPMN (BD-IPMN), and concomitant PDACs are frequently found at the initial assessment of BD-IPMN or during surveillance of BD-IPMN with or without resection (19). Interestingly, most cases of BD-IPMN having concomitant PDAC are benign (5). On the other hand, we have recently reported 3 cases of MD-IPMN having concomitant PDAC (18), and therefore, both BD- and MD-IPMNs are the risk factors for the development of concomitant PDAC. We previously reviewed the clinical data of 236 patients with IPMNs including 22 synchronous or metachronous distinct PDACs and found that worsening diabetes mellitus and abnormal serum CA19-9 level were the independent predictors for the presence of PDAC concomitant with IPMN (7). However, the problem is that the most patients having such predictors are diagnosed as having advanced stage distinct PDAC.
Diagnosis of PDAC concomitant with IPMN
Standard diagnostic modalities during initial assessment of IPMNs include enhanced computed tomography (CT), magnetic resonance imaging / cholangiopancreatography (MRI / MRCP), and endoscopic ultrasonography (EUS), as described in the international consensus guidelines 2012 for the management of IPMN (19). On the other hand, a standard surveillance protocol after resection of IPMN or for IPMN observed without resection remains undetermined. We have been using alternate CT and MRCP at a 6-month intervals for the surveillance after resection of non-malignant IPMN or of benign-looking IPMN observed without resection; however, we sometimes experienced a patient found to have an unresectable concomitant PDAC with distant metastasis (13, 17), indicating the requirement for more sensitive diagnostic tools or shorter intervals of the examination for the early diagnosis of concomitant PDAC.
Kamata et al.(9) have recently conducted a prospective surveillance study of 102 benign-looking IPMNs and reported metachronous development of 7 concomitant PDACs (7%). In this report, they showed the utility of EUS for the early detection of concomitant PDACs which were not diagnosed by CT and MRI / MRCP. We also found 23 synchronous or metachronus PDACs occurring in 20 patients in a series of 179 patients who underwent resection of IPMNs (14). Seven of the 23 PDACs (30%) were of early stages, i.e., stage 0 to I according to Japanese General Rules for Pancreatic Cancer (Table 1) (8). Sensitivities of CT, MRI / MRCP, and EUS to detect the stage 0 to I concomitant PDACs were 16%, 29%, and 29%, respectively, while sensitivity of endoscopic retrograde pancreatography (ERP) / pancreatic juice cytology was 86% (14). Of note, 3 early-stage PDACs were diagnosed only by ERP / cytology (Figure 1, 2), indicating that ERP has an important role in the early diagnosis of distinct PDACs in patients with IPMNs (14).
We also reported the usefulness of intraoperative irrigation cytology of the remnant pancreas during partial pancreatectomy for IPMN to detect unexpected concomitant PDAC which could be overlooked by preoperative CT, MRI, or EUS (12). The method of intraoperative irrigation cytology is that, after division of the pancreas, a 4-French Polyvinyl chloride tube is inserted into the main pancreatic duct of the remnant pancreas from the cut edge, and fluid is obtained by saline irrigation through the tube (12, 15) (Figure 3). Even when the first and/or second cytology are positive, the saline irrigation is repeated 3 times. If the third cytology still shows a positive result, then the patient undergo additional resection of a 2-cm segment of the remnant pancreas. If the patient has positive cytology results in the first and second irrigation while a negative result in the third cytology, additional resection will not be performed and the positive results in first and second irrigation are considered as contamination of cancer cells derived from the primary lesion(s) in the resected pancreas. Among 48 patients who underwent intraoperative irrigation cytology, 5 patients had a positive result in third intraoperative irrigation cytology, and all of these patients had concomitant PDACs in the additionally resected specimens, including 4 stage 0 and one stage III PDACs (8, 12) (Table 1).
On the other hand, there was one patient who had development of PDAC in the remnant pancreas 24 months after operation despite the negative intraoperative irrigation cytology result, indicating the possibility of false negative result of this procedure. Thus, although useful, this procedure may not be completely safe to rely on when there is a negative intraoperative cytology result (10).

Figure 1. Early stage pancreatic ductal adenocarcinoma concomitant with intraductal papillary mucinous neoplasm. A. Schema of the location of the intraductal papillary mucinous neoplasms (IPMNs) and pancreatic ductal adenocarcinoma (PDAC). This patient underwent distal pancreatectomy, while IPMN in the pancreas head was left without resection. B. Endoscopic retrograde pancreatography (ERP) reveals that cystic dilation of the branch duct in the head and body of pancreas (open arrows), and stenosis of main pancreatic duct with dilation of its distal side (closed arrow). Brushing cytology of stenotic lesion showed adenocarcinoma. Pathology of resected specimen were low grade dysplasia of IPMN (C), and stage I concomitant PDAC (D). Reprinted from Ref 14 with permission of the publisher.
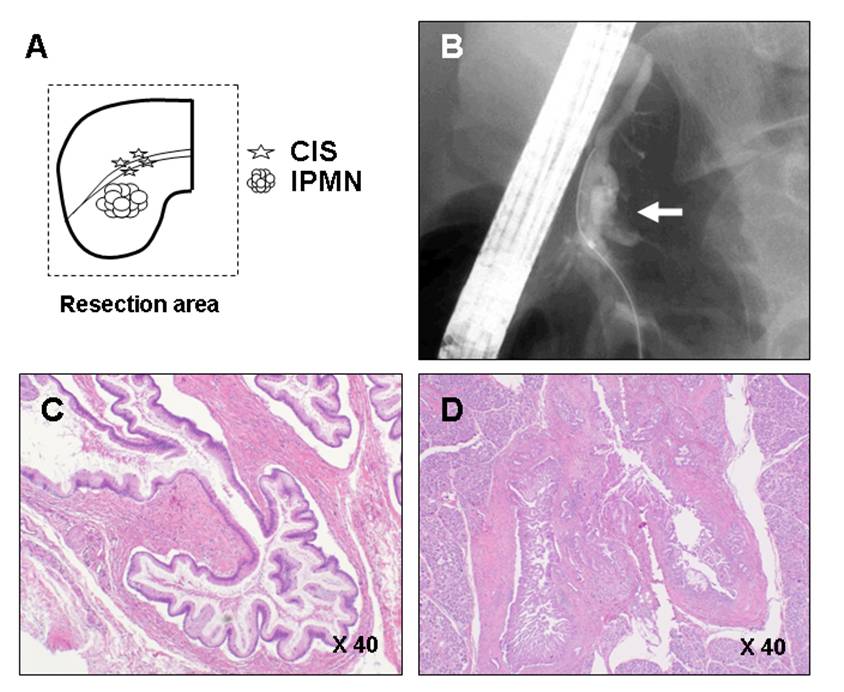
Figure 2. Pancreatic ductal adenocarcinoma concomitant with intraductal papillary mucinous neoplasms detected only by pancreatic juice cytology. A. Schema of the location of the intraductal papillary mucinous neoplasms (IPMNs) and ductal adenocarcinoma in situ (CIS). This patient was the same as demonstrated in Figure 1 and studied of 4 years after the distal pancreatectomy. This patient underwent remnant pancreatectomy for those lesions. CIS was not detected by computed tomography, magnetic resonance imaging, and endoscopic ultrasonography. B. Endoscopic retrograde pancreatography (ERP) reveals that cystic dilation of the branch duct in head of pancreas (open arrows), and there is no abnormality in main pancreatic duct except for the presence of mucin. However, pancreatic juice cytology revealed adenocarcinoma. Pathology of resected specimen were intermediate grade dysplasia of IPMN (C), and stage 0 of non-invasive ductal carcinoma (D). Reprinted from Ref 14 with permission of the publisher.

Figure 3. Intraoperative irrigation cytology of the pancreatic duct. During pancreatoduodenectomy for IPMN in the pancreas head, pancreas is divided at the level of portal vein, and then a 4Fr polyvinyl tube (arrow) is inserted into the pancreatic duct in the remnant pancreas (distal pancreas), and fluid for cytology is obtained by gentle irrigation of 1 to 2mL of saline through a tube with a syringe. Reprinted from Ref 12 with permission of the publisher.

Molecular aspects to predict the development of distinct PDAC
We previously evaluated the expression status of MUC1, MUC2, MUC5AC, MUC6, and CDX2 by immunohistochemical assessment and the mutational status of GNAS in 179 resected IPMNs including PDACs concomitant with IPMNs (5). In that report, occurrence of concomitant PDACs was more frequently observed in gastric-type IPMNs (18/110, 16.4%) than in intestinal (1/49, 2.0%), pancreatobiliary (1/17, 5.9%), or oncocytic-type (0/3, 0%) (p<0.05) (5). PDACs with IPMNs frequently had positive immunostaining for MUC1, MUC5AC, and MUC6, but were negative for MUC2, and CDX2 (5). Mutation of GNAS within codon 201 was not detected in 5 of 6 PDACs concomitant with IPMNs (5). Those data indicate that PDACs concomitant with IPMN frequently arise in the pancreas with benign gastric-type IPMN without GNAS mutations (Figure 4).
Challenge to the early diagnosis of PDAC concomitant with IPMN
A surveillance protocol of IPMNs for early detection of concomitant PDACs has not been established yet. As described above, alternate CT and MRI / MRCP at a 6-month interval seems to be insufficient to detect early stage PDAC concomitant with IPMNs (13). The Japan Pancreatic Society is conducting a nationwide prospective surveillance study for BD-IPMN to investigate the real prevalence of the development of concomitant PDAC in patients with BD-IPMN, and to clarify the efficacy of the surveillance protocol using alternate CT and MRI / EUS at a 6-month interval [UMIN000007349]. Although ERP/pancreatic juice cytology seems to have an important role to detect early stage PDAC concomitant with IPMNs, as described above (14), it also has a risk of post-ERP pancreatitis, and thus the international consensus guidelines 2012 (19) do not recommend ERP as a routine diagnostic modality during the assessment of IPMNs. ERP should be applicable only for high-risk patients with IPMN who may have the development of concomitant PDACs.
Other investigators (1) have also made an effort for the early diagnosis of pancreatic cancer by intense surveillance of 216 high risk individuals who had family history of pancreatic cancer (90%, 6.4- to 32 –fold increased risk), family history of ovarian-breast cancer (9%, 3.5- to 5.9-fold increased risk) and Peutz-Jeghers syndrome (132-fold increased risk), and the most frequent prevalent pancreatic lesions during surveillance was small cyst (39%), most of which seemed to be BD-IPMNs. The detection rates of pancreatic abnormalities in these individuals in CT, MRI, and EUS were 11%, 33%, and 43%, respectively. Because of such suboptimal detection rate and radiation exposure in CT, these experts (2) recommended use of MRI / MRCP and EUS at 12 month intervals for baseline screening of high-risk individuals. If there is some abnormalities in the pancreas, then additional examinations such as CT, ERCP, EUS-guided fine needle aspiration cytology should be performed. However, the effect of this surveillance system on the early detection of PDAC remains unclear.
Because 60 to 70% of IPMNs including both invasive carcinoma and non-invasive lesions (low, intermediate to high grade dysplasia) are reported to have GNAS mutation (3, 4, 23), the remaining 30 to 40% of IPMNs without GNAS mutation are considered to be candidates to have the development of concomitant PDACs (5) (Figure 4). If this high-risk group can be detected by a non-invasive examination, then we would intensively survey these high-risk patients, for examples, by ERP / pancreatic juice cytology, to detect early-stage concomitant PDAC. One of the ways is to assess the mutational status of GNAS in the duodenal fluid. Kanda et al. (10) assessed the GNAS mutational status of the duodenal fluid in 78 patients having radiologically diagnosed IPMNs, and found that 50 patients (64%) were judged as having GNAS mutation in the duodenal fluid, although the status of GNAS mutation in IPMNs themselves were not assessed.

Figure 4. Possible high risk group for the development of pancreatic ductal adenoacrinoma concomitant with intraductal papillary mucinous neoplasms. Intraductal papillary mucinous neoplasm (IPMN) of the pancreas with MUC2 negative gastric subtype and GNAS wild type is considered as being high risk for the development of distinct pancreatic ductal adenocarcinoma (PDAC) in the same pancreas. Strict surveillance for this high risk group may contribute to increase in the number of the early detection of the PDAC.

Figure 5. Our approach to detect high risk patients for the development of concomitant pancreatic ductal adenocarcinoma using molecular marker in the duodenal fluid. A. Duodenal fluid is collected using the duodenoscope. B. Endoscopic finding of the duodenum near the duodenal papilla. Yellow-colored duodenal fluid is observed (arrow). C. Duodenal fluid is collected by a syringe through the catheter. D. Presence of GNAS mutation at codon 201 in the duodenal fluid is detected by cast PCR. Our preliminary assessment of GNAS mutation status in the duodenal fluid and matched resected specimen of intraductal papillary mucinous neoplasms (IPMNs) showed that GNAS status were the same between these two samples in 21 of 23 patients (91%) [6]. Therefore, the patients with IPMN who have GNAS wild type in the duodenal fluid may be at risk for the development of the distinct pancreatic ductal adenocarcinoma.
We preliminary evaluated the GNAS mutational status in the duodenal fluid and matched resected specimen of IPMNs, and found that GNAS status were the same between the duodenal fluid and resected specimen in 21 of 23 patients (91%) (6) (Figure 5). Of note, 3 of 4 patients having PDAC concomitant with IPMN showed wild type GNAS in duodenal fluid, and resected IPMN and PDAC (10). Collection of the duodenal fluid seems to be safe and non-invasive, and, therefore, the assessment of GNAS mutational status in the duodenal fluid will be useful to select the high-risk patients, and intensive surveillance for these high-risk patients may lead to more efficient early detection of PDAC, thereby improving the outcomes of the PDACs.
Summary
Patients having IPMNs have been recently recognized as at high-risk to develop concomitant PDAC, while most concomitant PDACs still have been diagnosed as advanced cancers. Therefore, establishment of screening methods to detect real high-risk group for the development of concomitant PDAC in IPMN patients and of surveillance protocol for the early detection of concomitant PDAC are urgently needed. In this chapter, we introduced our approach to the early diagnosis of concomitant PDAC focusing on the usefulness of pancreatic juice cytology under ERP as well as intraoperative irrigation cytology in the remnant pancreas during partial pancreatectomy and the determination of the real high-risk patients with IPMN by assessing GNAS mutation status.
References
- Canto M.I, Hruban R.H, Fishman E.K, Kamel I.R, Schulick R, Zhang Z, Topazian M, Takahashi N, Fletcher J, Petersen G, Klein A.P, Axilbund J, Griffin C, Syngal S, Saltzman J.R, Mortele K.J, Lee J, Tamm E, Vikram R, Bhosale P, Margolis D, Farrell J, Goggins M, American Cancer of the Pancreas Screening (CAPS) Consortium. Frequent detection of pancreatic lesions in asymptomatic high-risk individuals. Gastroenterology 142: 796-804, 2012. PMID: 22245846
- Canto M.I, Harinck F, Hruban R.H, Offerhaus G.J, Poley J.W, Kamel I, Nio Y, Schulick R.S, Bassi C, Kluijt I, Levy M.J, Chak A, Fockens P, Goggins M, Bruno M, International Cancer of Pancreas Screening (CAPS) Consortium. International Cancer of the Pancreas Screening (CAPS) Consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut 62: 339-347, 2013. PMID: 23135763
- Dal Molin M, Matthaei H, Wu J, Blackford A, Debeljak M, Rezaee N, Wolfgang C.L, Butturini G, Salvia R, Bassi C, Goggins M.G, Kinzler K.W, Vogelstein B, Eshleman J.R, Hruban R.H, Maitra A. Clinicopathological correlates of activating GNAS mutations in intraductal papillary mucinous neoplasm (IPMN) of the pancreas. Ann Surg Oncol 20: 3802-3808, 2013. PMID: 23846778
- Furukawa T, Kuboki Y, Tanji E, Yoshida S, Hatori T, Yamamoto M, Shibata N, Shimizu K, Kamatani N, Shiratori K. Whole-exome sequencing uncovers frequent GNAS mutations in intraductal papillary mucinous neoplasms of the pancreas. Sci Rep 1: 161, 2011. PMID: 22355676
- Ideno N, Ohtsuka T, Kono H, Fujiwara K, Oda Y, Aishima S, Ito T, Ishigami K, Tokunaga S, Ohuchida K, Takahata S, Nakamura M, Mizumoto K, Tanaka M. Intraductal papillary mucinous neoplasms of the pancreas with distinct pancreatic ductal adenocarcinomas are frequently of gastric subtype. Ann Surg 258: 141-151, 2013. PMID: 23532108
- Ideno N, Ohtsuka T, Tamura K, et al. Clinical significance of GNAS mutation for invasive pancreatic carcinoma distinct from/derived from intraductal papillary mucinous neoplasm (IPMN) of the pancreas. Pancreas (in press).
- Ingkakul T, Sadakari Y, Ienaga J, Satoh N, Takahata S, Tanaka M. Predictors of the presence of concomitant invasive ductal carcinoma in intraductal papillary mucinous neoplasm of the pancreas. Ann Surg 251: 70-75, 2010. PMID: 20009749
- Japan Pancreas Society. Classification of Pancreatic Carcinoma. 3rd ed, revised in English. Tokyo: Kanehara; 2011.
- Kamata K, Kitano M, Kudo M, Sakamoto H, Kadosaka K, Miyata T, Imai H, Maekawa K, Chikugo T, Kumano M, Hyodo T, Murakami T, Chiba Y, Takeyama Y. Value of EUS in early detection of pancreatic ductal adenocarcinomas in patients with intraductal papillary mucinous neoplasms. Endoscopy 46: 22-29, 2014. PMID: 24218310
- Kanda M, Knight S, Topazian M, Syngal S, Farrell J, Lee J, Kamel I, Lennon A.M, Borges M, Young A, Fujiwara S, Seike J, Eshleman J, Hruban R.H, Canto M.I, Goggins M. Mutant GNAS detected in duodenal collections of secretin-stimulated pancreatic juice indicates the presence or emergence of pancreatic cysts. Gut 62: 1024-1033, 2013. PMID: 22859495
- Maguchi H, Tanno S, Mizuno N, Hanada K, Kobayashi G, Hatori T, Sadakari Y, Yamaguchi T, Tobita K, Doi R, Yanagisawa A, Tanaka M. Natural history of branch duct intraductal papillary mucinous neoplasms of the pancreas: A multicenter study in Japan. Pancreas 40: 364-370, 2011. PMID: 21289527
- Mori Y, Ohtsuka T, Tamura K, Ideno N, Aso T, Kono H, Nagayoshi Y, Ueda J, Takahata S, Aishima S, Ookubo F, Oda Y, Tanaka M. Intraoperative irrigation cytology of the remnant pancreas to detect remnant distinct pancreatic ductal adenocarcinoma in patients with intraductal papillary mucinous neoplasm undergoing partial pancreatectomy. Surgery 155: 67-73, 2014. PMID: 24183345
- Ohtsuka T, Kono H, Tanabe R, Nagayoshi Y, Mori Y, Sadakari Y, Takahata S, Oda Y, Aishima S, Igarashi H, Ito T, Ishigami K, Nakamura M, Mizumoto K, Tanaka M. Follow-up study after resection of intraductal papillary mucinous neoplasm of the pancreas; special references to the multifocal lesions and development of ductal carcinoma in the remnant pancreas. Am J Surg 204: 44-48, 2012. PMID: 21996346
- Ohtsuka T, Ideno N, Aso T, Nagayoshi Y, Kono H, Mori Y, Takahata S, Oda Y, Aishima S, Igarashi H, Ito T, Ishigami K, Nakamura M, Mizumoto K, Tanaka M. Role of endoscopic retrograde pancreatography for early detection of pancreatic ductal adenocarcinoma concomitant with intraductal papillary mucinous neoplasm of the pancreas. J Hepatobiliary Pancreat Sci 20: 356-361, 2013. PMID: 22878836
- Ohtsuka T, Ookubo F, Tanaka M. Chapter 8-Diagnostic investigation using pancreatic juice. In: Tanaka M (ed): Intraductal Papillary Mucinous Neoplasm of the Pancreas. Pp. 103-113, Springer, Tokyo, 2014.
- Sawai Y, Yamao K, Bhatia V, Chiba T, Mizuno N, Sawaki A, Takahashi K, Tajika M, Shimizu Y, Yatabe Y, Yanagisawa A. Development of pancreatic cancers during long-term follow-up of side-branch intraductal papillary mucinous neoplasms. Endoscopy 42: 1077-1084, 2010. PMID: 21120776
- Tamura K, Ohtsuka T, Ideno N, Aso T, Kono H, Nagayoshi Y, Shindo K, Ushijima Y, Ueda J, Takahata S, Ito T, Oda Y, Mizumoto K, Tanaka M. Unresectable pancreatic ductal adenocarcinoma in the remnant pancreas diagnosed during every-6-month surveillance after resection of branch duct intraductal papillary mucinous neoplasm: A case report. JOP 14: 450-453, 2013. PMID: 23846946
- Tamura K, Ohtsuka T, Ideno N, Aso T, Shindo K, Aishima S, Ohuchida K, Takahata S, Ushijima Y, Ito T, Oda Y, Mizumoto K, Tanaka M. Treatment strategy for main duct intraductal papillary mucinous neoplasms of the pancreas based on the assessment of recurrence in the remnant pancreas after resection: a retrospective review. Ann Surg 259: 360-368, 2014. PMID: 23989056
- Tanaka M, Fernández-del Castillo C, Adsay V, Chari S, Falconi M, Jang J.Y, Kimura W, Levy P, Pitman M.B, Schmidt C.M, Shimizu M, Wolfgang C.L, Yamaguchi K, Yamao K, International Association of Pancreatology. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology 12:183-197, 2012. PMID: 22687371
- Tanno S, Nakano Y, Koizumi K, Sugiyama Y, Nakamura K, Sasajima J, Nishikawa T, Mizukami Y, Yanagawa N, Fujii T, Okumura T, Obara T, Kohgo Y. Pancreatic ductal adenocarcinomas in long-term follow-up patients with branch duct intraductal papillary mucinous neoplasms. Pancreas 39: 36-40, 2010. PMID: 19745777
- Tanno S, Nakano Y, Sugiyama Y, Nakamura K, Sasajima J, Koizumi K, Yamazaki M, Nishikawa T, Mizukami Y, Yanagawa N, Fujii T, Obara T, Okumura T, Kohgo Y. Incidence of synchronous and metachronous pancreatic carcinoma in 168 patients with branch duct intraductal papillary mucinous neoplasms. Pancreatology 10: 173-178, 2010. PMID: 20484955
- Uehara H, Nakaizumi A, Ishikawa O, Iishi H, Tatsumi K, Takakura R, Ishida T, Takano Y, Tanaka S, Takenaka A. Development of ductal carcinoma of the pancreas during follow-up of branch duct intraductal papillary mucinous neoplasms of the pancreas. Gut 57: 1561-1565, 2008. PMID: 18477671
- Wu J, Matthaei H, Maitra A, Dal Molin M, Wood L.D, Eshleman JR, Goggins M, Canto M.I, Schulick R.D, Edil B.H, Wolfgang C.L, Klein A.P, Diaz L.A, Jr, Allen P.J, Schmidt C.M, Kinzler K.W, Papadopoulos N, Hruban R.H, Vogelstein B. Recurrent GNAS mutations define an unexpected pathway for pancreatic cyst development. Sci Transl Med 3: 92ra66, 2011. PMID: 21775669
- Yamaguchi K, Ohuchida J, Ohtsuka T, Nakano K, Tanaka M. Intraductal papillary-mucinous tumor of the pancreas concomitant with ductal carcinoma of the pancreas. Pancreatology 2: 484-490, 2012. PMID: 12378117
- Yamaguchi K, Kanemitsu S, Hatori T, Maguchi H, Shimizu Y, Tada M, Nakagohri T, Hanada K, Osanai M, Noda Y, Nakaizumi A, Furukawa T, Ban S, Nobukawa B, Kato Y, Tanaka M. Pancreatic ductal adenocarcinoma derived from IPMN and pancreatic ductal adenocarcinoma concomitant with IPMN. Pancreas 40: 571-580, 2011. PMID: 21499212


