Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2015.9
| Attachment | Size |
|---|---|
| 902.16 KB |
1. Introduction
Chronic pancreatitis is an on-going fibro-inflammatory process that causes patchy loss of acinar cells while at the same time favouring the formation of intraductal calcium carbonate stones. It often presents as an attack that is indistinguishable from acute pancreatitis, representing paralysis of apical exocytosis in acinar cells, ‘pancreastasis’ (17, 19, 34), which in animal experiments is tied in with a burst of electron transfer reactions (loosely called free radical activity, FRA) (19, 129). Until all secretory parenchyma is obliterated, agonizing pain is usually the predominant symptom, whether accompanying recurrent attacks, or constant and disabling. Its treatment is largely empirical, such that addiction to narcotic analgesics is a compounding menace, because there is no consensus on disease pathogenesis (34). The concept that electrophilic stress is the detonator and inflammatory motor (23, 24) offers the opportunity for corrective micronutrient therapy (11, 20, 30, 66, 140, 154) and, thereby, pain control (28). This usage of micronutrients exploits more than ‘antioxidant’ properties.
2. Stresses and Stressors
Electrophilic stress
The phrase indicates the threat when electrophilic compounds (ie with a relative electron deficit) steal electrons from nucleophiles, as are most biological macromolecules. Xenobiotics (ie exogenous lipophilic substrates) are the major pathological source of electrophiles, by way of reactive xenobiotic species (RXS) that are inadvertently generated upon processing by cytochrome P450 monooxygenases (CYP). Highly reactive carbonyl products derived from oxidation of polyunsaturated fatty acids in cell membranes are the most relevant endogenous source (21).
Oxidative stress
This descriptor points to the threat from an unusually high concentration of reactive oxygen species (ROS), of which many are free radicals (ie. with an unpaired electron) (111). The best known are superoxide (O2- •) which is quenched by superoxide dismutase, hydrogen peroxide (H2O2) which is removed by catalase and glutathione (GSH) peroxidase, and the highly reactive hydroxyl radical (OH•) - leaving aside products of interaction with nitric oxide (46, 87). About 10% of molecular oxygen undergoes ROS-yielding stepwise reduction during such physiological processes as mitochondrial respiration, CYP-mediated processing of endogenous lipophilic compounds, phagocytosis, and synthesis of disulphide (S-S) bonds from cysteine that are needed for proper protein folding in the endoplasmic reticulum (ER) (143). Evidently cells can tolerate the burden, deliberately allowing low-grade oxidative stress for the cited and many other vital roles including signal transduction, calcium homeostasis, membrane turnover, redox control and genomic stability. A pathological excess of ROS - as is associated with CYP induction (65), ultraviolet irradiation, xanthine oxidase activity under conditions of ischaemia-reperfusion, and so on - threatens cell viability by jettisoning just those homeostatic mechanisms that physiological oxidative stress secures (21). Transition metals, iron in particular, promote electron transfer reactions (67). Insofar that ROS are integral to CYP function, the degree of electrophilic stress might be thought to mirror oxidative stress, but studies in the context of ageing show that the level of electrophilic stress can be disproportionately greater than that of its oxidative drive (179).
Reductive stress
This idiom describes abnormally high electron (reducing) pressure behind a blockade of an enzymic step in the ATP energy production staircase. The blockade may be due to absence of an enzyme, or to malfunction. When electron pressure is sufficiently high some of the electrons may react with O2 directly and generate ROS. Swings in electron pressure (redox potential) mimic and are reciprocally linked to swings in pH (proton pressure) (30, 58, 59). In fact, just as alkalosis is rarely if ever a problem unless deliberately induced because all metabolic processes tend to be acid-generating, so too reducing pressure / reducing stress seems to be the main route to oxidative stress, at least in the long term. The problem is epitomised by alcoholism, hypoxia, redox cycling compounds such as doxorubicin that cause electron dislocation, and uncouplers of electron flow, such as NSAIDs, cyclosporine, and cytokines.
Endoplasmic reticulum stress
If not quickly rectified, any of the above stresses activates the ER stress- unfolded protein response (UPR) which, in turn, exacerbates oxidative stress and elicits inflammation by activating stress response genes such as NF-kB (9, 122). The exocrine pancreas with its huge rate of protein synthesis is particularly vulnerable when subjected to congestion in the busy protein-trafficking lanes - an inevitable consequence of pancreastasis episodes, despite the acinar cell’s best efforts to compensate by endocrine re-routing of newly synthesised enzymes; removal of zymogen granules via the three-pronged strategy of centripetal dissolution, crinophagy and basolateral redirection; and down-regulation of enzyme synthesis (19, 30, 34, 52). The close integration between oxidative, electrophilic, ER, and inflammation stress is now regarded as the basis for many chronic diseases (177) and, increasingly, for chronic pancreatitis (97, 112, 123).
3. Electrophilic Stress Template
Component clauses
Since it was first mooted in 1983 (15), this disease model has evolved in line with new observations (16, 23). The 1998 version (24) views the acinar cell as the site of mounting electrophilic stress that steadily erodes methyl (CH3) and thiol (SH) - principally glutathione (GSH ) - moieties, as a result of CYP induction, concurrent exposure to a toxicant that yields RXS, and insufficiency of refurbishing micronutrients (Figure 1). A fourth factor must now be built into the equation, namely, gene mutations that might favour the cytoplasmic presence of trypsin (97): the enzyme, as also chymotrysin, is readily inhibited by GSH via SH-SS exchange (24) should it break loose of constraint by SPINK1 (serine protease inhibitor Kazal type 1) (118), but less GSH is then available for control of electrophilic / oxidative stress and other vital roles (24, 30, 162). The qualifying clauses help to explain why patients on CYP-inducing anticonvulsant drugs rarely develop chronic pancreatitis, or why profound electrophilic / oxidative stress but with low CYP activity in children with kwashiorkor results in painless loss of acini, not chronic pancreatitis (64).

Figure 1. A framework for the pathogenesis of chronic pancreatitis, showing how risk factors interact to generate electrophilic / oxidative stress while also promoting lithiasis.Note that the supply of critical micronutrients might be subnormal in absolute terms - due to inaffordability of source foodstuffs (eg in Soweto), problems associated with senility (22), or hostile culinary practices (eg as in India, resulting in destruction of ascorbic acid) - or relative to increased oxidant load. Abbreviations: C18:2, oils rich in bi-unsaturated fatty acids; CYP, cytochrome P450 mono-oxygenases; PRSS1, SPINK1, CFTR, mutation(s) in genes for cationic trypsinogen, the serine protease inhibitor Kazal type 1, and the cystic fibrosis transmembrane conductance regulator, respectively; ROS reactive oxygen species; RXS reactive xenobiotic species; GSH, glutathione in bioactive form; CH3 activated methyl groups; Vit C (AA) the bioactive ascorbic acid form of vitamin C; GP-2, secreted component of zymogen granule membranes analogous to the renal cast protein (34); PAP, pancreatitis associated protein activated by electrophilic stress (34). Encircled plus or minus symbols represent increases or decreases, respectively.
The concept does allow for a steady build-up of ROS alone, as in elderly people (22), and patients with hereditary pancreatitis (57, 98, 162).
Within this framework, each burst of electron transfer reactions hinders apical exocytosis to trigger an attack of pancreatitis by interfering with the methionine-to -GSH metabolic pathway, which interacts closely with ascorbate and selenium. The diversion of free radical oxidation products (FROP) into the interstitium causes mast cells to degranulate (Figure 1) (25, 34), thereby provoking inflammation, the activation of nociceptive mechanisms that promote a chronic pain syndrome (28), and pro-fibrotic interactions: here it is worth noting that RXS (including from opiates), bile salts and radiocontrast media evoke a non-IgE anaphylactoid response (25). Meanwhile, the acinar cell generates its own pro-inflammatory mediators under the influence of redox-sensitive signalling cascades (87), but pancreatitis is said not to ensue when basolateral exocytosis is prevented (52).
Cystic fibrosis, usually due to severe mutation in bothalleles of the cystic fibrosis transmembrane conductance regulator (CFTR) gene, causes an accelerated non-calcific form of chronic pancreatitis that begins in utero: oxidative stress and inflammation are now regarded as integral features of the disease, driven by unfolded CFTR via the ER stress-UPR system (121, 177). This is not the position depicted in Figure 1, which instead seeks to understand the increased frequency of CFTR mutation(s), with or without mutation in SPINK1, among patients with idiopathic chronic pancreatitis (97), especially the tropical variant (102).
Thus, neonatal hypertrypsinogenaemia in CFTR carriers, and the enhanced susceptibility to experimental pancreatitis so conveyed, suggests hindrance to CFTR-facilitated apical exocytosis in the acinar cell under conditions of excessive FRA (30). Moreover, as predicted (24), CFTR is easily inactivated by oxidants (30), which would have the same impact as pancreas-selective mutations in CFTR (85), compromising the delivery via ductal cells of bicarbonate into pancreatic juice and, thereby, contributing to lithogenicity (34, 81). The ability of the antioxidant curcumin to rescue DF508-CFTR localization in cell lines (88) suggests that oxidants might be responsible for the cytoplasmic mislocalization of CFTR observed in alcoholic, idiopathic and autoimmune pancreatitis (81). Of interest, the CFTR channel also transports the antioxidants GSH (30, 121) and thiocyanate (174).
The template (Figure 1) envisages permutations and combinations among the aforesaid factors plus oxidant attack on CFTR in ductular epithelium as determining outcome - whether recurrent acute pancreatitis, small-duct chronic pancreatitis, or large-duct disease with or without calculi; age at onset; and rate of progression. The worst combination appears to be in patients with tropical pancreatitis (30, 102, 103). The populist notion of pancreatic autodigestion by prematurely activated trypsin in acinar cells has no part in the philosophy (17, 25, 26, 118), and is increasingly challenged by its former proponents (124). Although not in the schema, it is conceivable that RXS via CYP might be involved in the genesis of autoimmune pancreatitis, as in autoimmune hepatitis (60): this becomes plausible with the finding from studies in hepatocytes that newly synthesised CYP enters the secretory pathway to arrive at the outer surface of the plasma membrane (119). As to a connection with ER stress, many xenobiotics have been shown to influence the UPR signalling route, with either pro-survival or pro-death features - unsurprising given that resident CYP straddle ER membranes (84).
CYP induction / Concurrent toxicants
The liver is the main site of xenobiotic processing by CYP although since around 1986 it has become increasingly evident that many organs including the pancreas have the archaic, dormant but inducible machinery (16, 21). The detoxification of xenobiotics and excretion of hydrophilic metabolites is brought about by an initial oxidative step that utilises ROS, followed by conjugation of the intermediate metabolite with glucuronic acid, inorganic sulphur, acetyl groups or GSH via GSH transferases (GST) (70, 154). The phenomenon of ‘enzyme induction’ ensures increased availability of the particular CYP isoform that is appropriate for the substrate in question. This is accomplished by increased synthesis of haem for incorporation into CYP and haem oxygenase (90). The latter degrades excess toxic haem with release of ferritin, bilirubin (via biliverdin) and carbon monoxide. Haem oxygenase is up-regulated by numerous other stressors that share a capacity to decrease tissue GSH. The enzyme is carried in blood and is a potent antioxidant, as also are the catalytic end-products: moreover, it strongly inhibits mast cells (27). Membrane lipids are integral to proper CYP function (21). So too is the trace element selenium, a deficit of which causes hepatic haem to be wasted down the bilirubin route (47).

The information in Table 1 is a distillate of many studies in patients with chronic pancreatitis, itemised in earlier reviews (16, 18, 23, 34), now incorporating more recent observations in patients with mainly alcoholic disease (138, 5). The findings are readily rationalized by the electrophilic stress concept (Figure 1), but not by any other theory on pathogenesis (34). The information reveals that induction of the xenobiotic-processing machinery in liver and pancreas is not innocuous, despite mobilization of several natural antioxants (67, 111). Studies on surgically resected specimens afford direct evidence of CYP induction (55, 147, 168), and also on-going oxidative stress : structural aberrations by microscopy (Table 1) (24), FRA signals (155), increased FROP with decreased GSH (5, 133), increased concentrations of pro-oxidant metals (copper, iron) but decreased levels of antioxidant metals (zinc, selenium) (5); and markers of the ER stress-UPR (123). The last finding might be expected to involve disrupted calcium homeostasis (177), but studies in isolated rat acini indicate that this is not a factor in toxicity from induced CYP (37). As to whether oxidative stress contributes to the sclerosing ductal lesions, its involvement in primary sclerosing cholangitis is worth registering (43), in that similar lesions are not infrequently present in patients with ordinary chronic pancreatitis (18). Moreover, pancreatic juice (130), bile (86), and duodenal aspirates (56, 68) from patients with chronic pancreatitis have high concentrations of irritant lipid oxidation products.
The key point is that the pancreas falls clinical victim while liver injury is generally silent - but why? The best explanation is that xenobiotics hit the gland directly via the arterial route, whereas ingested toxicants first encounter the liver which is best equipped to deal with RXS, via its huge complement of GSH and GST (21, 170). This is illustrated by experimental studies in the 1950s using a subcutaneous dose of carbon tetrachloride, which is processed by CYP to yield RXS : damage in advance of liver injury; lesions that could be “produced at will” by varying the dose, ranging from patchy lesions of early chronic pancreatitis with or without concretions, through to ‘pancreatic cirrhosis’ or a cystic fibrosis- like picture (165). The theme is reinforced by more recent studies with nitriles akin to those in dietary cassava (manioc, tapioca), and the occupational chemical dibutyltin (170), showing that prior induction of CYP2E1 by a small dose of ethanol augments dibutyltin injury (101) - as is also true for hepatotoxicity from volatile hydrocarbons (131, 149). As to chronic exposure to ethanol, laboratory studies show that increased FRA precedes pancreatic injury (71). In the drug metabolism studies from Manchester, UK (Table 1), heightened theophylline clearance was the predominant finding in patients with idiopathic or alcoholic disease (18), indicating induction of CYP1A (70) - as by C18:2 fatty acids (eg in corn, peanut or linseed oil), polycyclic aromatic hydrocarbons and halogenated hydrocarbons (94).
Since a proportion of arterial blood first enters islets cells, it is likely that RXS generated in CYP-induced islets are delivered to some acini by the insulo-portal conduit, so adding to their RXS burden, and potentially explaining the patchy distribution of lesions (24, 147, 165). The abundance of GST in islet cells affords insurance against injury in the short term, whereas a dearth of GST in acinar cells - whether absolute (161), or relative to increased need from CYP induction (55, 147, 168) - renders them vulnerable (30). Moreover, relatively long-lived FROP and RXS generated in the CYP-induced liver (Table 1) could aggravate pancreatic injury if they find their way there via refluxed bile (128), or the bloodstream (18).
Of all the findings in Table 1, the increase in bilirubin is most revealing because it indicates induction of haem oxygenase to combat severe oxidative stress. A further surge accompanies a pancreatitis relapse (18), mimicking the abrupt enzyme rise when phenobarbitone-treated rats are exposed to RXS from halothane gas (152). The combination of induced CYP1A, increased copper, and induced haem oxygenase is a unique exposé of environmental toxicology in humans (83, 110). The three findings cannot be dismissed as a consequence of impaired pancreatic function because there was no correlation with its degree as gauged by secretin-pancreozymin tests (18). However, the normalization of copper and bilirubin data by long-term treatment with pancreatic extracts (18) is of the utmost interest, now that a paper documenting the antioxidant potential of such extracts has been unearthed (see below). Both bile and pancreatic juice inhibit copper absorption in experimental work, but studies using radioisotopes did not show any difference in copper absorption by healthy volunteers, patients with untreated chronic pancreatitis or those on pancreatic extracts (H Sharma, P Tasker, JM Braganza, unpublished).
Reports from the UK (Manchester) (33, 100), south India (Madras) (32) and south Africa (Soweto) (73, 138) have revealed regular close exposure to volatile hydrocarbons in patients with chronic pancreatitis, whether in the occupational environment, domestic setting (from kerosene or paraffin lamps and cookers in confined spaces), or both. In the first UK study, patients noted freedom from attacks when away from the workplace (33). The six-fold decline between 1962 and 1987 in annual hospital admissions with the disease in Kerala province, south India, coincides with the introduction of electricity, which removed the dependence on traditional lighting (34). An investigation at Soweto concluded that exposure to occupational chemicals distinguishes patients labelled as ‘alcoholic chronic pancreatitis’ from alcoholic controls with similar cigarette usage and equally poor diet (138). These observations add weight to case reports cited in previous reviews (21, 79, 91). Although the direct pancreatic toxicity of petrochemicals is documented in lower species (21), and hepatotoxicicity from kerosene was recorded some time ago (148), the field of inhalation toxicology to the pancreas has been a vacuum until recent evidence of injury from cigarette smoke in rodents (34).
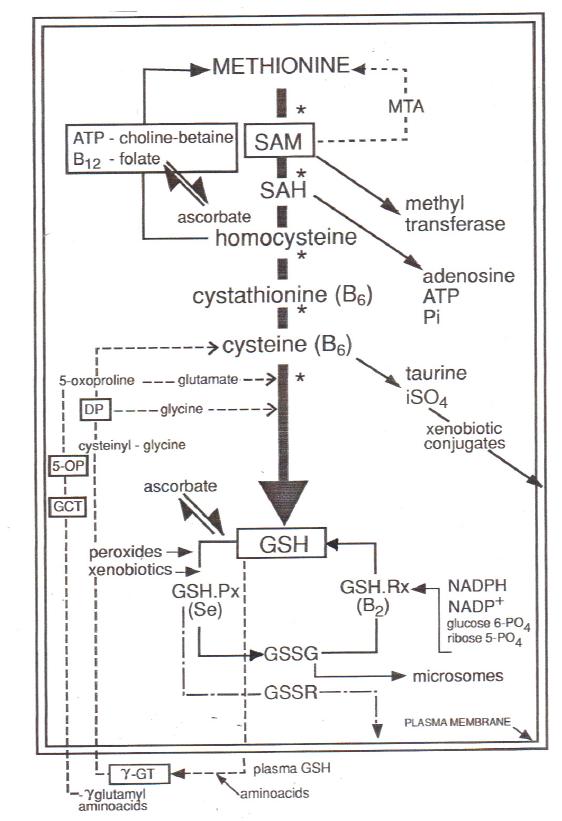
Figure 2. Reproduced from reference (25) with kind permission from the publisher (S. Karger AG, Basel, Germany).The pathway of methionine metabolism wherein key metabolites are SAM, sulphadeosnsyl methionine; SAH, sulph adenosyl homocysteine; and GSH, glutathionine. Other abbreviations: MTA methylthioadenosine; ATP adenosyl triosephosphate; Pi, activated phosphate groups; iSO4, inorganic sulphate; B2, B6, B12, riboflavin, pyridoxine and cobalamin, respectively; GSH.Px, glutathione peroxidase; GSH.Rx, glutathione reductase; Se, selenium; GSSG, oxidised glutathione on engaging with peroxides; GSSR, conjugates of glutathione with electrophiles from xenobiotics; NADPH and NADP, reduced and oxidized forms of nictotinamide adenosine phosphatase, respectively; DP, 5-OP and GCT, enzymes involved in the synthesis of glutathione (170). Asterisks indicate enzymes that are known to be vulnerable to electrophilic / oxidative stress.
This should soon be rectified, scepticism notwithstanding (144), because the health risk from volatile petrochemicals is currently under intense scrutiny (109, 117, 156).
Methyl / Thiol insufficiency
In theory, there are many ways in which a burst of electron transfer reactions can impede apical exocytosis in acinar cells (21), but the evidence in patients with chronic pancreatitis points to a breakdown in the delivery of CH3 and SH moieties (Figure 2) (21, 23-25). The concept of methionine-dependent exocytosis was enunciated in the 1950s (77, 165, 166) and is now pinpointed to methylation of membrane components, probably of a prenylated cysteine residue (40).
The supply of CH3 moieties depends on de novo synthesis from S-adenosylmethionine (SAM) via a folate-dependent enzyme which catalyses the production of S-adenosyl homocysteine (SAH), or by re-methylation of the next metabolite, homocysteine, via choline-betaine and vitamin B12-folate cycles that need ATP and are facilitated by ascorbic acid , the bioactive from of vitamin C (30). Hence folic acid lack inhibits secretion too (6). GSH derived from homocysteine by the transsulphuration pathway is another absolute requirement for apical exocytosis, not least by protecting participating enzymes in the methionine metabolic route (24) while also sparing critical protein thiols (93). Furthermore, in vitro studies identify the need of CFTR for exocytosis (115), while in vivo experiments show that a surfeit of magnesium stabilizes the exocytosis machinery by antagonizing calcium (132).
Both steps in the onward route from homocysteine to cysteine are powered by vitamin B6 (pyridoxine)-dependant enzymes. Pyridoxal-5’-phosphate is also a co-factor for two other enzymes involved in the synthesis of the gaseous mediator hydrogen sulphide (H2S) from homocysteine and cysteine (172), seemingly provoked when the progression to GSH is impeded. Cysteine is pluripotent : rate-limiting component in GSH synthesis, source of taurine and inorganic sulphate that facilitate the removal of RXS (Figure 2), key to proper protein folding in the ER (143), and seemingly even more important than GSH for redox control (105). The same is true for GSH: besides facilitating exocytosis and inhibiting proteases, it helps in redox control, serves as a reservoir for cysteine, mops up hydrogen and lipid peroxides, detoxifies RXS and contributes to the extracellular antioxidant shield. Whereas its utilisation in peroxide control is soon made good via interlocking GSH peroxidase-GSH reductase, NADPH-NADP, and glucose 6 phosphate-ribose 5 phosphate shuttles, it is permanently excreted from cells in conjugates with RXS (Figure 2) (21, 170). In these circumstances, the ability of ascorbic acid to substitute for GSH by redox and non-redox pathways is invaluable (30), as is heightened activity of gamma glutamyl transpeptidase (γGT) in the plasma membrane, which enables the uptake of reconstituting amino acids from the plasma GSH pool (138, 170) - but these resources are finite.
The pathway of methionine metabolism also impacts on the correction of reductive stress by biomolecules with electrophilic methyl groups. These include SAM, phosphatidylcholine, betaine and carnitine (58).They appear to act by binding to positively charged nitrogen or sulphur moieties, a poising mechanism that is demonstrable in vitro when the reaction mix includes catalytically active iron, H2O2 and ascorbic acid: carbon dioxide and carbon monoxide are formed from the ascorbate molecule in parallel with generation of methane gas. It is now recognized that albumin acts as a sacrificial antireductive protein which when modified by OH• radicals emits a signal to proteolytic degradation and elimination (89).
In patients with chronic pancreatitis there is clear evidence of oxidant-associated breakdown in methionine metabolism. Thus, during a relapse, neutrophils show low GSH but an increase in the oxidized form (96), while urine and / or blood analysis point to a metabolic block in the transsulphuration pathway distal to cysteine - leading to surges in cysteine and more proximate metabolites (96, 160), but a fall in inorganic sulphur. By the third day subnormal methionine and a further decline in sulphur levels hint at poor pre-morbid intakes of sulphur amino acids (30). These twin problems of hindrance to methionine metabolism within an oxidative environment and methionine insufficiency are shown in studies of patients with quiescent disease, whether alcoholic or idiopathic. (a) Peripheral blood displays a strong tendency to produce ROS (51, 151, 167). (b) Plasma / serum contains excessive amounts of protein carbonyls (167), and also lipid-based FROP as reported in papers that are too many to cite individually. (c) Erythrocytes have subnormal levels of certain antioxidant enzymes, and GSH (10, 62, 82). (d) Transmethylation and transsulphuration pathways remain fractured (63). (e) 11C methionine scanning demonstrates good pancreatic uptake of the amino acid but then its regurgitation coupled with impaired enzyme secretion into the duodenum (150, 153). (f) Subnormal plasma concentrations are reported of sulphur amino acids (61) and thiols derived via the transsulphuration route (125, 167), including GSH (138, 167). Plasma homocysteine level may remain normal (167), or increase in conjunction with subnormal folic acid (63), vitamin B6 (35) or vitamin B12 (138). (g) H2S appears in exhaled air (107). It is not known whether any of these aberrations has a bearing on displacement of Munc18c into the cytosol of intact acinar cells noted in the resected specimen of a patient with stable disease: this ‘SM protein’ is involved in pathological basolateral exocytosis (52).
4. Towards Treatment
Clues for a prescription
Non-enzymic endogenous defences to electrophilic/oxidative stress are already up-regulated in patients with chronic pancreattis (Table 1), and it is known that the acinar cell has liitle copper-superoxide dismutase (30). The inference is that micronutrient antioxidants fall short in the face of the persisting assault from RXS / ROS (31, 47). Many trace elements, the sulphur amino acids, and several - perhaps all - vitamins contribute in one or more ways to the antioxidant repertoire of tissues (64). Analysis of habitual diets is the only way to glean which items might be crucially lacking in the face of an increased oxidant load, not merely less than in healthy controls. The axiom - which extends to blood levels - cannot be overstated, while appreciating the difficulty in estimating that load (21, 111). Low blood levels reflect the net result of intake, absorption, tissue sequestration and excretion.
Studies at Manchester, UK, identified lower habitual intakes of selenium, vitamin C, riboflavin and vitamin E in patients with idiopathic chronic pancreatitis than in age and gender-matched controls, Selenium was the best discriminator on step-wise analysis, and when examined against theophylline clearance (as marker of CYP1A-related oxidant load) (70), afforded good separation between patients and controls (120). When the studies were extended to a control group with similarly high theophylline clearance - patients with epilepsy on anticonvulsant CYP inducers - a second discriminant function emerged that was equally weighted on lower methionine and vitamin C by the chronic pancreatitis set (158). This finding accords with in vitro studies showing heterosynergism between ascorbic acid and sulphur antioxidants (135). Moreover, whereas the epilepsy group was in a care centre cocooned from environmental toxicants, regular exposure to volatile hydrocarbons was noted in four others who were in employment and developed chronic pancreatitis (158). There are no comparable studies of habitual diets in patients with chronic pancreatitis.
The antioxidant role of selenium is generally linked to its presence at the active centre of enzymes that are redox catalysts, of which the best known are GSH peroxidase which removes H2O2 and lipid peroxides, and thioredoxin reductase which is homologous to GSH reductase and critical for redox regulation of protein function and signalling (4). However, there is evidence that the element serves other important roles in the detoxification of xenobiotics (31). Ascorbic acid is pluripotent in combating electrophilic / oxidative stress. It can substitute for GSH; facilitates the homocysteine remethylation cycle (Figure 2); scavenges electrophiles; acts as ‘Michael donor’ in reactions with acrolein and genotoxic FROP (30); protects against OH• in plasma (67); and quenches mast cell histamine (76) which generates H2O2 (108). Not only does the last factor rationalize the virtual absence of ascorbic acid in plasma samples from patients admitted with a pancreatitis attack (14, 136), but it also underlines the involvement of mast cells (25-27).
Lower concentrations of selenium have been noted in serum / plasma of groups with chronic pancreatitis compared to control groups in diverse geographic areas (5, 31, 106, 116, 138, 163, 164, 175), with subnormal GSH peroxidase activity when selenium level is very low (163). At Manchester the lowest selenium values accompanied painful disease, and levels fell progressively over four days upon repeated exposure to CYP substrates used for drug kinetic studies (31). There is debate as to whether (116), or not (173, 175), malabsorption contributes to the decrease. However, treatment with pancreatic extracts is expected to augment the intake of selenium, zinc, magnesium and methionine, because the gland is a repository of these: the metals should survive the purification procedure and sulphur amino acids might, whereas the vitamins could be lost. This deduction is supported by a hitherto buried study in patients with cystic fibrosis, in whom increases in plasma selenium and erythrocyte GSH peroxidase activity could be attributed to substantial amounts of selenium in commercial preparations (173). The finding has obvious repercussions on usage of pancreatic extracts to ease pancreatic pain in patients with small-duct chronic pancreatitis, indicating micronutrient antioxidant therapy by proxy (29).
Vitamin C assays by spectrophotometric methods do not indicate the percentage in the bioactive ascorbic acid form, as is enabled when an HPLC assay is run in parallel. Thus,the respectable control levels of plasma vitamin C reported in India - at Madras (36), Delhi (10), and Cochin (62) - are misleading if the Madras results can be generalized. Here the samples contained very little ascorbic acid, the discrepancy likely due to hostile culinary practices that might also destroy β-carotene. By contrast, in controls at Soweto the low level of ascorbic acid in plasma was proportionate to that of vitamin C, reflecting inaffordability of fresh fruit and vegetables, and fell further in the oxidizing milieu of chronic pancreatitis (138). At Manchester, ascorbic acid values were negligible in patients with calcific disease or cysts/pseudocysts (20). Against this background, the good value for ascorbic acid reported in French patients seems anomalous (116), and begs the question as to what fraction this represented of total vitamin C.
The triple whammy in the genesis of pancreatic electrophilic stress - CYP induction, concurrent exposure to volatile toxicants, and insufficiency of particular micronutrients - was highlighted by a report on patients with idiopathic disease at Manchester (128). Low protein intake, as in Madras and Soweto, impairs CYP induction: thus, the rate of theophylline clearance in Madras controls was lower than in Manchester controls, but significantly increased in local non-alcoholic patients (44), alongside regular close exposure to kerosene fumes and little ascorbic acid (36, 103). At the time of the studies at Soweto, it was not known that chlorzoxazone is a probe of CYP2E1 induction, as by ethanol (92): in these patients, with predominantly alcoholic disease, theophylline clearance was similar to that in healthy controls, but the impact of RXS evident from a fall in plasma GSH, increased oxidation of plasma ascorbic acid, decreased urinary inorganic sulphate and increased D-glucaric acid (138). A subnormal concentration of zinc in serum (5, 138) and erythrocytes (62, 116) correlates with reduced exocrine secretory capacity (62), rather than poor intake, and has no bearing on CYP function.
Potential benefit
Treatment with a combination of methionine, vitamin C and selenium should, in theory, help patients with painful chronic pancreatitis by several means - protecting the acinar exocytosis machinery; controlling rogue trypsin; removing RXS from halogenated hydrocarbons (166) and petrochemicals (157 ) ; shielding CFTR (30); curbing NF-kB activation and cytokine production (13); rectifying reductive stress, as by cyclosporine (137); inhibiting ER stress and activation of the UPR (74, 75, 95); reducing the oxidative drive to stellate cells (49); and stabilizing mast cells (26, 28, 29), mediators from which are not only pro-fibrotic, but also are implicated in converting peripheral pain sensitization to the unrelenting pain from central sensitization (28).
Insofar that recurrent acute pancreatitis (ie with histological restitution six or more weeks after the last episode, as gauged by normal secretory and imaging studies (16) and chronic pancreatitis are now regarded as a disease continuum (34), the following areas of overlap as well as subtle differences are interesting. Genetic studies show an increased frequency of CFTR with or without SPINK1 mutationamong patients with pancreas divisum, type-1 hyperlipidaemia or hyperparathyropidism (30). A pilot study disclosed regular exposure to volatile hydrocarbons in patients without gallstones (33). Drug disposal studies indicated induction of CYP and ancillary systems in several of them (18). Analysis of secretin-stimulated duodenal aspirates identified Increased lipid peroxidation, albeit less than in chronic pancreatitis (68). Dietary inventories showed that datapoints lay on or close to the aforementioned discrimination line based on selenium intake versus theophylline clearance (54). Oxidative stress is recognized in the pathogenesis of recurrent pancreatitis due to deficiency of lipoprotein lipase (69). Plasma / serum profiles of micronutrients were within normal limits, in contrast to the deficiency profiles in chronic pancreatitis (106). In other words, there seems to be a better match between the availability of micronutrient antioxidants and the degree of electrophilic / oxidative stress in patients with recurrent acute pancreatitis, despite the need to protect a larger mass of functional parenchyma. However, this might not be the only explanation for why recurrent acute pancreatitis does not always progress to chronic pancreatitis (30, 69, 106, 123, 176).
Pilot studies
In the 1980s there was no commercial preparation that would deliver methionine, vitamin C and selenium simultaneously. Methionine tablets were available from the Evans Medical Ltd, Horsham, UK (to treat paracetamol poisoning, which is caused by CYP-derived RXS), as was a nutraceutical from Wassen International, Leatherhead, UK that would provide the other two micronutrients, but along with β-carotene and α-tocopherol ( ie vitamin E). By trial and error in 23 patients (20) - including five described in case reports of small-duct chronic pancreatitis or large-duct disease without or with huge calculi (128, 158) - total daily doses that most often reduced attack frequency and / or background pain were identified. These were 2 gm methionine (although patients exposed to occupational chemicals tended to need twice as much), 600 μg organic selenium, 0.54 gm vitamin C and, inevitably, also 9000 IU of β-carotene and 270 IU of vitamin E. Side effects were usually minimal (eg. nausea, skin discolouration from β-carotene). Schizophrenia has been reported when methionine dose exceeds 10 gm/day: a patient with a strong family history developed symptoms on 4 gm/day.
A number of exclusion criteria for future placebo-controlled clinical trials could be delineated: suspected pancreatic cancer; over-the counter vitamins / antioxidants; children; pregnant women; pain that could be explained by concurrent illness such as gallstones, peptic ulcer or somatic causes which are neither expected to nor do respond to micronutrient therapy (JM Braganza unpublished); large pseudocysts, or bile duct stones that need invasive intervention; such advanced disease that oxidants have no target (51), as evident from steatorrhoea, secretin tests and nowadays by assay of faecal elastase (29, 34,62); family history of schizophrenia; chronic renal failure or liver failure; addiction to narcotic analgesics in that the associated pain mimics that of chronic pancreatitis and because addicts might have ulterior motives for sickness behaviour (28). Clearly, the earlier that micronutrient therapy starts in patients with (non-gall stone) relapses, the greater should be the chance of success. These exclusion criteria can be relaxed outside trials when regular clinical and biochemical monitoring is possible (99).
Assessing outcome
The goal of treatment is to correct electrophilic / oxidative stress and thereby to control pancreatic pain (28). Hence it stands to reason that there must be evidence of such stress pre-recruitment, or its confident expectation on the basis of numerous published reports - provided that ineligible patients are excluded and that patients do not change their lifestyles for the duration of a trial. Furthermore, any reduction in pain on active treatment should ideally be shown to occur pari passu with correction of such stress, so as to distinguish true improvement from the 20% rate of amelioration by placebo (41). Low plasma/serum level of one or more micronutrients in isolation merely indicates a propensity to oxidative stress. Likewise, increases in plasma levels upon supplementation thereof without reference to oxidant load are not only meaningless, but in healthy controls is without benefit (3), and could be harmful by abolishing low-grade oxidative stress that is so essential physiologically.
In order to assess oxidant load, clinical investigators seek biochemical ‘fingerprints’ of persisting stress. The choice, from the immense library (111), must be guided by the perceived primary target of attack, and practicality. If lipids are the all-important target, the best current marker is F2 isoprostanes (86), and ‘thiobarbituric acid reacting substances (TBARS)’ the least specific albeit most popular. These are products of the classical lipid peroxidation pathway, but there is another route that accounts for the bulk of so-called ‘diene conjugates’ in biological fluids. This is the isomerisation pathway, and is easily mimicked in vitro by irradiating linoleic acid (9, 12 LA) in the presence of albumin. Moreover, the route seems to be under the control of a selenium-dependent enzyme, in that only one isomer (9 cis,11 trans) is present in bile, duodenal juice and serum when four are possible (42, 146), and also that consideration of serum selenium alongside the percent molar ratio of the isomer (9,11 LA’) to the parent fatty acid (% MRLA) enabled good discrimination between data from patients with cystic fibrosis and controls (126). The isomer’s stability under ordinary freezing conditions and ease of batch analysis by automated HPLC makes it an attractive marker, but potential invalidation by food sources and bacterial contamination must be remembered.
As argued recently (28), the triggering attack in pancreatitis is on enzymes and receptors that are protected by ascorbic acid interacting with GSH. Hence, useful measures in plasma/serum might include the percent oxidized ascorbic acid relative to total vitamin C (138); GSH coupled with y-GT activity (138); protein carbonyls (167); and allantoin, signifying oxidation of uric acid which, along with albumin, glucose and bilirubin constitute the bulk antioxidants. Convenient tests that measure ‘total plasma antioxidant activity’ by a commercial kit, or so-called ‘TRAP’ and ‘FRAP’ assays that have been used in clinical trials (10, 80), are misleading in monitoring micronutrient therapy because they are largely influenced by those compounds : ascorbic acid contributes 20% to the FRAP reading, vitamin E 5%, β-carotene very little and thiols none (30). If a nomogram is available to monitor treatment (126, 160), so much the better. Since dysregulated methionine metabolism due to RXS seems to underlie most cases of chronic pancreatitis, an index of its repair by treatment would be very helpful - as by analysing a metabolite(s) (63, 96, 160) , and / or 11C methionine scanning (150,153). These resources are scarce, but a sustained increase in erythrocyte GSH upon micronutrient therapy appears to be an indirect pointer (10). It is generally accepted that the identification of oxidative stress and its correction should involve more than a single index of attack on a single target (21).
As to clinical recording, exclusion criteria should be specified, as also criteria for diagnosing chronic pancreatitis, recognizing that minimal changes on endoscopic pancreatography or endoscopic ultrasonography are insufficient without a test of secretory capacity (34). The frequency of attacks, pain scores using visual analogue scales for the most common local descriptors of pain, the best available quality-of-life measure that befits pancreatic pain (139), and analgesic usage are indices of therapeutic efficacy, or otherwise. Questionnaires should be kept to a minimum and applied by the same clinician, given that these patients co-operate voluntarily although they may be in much pain. Monitoring of compliance is best achieved by an objective measure: since the detection of ascorbic acid by urinary dipstick is not quantitative, compliance might only be determined retrospectively when the results of blood analysis in the post-active treatment phase are known. The wide inter-individual variability in disease pattern favours a switch-over trial design which, however, is foiled by the carry-over effect of today’s high potency material eg. ‘Antox’ (Pharmanord, Morpeth, UK), although the average daily dose of selenium is half that used initially (134).
5. Clinical Trials
Meta-analyses
The first independent appraisal of antioxidant therapy for pain control in patients with
chronic pancreatitis covered reports on randomized controlled trials (RCT) up to 2009, and concluded that the identified micronutrient combination ( Section 3) improved outcome in each of three placebo-controlled studies (159, 80, 10), but that meta-analysis was impossible because different instruments were used to measure pain (104). In the past 12 months, three meta-analyses have appeared, covering studies until October ’12 (1), December ’12 (39), and February ’14 (178). Despite the indiscriminate inclusion of RCTs judged satisfactory on mechanistic grounds, without considering the basis for treatment or legitimacy, each report concluded that active treatment reduced pain, especially using the micronutrient combination (39), and that although side-effects in up to 19% were usually mild ( eg headache, nausea, allergy, constipation, diarrhoea), they did cause some patients to withdraw from the trial. The first trial, published in 1990 (159), was the only one to tick all the boxes in the report under the Cochrane banner (1) : an attempt to gauge quality-of-life used questionnaires based on patients with chronic backache, inappropriate in retrospect but the best available at the time. Interestingly, reports that were deemed fit for inclusion were not the same in these meta-analyses. All authors called for further large-scale studies.

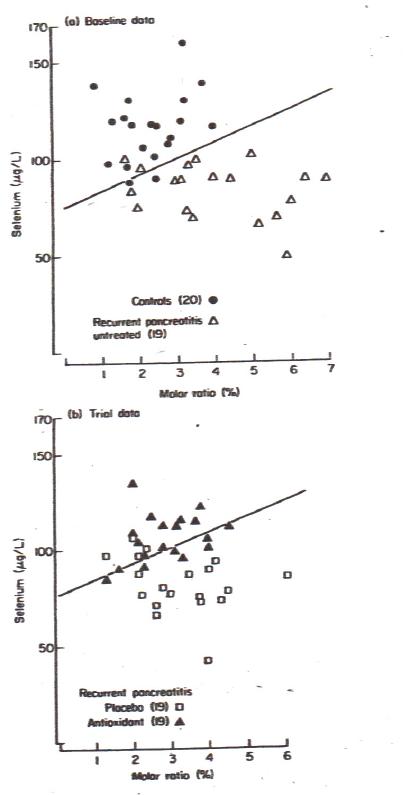
Figure 3. Reproduced from reference (160) with kind permission from the publisher (Wiley, West Sussex).Discriminant analysis of serum selenium and the % molar ratio of the 9 cis 11 trans isomer of linoleic acid to the parent compound in controls and patients with recurrent pancreatitis. Discriminant function: selenium= 8.76 x % MR + 75.74. Note that this function also applies to data from placebo-treated patients but fails to distinguish between controls and antioxidant-treated patients. Moreover, the discriminant function is not dissimilar to that recorded in patients with cystic fibrosis (126).
Since the raison d’etre for a micronutrient prescription is to boost the supply of CH3 and SH moieties (Section 3), the inefficacy of the micronutrient curcumin (despite curbing lipid peroxidaton and being a potent inhibitor of mast cells) and allopurinol (inhibitor of xanthine oxidase) in clinical trials indicates that these substances do not achieve the goal: of note, curcumin did not increase GSH in erythrocytes. Yet, the curcumin trial (53) was included in each meta-analysis, and the allopurinol trial (8) in two (1, 39). The Cochrane report (1) correctly separated a study showing the benefit of allopurinol or another antioxidant, dimethyl sulfoxide, in patients with a pancreatitis relapse (127), wherein many other factors such as pancreatic ischaemia come into play (23, 25), but was included in the other two appraisals (39, 178). However, that analysis included two studies published in abstract only (1), and a Polish language report of vitamin C / E treatment versus no treatment (ie. not the full micronutrient package) (72), which was also included in another meta-analysis (178).
Subjective assessments during this period concluded that micronutrient antioxidant therapy was convincing (11, 154); had potential (66, 140); could be useful as adjuvant therapy (45); was poor, based on very limited experience (38); or useless (113).
Micronutrient combination
Table 2 summarisesinformation on studies of the micronutrient combination, whether (10, 50, 80, 145, 159, 160) or not (48, 162) placebo-controlled, excluding studies reported only in abstract. In the original cross-over trial clinical improvement was accompanied by migration of datapoints towards the control zone in the nomogram relating serum levels of a lipid oxidation marker and selenium (Figure 3) (160). Amelioration of oxidative stress concurrently with clinical improvement was shown in two other trials (10, 50), of which one also noted reduction in markers of fibrosis (50). Clear benefit from active treatment accrued in all but one trial (145), although its authors argued strongly in favour just two years earlier (Table 2), in a cross-sectional study of patients already on micronutrient supplements versus no supplements under a later policy (see below) (139). Unfortunately, serious flaws render the second report invalid (28), while diluting the value of micronutrient therapy in each meta-analysis.
Of special note in Table 2 is the observation that the combination of SAM (instead of methionine) (Figure 2), plus vitamins A, C, E, and magnesium was beneficial in three children with hereditary pancreatitis (162). Combination therapy was also highly successful in abolishing attacks in patients with lipoprotein lipase deficiency (69), interesting because in this instance xanthine oxidase rather than induced CYP is the likely source of increased FRA (23). Intravenous treatment with N-acetylcysteine - in lieu of methionine - as a more immediate precursor of GSH (Figure 2), plus the other micronutrients by appropriate routes, resulted in rapid pain relief and contraction of the inflammatory calcific head mass in an emaciated patient with impedence to gastric outflow and silent haemochromatosis (142).
Two RCTs are in progress : NCT01528540 is testing the micronutrient cocktail plus pregabalin ( a presynaptic voltage-gated blocker of the calcium channel) or placebo in all-comers; EUROPAC-2 is a three-armed trial involving a potent commercial preparation of the antioxidant combination (‘Antox version 1.2’, Pharmanord, UK), versus magnesium , versus placebo in patients with hereditary or idiopathic disease.
Other observations
(a) SAM alone, or with selenium and vitamin E, was ineffective in double-blind placebo-controlled RCTs from the UK (12). (b) This was also true for selenium and vitamin C in a single-blind RCT from India, the report of which gave no diagnostic criteria, stated gall stones / common bile duct stones as the commonest aetiological factor, did not specify if these problems were on-going, and administered proton pump inhibitors plus pancreatic extracts - both with antioxidant potential - to treatment and no-treatment groups (78). (c) By contrast, in an open observational study from the Czech Republic, vitamin C (0.5 gm/day) plus vitamin E (100 mg/day) for 12 months resulted in substantial pain reduction, to the point of abolition in 44% of 70 patients with mainly alcoholic disease. This was largely attributable to vitamin C, in that vitamin E levels were inexplicably unchanged by treatment, and with the greatest decrements in ROS generation and lipid peroxides among patients with the most functional parenchyma, judging by grade of pancreatogram abnormality (51). (d) A study reported in Polish (72) is cited in two meta-analyses (1, 178). The prescription (C 0.4 gm/day, E 300mg/day) or no treatment was administered in an open RCT for six months in 91 patients with alcoholic disease, while blood antioxidant levels were monitored. The English abstract reports that 68% of the group of 46 on active treatment became pain-free versus 31% of 45 untreated (p=0.002). The treated group also had fewer pancreatitis relapses (p=0.03) and their weight improved (p= 0.001), as did pancreatic exocrine and endocrine function (p=0.001, p=0.015 respectively). (e) An anecdotal account from the USA showed impressive improvement in three patients treated with a grape-seed extract (7).
Clinical improvement in the latter three studies suggests that methionine intake in those patients was adequate, such that ascorbic acid or potent antioxidants in the grape seed extract protected enzymes in the methionine metabolic route (Figure 2). Moreover, the Czech study shows that it is illogical to expect relief from oxidative stress-induced inflammatory pain in patients whose exocrine pancreatic function is severely compromised.
Long-term treatment
The long-term value of combination micronutrient therapy has been documented in reports from Delhi (102) and Manchester (99, 141). However, the importance of correcting reductive stress is illustrated by the finding that in three Manchester patients with recurrent acute pancreatitis, who were referred after the trials, standard treatment failed: the addition of folate to provide more methyl groups did not help, but a choline supplement did (JM Braganza unpublished). This is in tune with the experimental observation that polyenylphosphatidyl choline protects against pancreatic oxidative stress in alcohol-treated rats (2). When choline intake falls short, this phospholipid can be synthesised from phosphatidyl ethanolamine, but incurs severe pressure on ‘the universal methyl donor’, SAM (Figure 2) (169). There is now a convergence of thought on mechanisms of liver and pancreatic damage from a protracted excess of alcohol. The combination of three methyl donors - SAM, folate, betaine (Figure 2) - alleviated alcoholic liver injury, while at the same time lowering the elevated SAM / SAH ratio and homocysteine level (114), but the aforesaid evidence also points to the critical importance of ascorbic acid in protecting the exocrine pancreas, as shown by its ameliorating effect in animal models of mild and severe pancreatitis (30). The inescapable conclusion is that the choline-deficient ethionine-supplemented dietary (CDE) model of acute pancreatitis, which is easily modified to cause inflammatory fibrosis, is highly relevant to clinical pancreatitis.
A surgical audit in 1992 involved 94 patients attending the Manchester Pancreato-Biliary Unit, with a mean follow-up period of 30 months on micronutrient therapy, and more than five years in 22% of cases (99). Imaging studies revealed that 85 % had ‘large-duct disease‘ (moderate or advanced change pancreatitis by endoscopic pancreatography), with or without calculi , and 15 % had ‘small-duct disease’, usually identified by secretin-pancreozymin tests. No patient needed duct decompression or resective pancreatic surgery during 248 patient-years of follow up; the total number of days spent in hospital while on treatment was significantly lower than in the preceding year; 78% of patients became pain-free and a further 7 % had a substantial reduction in pain (although several continued to take simple analgesics as fearful of an attack); just two patients had continuous pain compared to 29 before micronutrient therapy; and of the 76 patients previously in employment, 88% were back at work and 80 % of these were doing the same job. Of the 42 patients who drank alcohol excessively, a third continued to drink as previously, half had abstained altogether, and the others had reduced their intake to ‘safe’ limits (99).
This excellent result, which was continued through to 1998, such that surgery to treat the pain of chronic pancreatitis was virtually obsolete, accrued through strict guidelines: patient-controlled devices to deliver morphine forbidden; routine endoscopic sphincterotomy, pancreatic duct stents or attempts at clearance of pancreatic calculi firmly rejected; psychiatric help sought early when dependence on narcotic analgesics or despair at social upheaval loomed; input of primary care practitioners solicited; nutritionist, social worker and pharmacist engaged from the outset. The prescription of opiates in patients who were already dependent at referral was devolved to the pain team. A weekly medical-surgical clinic was preceded by an interdisciplinary discussion on patients to be seen that day, co-ordinated by a medical registrar, nurse specialist, and biochemist. Each doctor had a printout of previous antioxidant and % MRLA data, so that selenium dosage could be adjusted with reference to the nomogram (Figure 3), while whole blood GSH helped to assess methionine adequacy, and that of vitamin C by reference to the % oxidation of contained ascorbic acid (JM Braganza, unpublished).
The full prescription was usually needed for six months while dietary advice was given on antioxidant -rich foodstuffs (99). Negotiation with executives from Pharmanord, UK, resulted in a combination tablet, ‘Antox’, that reduced the number to an average of four per day compared to 14 per day in the original trial. Further improvements were made by the company in order to increase the daily dose of methionine, while limiting β-carotene to the shell, because of cosmetic distress from a yellow hue, resulting in ‘Antox version 1.2’. Treatment failure in around 10% of about 300 patients was associated with large cysts / pseudocysts, non-compliance in unreformed alcoholics, undiagnosed neoplasia in two (adenocarcinoma, papillary mucinous) and in the described patients with recurrent acute pancreatitis (JM Braganza, unpublished).
After 1998, new Consultants brought to bear their previous experience, such that patient-controlled pumps to deliver morphine were introduced, morphine prescriptions soared, invasive intervention increased, and the micronutrient prescription ceased. This change in practice is witnessed by the high daily dose of morphine and 54% prior intervention rate in the recent negative trial of combination micronutrient therapy (Table 2) (145), compared to none on either count in the 1990 report (159).
6. Conclusion
Chronic pancreatitis seems to represent ‘hepatization’ of the gland - a reversion to its ancestral roots, as a result of chronic exposure to xenobiotics that strike parenterally (16, 165). Choline intake and status in patients on their habitual diet were unfortunately not assessed in the Manchester studies, but are needed urgently. In the meantime, the addition of a choline supplement to the successful micronutrient cocktail is judicious, and probably should take precedence over calls to prescribe zinc, folate or magnesium. The perception of electrophilic / oxidative stress in acinar cells as the ‘obligate intermediate phenotype’ in the pathogenesis of chronic pancreatitis (Figure 1) fulfils a set of postulates derived from Koch’s classical work on tuberculosis, as modified for a polygenic disease (23,24). Hence, it is difficult to see the need for ‘a new framework for 21st century medicine‘ (171).
7. References
- Ahmed Ali U, Jens S, Busch O.R.C, Keus F, van Goor H,.Gooszen H.G, Boermeester M.A. Antioxidants for pain in chronic pancreatitis. Cochrane Database Syst Rev 8: CD008945, 2014. PMID: 25144441
- Aleynik S, Leo M.A, Aleynik M.K, Lieber C.S. Alcohol-induced pancreatic oxidative stress: protection by phospholipid repletion. Free Radic Biol Med 26: 609-619, 1999. PMID: 10218649
- Anderson D, Phillips B.J, Yu TW, Edwards A.J, Ayesh E.R, Butterworth K.R. The effects of vitamin C supplementation on bilomarkers of oxygen radical generated damage in human volunteers with ‘low’ or ‘high’ cholesterol levels. Environ Mol Mutag 30: 161-174, 1997. PMID: 9329641
- Arnẽr E.S.J, Holmgren A. Physiological functions of thioredoxin and thioredoxin reductase. Eur J Biochem 267: 6102-6109, 2000. PMID: 11012661
- Arumugam G, Padmanaban M, Krishnan D, Panneerselvam S, Rajagopal S. Influence of copper, iron, zinc and Fe3+ haemoglobin levels on the etiopathogenesis of chronic calcific pancreatitis - a study in patients with pancreatitis. Biol Trace Elem Res 142: 424-434, 2011. PMID: 20809271
- Balaghi M, Wagner C. Folate deficiency inhibits pancreatic amylase secretion in rats. Am J Clin Nutr 61:90-96, 1995. PMID: 7529961
- Banerjee B, Bagchi D. Beneficial effects of a novel IH636 grape seed proanthocyanidin extract in the treatment of chronic pancreatitis. Digestion 63: 203-206, 2001. PMID: 11351148
- Banks P.A, Hughes M, Ferrante M, Noordhoek E.C, Ramagopal V, Slivka A. Does allopurinol reduce pain of chroniuc pancreatituis? Int J Pancreatol 22: 171-176, 1997.PMID: 9444547
- Bhandary D, Marahatta A, Kim H-R, Chae H-J. An involvement of oxidative stress in endoplasmic stress and its associated diseases. Int J Mol Sci 14: 434-456, 2013. PMID: 23263672
- Bhardwaj P, Garg P.K, Maulik S.K, Saraya A, Tandon R.K, Acharya S.K. A randomized controlled trial of antioxidant supplementation for pain relief in patients with chronic pancreatitis. Gastroenterology 138: 149-159, 2009. PMID: 18952082
- Bhardwaj P, Yadav R.K. Chronic pancreatitis: role of oxidative stress and antioxidants. Free Radic Res 47: 941-949, 2013. PMID: 23668832
- Bilton D, Schofield D, Mei G, Kay P.M, Bottiglieri T, Braganza J.M. Placebo-controlled trials of antioxidant therapy including S-adenosylmethionine in patients with recurrent non-gallstone pancreatitis. Drug Invest 8: 10-20, 1994.
- Blanchard J.A, Barve S, Joshi-Barve S, Talwalker R, Gates L.K. Antioxidants inhibit cytokine production and suppress NF-kappa B activation in CAPAN-1 and CAPAN-2 cell lines. Dig Dis Sci 46; 2768-2772, 2001. PMID: 11768272
- Bonham M.J, Abu-Zidan F.M, Simovic M.O, Sluis K.B, Wilkinson A, Winterbourne C.C, Windsor J.A. Early ascorbic acid depletion is related to the severity of acute pancreatitis. Br J Surg 86: 1296-1301, 1999. PMID: 10540137
- Braganza J.M. Pancreatic disease: a casualty of hepatic ‘detoxification’ ? Lancet ii:1000-1003, 1983. PMID: 6138545
- Braganza JM. The pancreas. In: Pounder RE (ed)Recent Adv Gastroenterol 6: 251-280, 1986. Churchill Livingstone, London.
- Braganza J.M. Free radicals and pancreatitis. In: Rice-Evans C, Dormandy T.L (eds) Free radicals : chemistry, pathology and medicine : pp 357-381, 1988. Richelieu Press, London.
- Braganza J.M. The role of the liver in exocrine pancreatic disease. Int J Pancreatol 3: S19-42, 1988. PMID:3062099
- Braganza J.M. Experimental acute pancreatitis. Curr Opin Gastroenterol 6: 763-768, 1990.
- Braganza J.M. Antioxidant therapy for pancreatitis: clinical experience. In: Braganza J.M (ed) The pathogenesis of pancreatitis. pp 178-197, 1991, Manchester University Press, Manchester.
- Braganza J.M. Toxicology of the pancreas. In: Braganza J.M. (ed) The pathogenesis of pancreatitis pp 66-85, 1991. Manchester University Press, Manchester.
- Braganza J.M. The pancreas. In: Brocklehurst J.C, Tallis R, Fillit J (eds) Textbook of geriatric medicine and gerontology, 4th edition, pp 527-535, 1991. Churchill Livingstone, London.
- Braganza J.M. The pathogenesis of chronic pancreatitis. QJM 89: 243-250, 1996. PMID: 8733510
- Braganza J.M. A framework for the aetiogenesis of chronic pancreatitis. Digestion 59 (suppl 4): 1-12, 1998. PMID: 9832631
- Braganza J.M. Towards a novel treatment strategy for acute pancreatitis. 1. Reappraisal of the evidence on aetiogenesis. Digestion 63: 69-91, 2001. PMID: 11244246
- Braganza J.M. Towards a novel treatment strategy for acute pancreatitis. 2. Principles and potential practice. Digestion 63: 143-162, 2001. PMID: 1351142
- Braganza J.M. Mast cell control: likely modus operandi of panhaematin in experimental pancreatitis. Gut 61: 632, 2012. PMID: 21757450
- Braganza J.M. Micronutrient therapy for chronic pancreatitis: Premises and pitfalls. JOP 14:304-308, 2013. PMID: 23846913
- Braganza J.M. Pancreatic extracts for painful chronic pancreatitis: Micronutrient antioxidant therapy by proxy. JOP 15: 541-543, 2014. PMID: 25435568
- Braganza J.M, Dormandy T.L. Micronutrient therapy for chronic pancreatitis: Rationale and impact. JOP 11: 99-112, 2010. PMID: 20208316
- Braganza J.M, Hewitt C.D, Day J.P. Serum selenium in patients with chronic pancreatitis: lowest values during painful exacerbations. Trace Elements Med 5: 79-84, 1988.
- Braganza J.M, John S, Padmayalam I, Mohan V, Chari S, Madanagopalan M. Xenobiotics and tropical chronic pancreatitis. Int J Pancreatol 7: 231-245, 1990.
- Braganza J.M, Jolley J.E, Lee W.R. Occupational chemicals and pancreatitis: a link ? Int J Pancreatol 1: 9-19, 1986. PMID: 3320224
- Braganza J.M, Lee S, McCloy R.F, McMahon M.J. Seminar. Chronic pancreatitis. Lancet 377: 1184-1197, 2011. PMID: 21397320
- Braganza J.M, Odom N, McCloy R.F, Ubbink J.B. Homocysteine and chronic pancreatitis. Pancreas 39: 1303-1304, 2010. PMID: 20944492
- Braganza J.M, Schofield D, Snehalatha C, Mohan V. Micronutrient antioxidant status in tropical compared with temperate-zone chronic pancreatitis. Scand J Gastroenterol 28: 1098-1104, 1993. PMID: 8303214
- Bruce J.I.E, Elliott A.C. Pharmacological evaluation of the role of cytochrome P450 in intracellular calcium signalling in pancreatic acinar cells. Br J Pharmacol 131: 761-771, 2000. PMID: 11030726
- Burton F. Alkaade S, Collins D, Muddana V, Slivka A, Brand R.E, Gelrud A, Banks P.A, Sherman S, Anderson M.A, Romagnuolo J, Lawrence C, Baillie T.B, Gardner T.B, Lewis M.D, Amann S.T, Lieb J.G, O’Connell M.O, Yadav D, Whitcomb D.C. Forsmark C.E. for the North American Pancreatic Study group. Use and perceived effectiveness of non-analgesic medical therapies for chronic pancreatitis in the United States. Aliment Pharmacol Ther33:149-159, 2011. PMID: 21083584
- Cai G.H, Huang J, Chen J, Wu H.H, Dong Y.L, Smith H.S, Li Y.Q, Wang W, Wu S.X. Antioxidant therapy for pain relief in patients with chronic pancreatitis: systematic review and meta-analysis. Pain Physician 16: 521-532, 2013. PMID: 24284838
- Capdevila A, Decha-Umphai W, Song K.H, Borchardt R.T, Wagner C. Pancreatic enzyme secretion is blocked by inhibitors of methylation. Arch Biochem Biophys 345:47-55, 1997. PMID: 9281310
- Capurso G, Cocomello L, Benedetto U, Camma C, Delle Fave G. Meta-analysis: the placebo rate of abdominal pain remission in clinical trials of chronic pancreatitis. Pancreas 41: 125-131, 2012. PMID: 22513290
- Cawood P, Wickens D.G, Iversen S.A, Braganza J.M, Dormandy T.L. The nature of diene conjugation in human serum, bile and duodenal juice. FEBS Lett 62: 239-243, 1983. PMID: 6628668
- Cecere A, Tancredi L, Gattoni A. Primary sclerosing cholangitis. Panminerva Med 44: 313-323, 2002. PMID: 12434113
- Chaloner C, Sandle L.N, Mohan V, Snehalatha C, Viswanathan M, Braganza J.M. Evidence for induction of cytochrome P4501 in patients with tropical pancreatitis. Int J Pharmacol Ther Toxicol 28: 235-240, 1990. PMID: 2376424
- Chauhan S.S, Pannu D.S, Forsmark C.E. Antioxidants as adjuvant therapy for pain in chronic pancreatitis. Practical Gastroenterology: Nutrition issues in Gastroenterology series #103: 2012
- Chvanov M, Petersen O.H, Tepkin A. Free radicals and the pancreatic acinar cells: role in physiology and pathology. Philos Trans R Soc Lond B Biol Sci 360: 2273-2284, 2005. PMID: 16321797
- Correia M.A, Burk R.F. Defective utilization of haem in selenium-deficient rat liver. Biochem J 214: 53-58, 1983. PMID: 6615473
- De las Heras Castaño G, Garcia de la Paz A, Fernández M.D, Fernández Forcelledo J.L. Use of antioxidants to treat pain in chronic pancreatitis. Rev Esp Enferm Dig 92: 831-835, 2000. PMID: 10985097
- De Las Heras-Castaño G, García-Unzueta T, Dominguez-Diez A, Fernández-Gonzáles M.D, García de la Paz A.M, Mayorga-Fernández M, Fernández-Fernández F. Pancreatic fibrosis in rats and its response to antioxidant treatment. JOP 6: 316-324, 2005. PMID: 16006681
- Dhingra R, Singh N, Sachdev V, Upadhya A.D, Saraya A. Effect of antioxidant supplementation on surrogate markers of fibrosis in chronic pancreatitis. A randomized placebo-controlled trial. Pancreas 42: 589-595, 2013. PMID: 23531998
- Ditẽ P, Prẽcehtelová M, Novooytmny L, Soska V, Źaková A, Lata J. Changes of reactive substance in patients with morphologically different degrees of chronic pancreatitis and effects of long-term therapy with natural antioxidants. Gastroenterologia Polska 10: 379-383, 2003.
- Dolai S, Liang T, Cosen-Binker L.I, Lam P.P.L, Gaisano H. Regulation of physiologic and pathologic exocytosis in pancreatic acinar cells. The Pancreapedia: Exocrine Pancreas Knowledge Base, 2012. DOI: 10.3998/panc.2012.12
- Durgaprasad S, Ganesh Pai C, Vasanthkumar, Alvres J.F, Namitha S.A. A pilot study of the antioxidant effect of curcumin in tropical pancreatitis. Indian J Med Res 122: 315-318, 2005. PMID: 16394323
- Foster J.R. Toxicology of the exocrine pancreas. In: Ballantyne B, Marrs T.C, Syverson J (eds). General and Applied Toxicology 3nd edition, pp 1411-1456, 2009. Wiley, New Jersey.
- Foster J.R, Idle J.R, Hardwick J.P, Bars R, Scott P, Braganza J.M. Induction of drug-metabolizing enzymes in human pancreatic cancer and chronic pancreatitis. J Pathol169:457-463, 1993. PMID: 8501544
- Ganesh Pai C, Sreejayan, Rao M.N. Evidence for oxidative stress in chronic pancreatitis. Indian J Gastroenterol 18: 156-157, 1999. PMID: 10531717
- Georgelin T, Schnee M, Sagniez M, Bailly F, Naudot I, Soulard F.M, Martin F. Antioxidant status in patients with hereditary chronic pancreatitis (HCP) and alcoholics. Gastroenterology 114: A461, 1998.
- Ghyczy M, Boros M. Electrophilic methyl groups present in the diet ameliorate pathological states induced by reductive and oxidative stress: a hypothesis. Br J Nutr 85:409-414, 2001. PMID: 11348555
- Ghyczy M, Torday C, Boros M. Simultaneous generation of methane, carbon dioxide, and carbon monoxide from choline and ascorbic acid: a defence mechanism against reductive stress ? FASEB 17:1124-1126, 2003. PMID: 12692080
- Gilbert K.M. Xenobiotic exposure and autoimmune hepatitis. Hepatitis Research and Treatment 2010.
- Girish B.N, Rajesh G, Vaidyanathan K, Balakrishnan V. Alterations in plasma amino acid levels in chronic pancreatitis. JOP 12: 11-18, 2011. PMID: 21206095
- Girish B.N, Vaidyanathan K, Rajesh G, Balakrishnan V. Effects of micronutrient status on oxidative stress and exocrine pancreatic function in patients with chronic pancreatitis. Indian J Biochem Biophys 49: 386-391, 2012. PMID: 23259326
- Girish B.N, Vaidyanathan K, Rao N.A, Rajesh G, Reshmi S, Balakrishnan V. Chronic pancreatitis is associated with hyperhomocysteinaemia and derangements in transsulfuration and transmethylation pathways. Pancreas 39: e11-16, 2010. PMID: 20050230
- Golden M.H.N. The exocrine pancreas in severe malnutrition. In: Braganza J.M. (ed) The pathogenesis of pancreatitis. pp 139-155, 1991. Manchester University Press, Manchester.
- Gonzalez F.J. Role of cytochromes P450 in chemical toxicity and oxidative stress: studies with CYP2E1. Mutat Res 568: 101-110, 569. PMID: 15603755
- Grisby B, Dodriguez-Rilo H, Khan K. Antioxidants and chronic pancreatitis: theory of oxidative stress and trials of antioxidant therapy. Dig Dis Sci 57: 835-841, 2012. PMID: 22302241
- Gutteridge J.M.C. Lipid peroxidation and antioxidants as biomarkers of tissue damage. Clin Chem 41: 1819-1828, 1995. PMID: 7497639
- Guyan P.M, Uden S, Braganza J.M. Heightened free radical activity in pancreatitis. Free Radic Biol Med 8: 347-354, 1990. PMID: 2379863
- Heaney A.P, Sharer N, Rameh B, Braganza J.M, Durrington P.N. Prevention of recurrent pancreatitis in familial lipoprotein lipase deficiency with high-dose antioxidant therapy. J Clin Endocrin Metab 84: 1203-1205, 1999. PMID: 10199753
- Houston J.B. Cytochromes P450 in chronic pancreatitis. In: Braganza J.M.(ed) The pathogenesis of pancreatitis pp 103-114, 1991. Manchester University Press, Manchester.
- Iimuro Y, Bradford B.U, Gao W, Kadiska M, Mason R.P, Stefanovic B, Brenner D.A, Thurman R.G. Detection of α-hydroxyethyl free radical adducts in the pancreas after chronic exposure to alcohol in the rat. Mol Pharmacol 50: 656-661, 1996. PMID: 8794907
- Jarosz M, Orzeszko M, Rychlik E, Kozuch M. [Polish] Antioxidants in the treatment of chronic pancreatitis Gastroenterologia Polska 17: 41-46, 2010.
- Jeppe C.Y, Smith M.D. Transverse descriptive study of xenobiotic exposures in patients with chronic pancreatitis and pancreatic cancer. JOP 9: 235-239, 2008. PMID: 18326937
- Ji Y-L, Wang Z, Wang H, Zhang C, Zhang Y, Zhao M, Chen Y-H, Meng X-H, Xu D-X. Ascorbic acid protects against cadmium-induced reticulum stress and germ cell apoptosis in testes. Reproductive Toxicology 34: 357-363, 2012. PMID: 22569276
- Ji Y-L, Wang H, Zhang C, Zhang Y, Zhao M, Chen Y-H, Xu D-X. N-acetylcysteine protects against cadmium-induced germ cell apoptosis by inhibiting endoplasmic reticulum stress in testes. Asian J Androl 15: 290-296, 2013. PMID: 23353715
- Johnston C.S. The antihistamine action of ascorbic acid. Subcell Biochem 25: 189-213, 1998. PMID: 8821975
- Kahn D.R, Carlson A.B. On the mechanism of experimentally induced ethionine pancreatitis. Ann Surg 150: 42-49, 1959. PMID: 13661828
- Khariong P.D.S, Hajong R, Kundu D, Tongper D, Mibang N, Picardo P.J. A comparative study into pain treatment in chronic pancreatitis between non operative conventional treatment and those treated with antioxidants. IOSR-JDMS 6: 91-99, 2013.
- Khurana V, Barkin J.S. Pancreatitis induced by environmental toxins. Pancreas 22:102-195, 2001. PMID: 11138962
- Kirk G.R, White J.S, McKie L, Stevenson M, Young I, Clements W,D, Rowlands B.J. Combined antioxidant therapy reduces pain and improves quality of life in chronic pancreatitis. J Gastrointest Surg 10: 499-503, 2006. PMID: 16627214
- Ko S.B, Azuma S, Yoshikawa T, Yamamoto A, Kyokane K, Ko M.S, Ishiguro H. Molecular mechanisms of pancreatic stone formation in chronic pancreatitis. Front Physiol 3: 415: 2012. PMID: 23133422
- Kodydkova J, Vavrova L, Stankova B, Macasek J, Krechler T, Zak A. Antioxidant status and oxidative stress markers in pancreatic cancer and chronic pancreatitis. Pancreas 42: 614-621, 2013. PMID: 23558240
- Korashy H.M, El-Kadi A.O. Modulation of TCCD-mediated induction of cytochrome P-4501A1 by mercury, lead, and copper in human HepG2 cell line. Toxicol In Vitro 22: 154-158, 2008. PMID: 17889500
- Lafleur M.A, Stevens J.L, Lawrence J.W. Xenobiotic perturbation of ER stress and the unfolded protein response. Toxicol Pathol 41: 235-262, 2013. PMID: 23334697
- LaRusch J, Jung J, General I.J, Lewis M.D, Park H.W, Brand R.E, Gelrud A, Anderson M.A, Banks P.A, Conwell D, Lawrence C, Romagnuolo J, Baillie J, Alkaade S, Cote G, Gardner T.B, Amann S.T, Slivka A, Sandhu B, Aloe A, Kienholz M.L, Yadav D, Barmada M.M, Bahar I, Lee M.G, Whitcomb D.C, and the North American Pancreatology Study Group. Mechanisms of CFTR functional variants that impair regulated bicarbonate permeation and increase risk for pancreatitis but not for cystic fibrosis. PLOS Genetics 10: 1-15, 2014. PMID: 25033378
- Leo M.A, Aleynik S.I, Siegel J.H, Kasmin F.E, Aleynik M.K, Leiber C.S. F2-isoprostanes and 4-hydroxynonenal excretion in human bile of patients with biliary tract and pancreatic disorders. Am J Gastroenterol 92: 2069-2072, 1997. PMID: 9362195
- Leung P.S, Chan Y.C. Role of oxidative stress in pancreatic inflammation. Antiox Redox Signal 11: 135-165, 2009. PMID: 18837654
- Lipecka J, Norez C, Bensalem N, Baudouin-Legros M, Planelles G, Becq F, Edelman A, Davezac N. Rescue of Delta508-CFTR (cystic fibrosis transmembrane conductance regulator) by curcumin: involvement of the keratin 18 network. J Pharmacol Exp Ther 317: 500-505, 2006. PMID: 16424149
- Lipinski B. Evidence in support of a concept of reductive stress. Br J Nutr 87: 93-94, 2002. PMID: 11895317
- Liu L.G, Yan H, Zhang W, Yao P, Zhang X.P, Sun X.F, Nussler A.K. Induction of heme oxygenase-1 in human hepatocytes to protect from ethanol-induced cytotoxicity. Biomed Environ Sci 17: 315-326, 2004. PMID: 15602829
- Longnecker D.S. Environmental factors and diseases of the pancreas. Environ Health Perspect 20: 105-112, 1977. PMID: 598342
- Lucas D, Ferrara R, Gonzalez E, Bodenez P, Albores A, Manno M, Berthou F. Chlorzoxazone, a selective probe for phenotyping CYP2E1 in humans. Pharmacogenetics 9: 377-388, 1999. PMID: 10471070
- Luthen R.E, Grendell J.H. Thiol metabolism and acute pancreatitis: trying to make the pieces fit. Gastroenterology 107: 888-892, 1994. PMID: 8076778
- Ma Q, Lu A.Y.H. Cyp1A induction and human risk assessment: an evolving tale of in vitro and in vivo studies. Drug Metab Dispos 35:1009-1016, 2007. PMID: 17431034
- Malhotra J.D, Miao H, Zhang K, Wolfson A, Pennathur S, Pipe S.W, Kaufman R.L. Antioxidants reduce endoplasmic stress and improve protein secretion. PNAS 105: 18525-18530, 2008. PMID: 19011102
- Martensson J, Bolin T. Sulfur amino acid metabolism in chronic relapsing pancreatitis. Am J Gastroenterol 81:1179-1184, 1986. PMID: 3788926
- Masamune A. Genetics of pancreatitis: the 2014 update. Tohoku J Exp Med 232: 69-77, 2014. PMID: 24522117
- Mathew P, Wyllie R, Van Lente F, Steffen R.M, Kay M.H. Antioxidants in hereditary pancreatitis. Am J Gastroenterol 91:1558-1562, 1996. PMID: 8759661
- McCloy R.F. Chronic pancreatitis at Manchester, UK. Focus on antioxidant therapy. Digestion 59 (suppl 4): 36-48, 1998. PMID: 9832634
- McNamee R, Braganza J.M, Hogg J, Leck I, Rose P, Cherry N.M. Occupational exposure to hydrocarbons and chronic pancreatitis: a case-referent study. Occup Environ Med 51: 631-637, 1994. PMID: 7951796
- Merkord J, Weber H, Jonas L, Nizze H, Heninghausen G. The influence of ethanol on long-term effects of dibutylytin dichloride (DBTC) in pancreas and liver of rats. Hum Exp Toxicol 17: 144-150, 1998. PMID: 9587782
- Midha S, Khaguria R, Shastri S, Kabra M, Garg P.K. Idiopathic chronic pancreatitis in India: phenotypic characterisation and strong genetic susceptibility due to SPINK1 and CFTR mutations. Gut 59: 800-807, 2010. PMID: 20551465
- Mohan V, Braganza J.M. Xenobiotics in tropical pancreatitis. In: Braganza J.M. (ed) The pathogenesisis of pancreatitis: pp115-128, 1991. Manchester University Press, Manchester.
- Monfared S.S.M.S, Vahidi H, Abdolghaffari A.H, Nikfar S, Abdollahl M. Antioxidant therapy in the management of acute, chronic and post ERCP pancreatitis: A systematic review. World J Gastroenterol 15: 4481-4490, 2009. PMID: 19777606
- Moreno M-L, Escobar J, Izquierdo-Ấlvarez A, Gil A, Pérez S, Pereda J, Zapico I, Vento M, Sabater L, Marina A, Martinez-Ruiz A, Sastre J. Disulfide stress : a novel type of oxidative stress in acute pancreatitis. Free Radic Biol Med 70 : 265-277, 2014. PMID: 24456905
- Morris-Stiff G.J, Bowrey D.j, Oleesky D, Davies M, Clark G.W, Puntis M.C. The antioxidant profiles of patients with recurrent acute and chronic pancreatitis. Am J Gastroenterol 94: 2135-2140, 1999. PMID: 10445540
- Morselli-Labate A, Fantini L, Pezzilli R. Hydrogen sulphide, nitric oxide and a molecular mass 66 u substance in the exhaled air of chronic pancreatitis patients. Pancreatology 7: 497-504, 2007. PMID: 17912017
- Nakos G, Gossrau R. Visualization of hydrogen peroxide (H2O2)-production from histamine. Ann Anat 177: 431-438, 1995. PMID: 7645739
- Odewabi A.O, Ogundahunsi O.A, Oyalowo M. Effect of exposure to petrochemical fumes on plasma antioxidant defense system in petrol attendants. Br J Pharmacol Toxicol 5: 83-87, 2014.
- Ossola J.O, Groppa M.D, Tomaro M.I. Relationship between oxidative stress and heme oxygenase induction by copper sulphate. Arch Biocem Biophys 227: 332-337, 1997. PMID: 9016830
- Palmieri B, Sblendorio V. Oxidative stress tests: overview on reliability and use. Part 1. Eur Rev Med Pharmacol Sci 11: 309-342, 2007. PMID: 18074940
- Pandol S.J, Gorelick F.S, Gerloff A, Lugea A. Alcohol abuse, endoplasmic reticulum stress and pancreatitis. Dig Dis 28: 776-782, 2010. PMID: 21525762
- Pezzilli R. Antioxidants are not useful in reducing both pain and inflammation in chronic pancreatitis. Recent Pat Inflamm Allergy Drug Discov 8: 19-23, 2014. PMID: 24397820
- Purohit V, Abdelmalek M.F, Barve S, Benevenga N.J, Halsted C.H, Kaplowitz N, Kharbanda K.K, Liu Q.Y, Lu S.C, McClain C.J, Swanson C, Zakhari S. Role of S-adenosylmethionine, folate, and betaine in the treatment of alcoholic liver disease: summary of a symposium. Am J Clin Nutr 86: 14-24, 2007. PMID: 17616758
- Quesnel L.B, Jaran A.S, Braganza J.M. Antibiotic accumulation and membrane trafficking in cystic fibrosis cells. J Antimicrob Chemother 1998; 41:215-221. PMID: 9533463
- Quillot D, Walters E, Bonte J-P, Fruchart J-C, Duriez P, Ziegler O. Diabetes mellitus worsens antioxidant status in patients with chronic pancreatitis. Am J Clin Nutr 81: 1117-1125, 2005. PMID: 15883437
- Rekhadevi P.V, Rahman M.F, Mahboob M, Grover P. Genotoxicity in filling station attendants exposed to petroleum hydrocarbons. Ann Occup Hyg 54: 944-954, 2010. PMID: 20956619
- Rinderknecht H. Pancreatic secretory enzymes. In: Go V.L.W, Gardner J.D, Brooks F.P, Lebenthal E, DiMagno E.P, Scheele G.A. (eds). The exocrine pancreas: biology, pathobiology and diseases, 2nd edition, pp 219-252, 1993. Raven Press, New York.
- Robin M-A, Descatoire V, Le Roy M, Berson A, Lebreton F-R, Maratrat M, Ballet F, Loeper J, Pessayre D. Vesicular transport of newly synthesised cytochromes P4501A to the outside of rat hepatocyte plasma membranes. JPET 294; 1063-1069, 2000. PMID: 10945860
- Rose P, Fraine E, Hunt L.P, Acheson D.W.K, Braganza J.M. Dietary antioxidants and chronic pancreatitis. Hum Nutr Clin Nutr 40: 151-164, 1986. PMID: 3957720
- Rottner M, Freyssinet J-M, Martinez M.C. Mechanisms of the noxious inflammatory cycle in cystic fibrosis. Respiratory Research 10: 23, 2009. PMID: 19284656
- Rutkowski D.T, Kaufman R.J. A trip to the ER: coping with stress. Trends Cell Biol 14: 20-28, 2004. PMID: 14729177
- Sah R.P, Garg S.K, Dixit A.K, Dudeja V, Dawra R.K, Saluja A.K. Endoplasmic stress is chronically activated in chronic pancreatitis. J Biol Chem 289: 27551-27561, 2014. PMID: 25077966
- Sah R.P, Saluja AK. Trypsinogen activation in acute and chronic pancreatitis: is it a prerequisite? Gut 60: 1305-1307, 2011. PMID: 21672938
- Sajewicz W, Milnerowicz S, Nabzdyk S. Blood plasma antioxidant defense in patients with pancreatitis. Pancreas 32: 139-144, 2006. PMID: 16552332
- Salh B, Webb K, Guyan P.M, Day J.P, Wickens J, Griffin J, Braganza J.M, Dormandy T.L. Aberrant free radical activity in cystic fibrosis. Clin Chim Acta 181: 65-74, 1989. PMID: 2721006
- Salim A.S. Role of oxygen-derived free radical scavengers in the treatment of recurrent pain produced by chronic pancreatitis. Arch Surg 126: 1109-1114, 1991. PMID: 1929842
- Sandilands D, Jeffrey I.J.M, Haboubi N.Y, MacLennan I.A.M, Braganza J.M. Abnormal drug metabolism in chronic pancreatitis. Treatment with antioxidants. Gastroenterology 98: 766-772, 1990. PMID: 2298375
- Sanfey H, Buckley G.B, Cameron J.L. The role of oxygen-derived free radicals in the pathogenesis of acute pancreatitis. Ann Surg 200:405-412, 1984. PMID: 6207783
- Santini S.A, Spada C, Bononi F, Foschia F, Mutignani M, Perri V, Giardina B, Silveri N. G, Costamagna G. Enhanced lipoperoxidation products in pure pancreatic juice: evidence for organ-specific oxidative stress in chronic pancreatitis. Dig Liver Dis 35: 888-892, 2003. PMID: 14703885
- Sato A, Nakajima T, Koyama Y. Effects of chronic ethanol consumption on hepatic metabolism of aromatic and chlorinated hydrocarbons in rats. Br J Ind Med 37: 382-386, 1980. PMID: 7192567
- Schick V, Scheiber J.A, Mooren F.C, Turi S, Ceyhan G.O, Schnekenburger J, Sendler M, Schwaiger T, Cehyan G.O, van den Brandt C, Fluhr G, Domschke W, Krüger B, Mayerle J, Lerch M.M. Effect of magnesium supplementation and depletion on the onset and course of acute experimental pancreatitis. Gut 63: 1469-1480, 2014. PMID: 24277728
- Schoenberg M.H, Büchler M, Pietrzyk C, Uhl W, Birk D, Eisele S, Marzinzig M, Beger H.G. Lipid peroxidation and glutathione metabolism in chronic pancreatitis. Pancreas 10: 36-43, 1995. PMID: 7899458
- Schofield D, Rameh B, Leach F.N, Braganza J.M. Antioxidant profiles after antioxidant regimens for ten weeks: implications for clinical trial design. Digestion 58 (suppl 2): 26, 1997
- Scott G. Antioxidants in vitro and in vivo. In: Braganza J.M. (ed) The pathogenesis of pancreatitis: pp 159-177, 1991. Manchester University Press, Manchester.
- Scott P, Bruce C, Schofield D, Shiel N, Braganza J.M, McCloy R.F. Vitamin C status in patients with acute pancreatitis. Br J Surg 80: 750-754, 1993. PMID: 8330166
- Scott P, Knoop M, McMahon R.F.T, Braganza J.M, Hutchinson I.V. S-adenosyl-L-methionine protects against haemorrhagic pancreatitis in partially immunosuppressed panreaticoduodenal transplant recipients. Drug Investigation 4 (suppl 4): 69-77, 1992.
- Segal I, Ally R, Hunt L.P, Sandle L.N, Ubbink J.B, Braganza J.M. Insights into the development of alcoholic chronic pancreatitis at Soweto, south Africa. Pancreas 40: 508-516, 2011. PMID: 21499204
- Shah N.S, Makin A.J, Sheen A.J, Siriwardena A.K. Quality of life assessment in patients with chronic pancreatitis receiving antioxidant therapy. World J Gastroenterol 16: 4066-4071. 2012. PMID: 20731021
- Shahedi K, Pandol S.J, Hu R. Oxidative stress and alcoholic pancreatitis. J Gastroenterol Hepatol Res 2: 335-342, 2013.
- Sharer N.M, Schwarz M, Malone G, Howarth A, Painter J, Super M, Braganza J.M. Mutations of the cystic fibrosis gene in patients with chronic pancreatitis N Engl J Med 339: 645-652, 1998. PMID: 9725921
- Sharer N.M, Taylor P.M, Linaker B.D, Gutteridge J.M.C, Braganza J.M. Safe and successful use of vitamin C to treat painful calcific chronic pancreatitis despite iron overload from primary haemochromatosis. Clin Drug Invest 10: 310-315, 1995.
- Shimizu Y, Handershot L.M. Oxidative folding: cellular strategies for dealing with the resultant equimolar production of reactive oxygen species. Antiox Redox Signal 11: 2317-2331, 2009. PMID: 19243234
- Siriwardena A.K. Reappraisal of xenobiotic-induced, oxidative stress-mediated cellular injury in chronic pancreatitis: a systematic review. World J Gastroenterol 20: 3033-3043, 2014. PMID: 24659895
- Siriwardena A.K, Mason J.M, Sheen A.J, Makin A.J, Shah N.S. Antioxidant therapy does not reduce pain in patients with chronic pancreatitis. The ANTICIPATE study. Gastroenterology 143: 655-673, 2012. PMID: 22683257
- Smith G.N, Taj M, Braganza J.M. On the identification of a conjugated diene component of duodenal bile as 9Z, 11E-octadecanoic acid. Free Radic Biol Med 10: 13-21, 1991. PMID: 20250295
- Standop L, Schneider M, Ulrich A, Büchler M.W, Pour P.M. Differences in immunohistochemical expression of xenobiotic-metabolizing enzymes between normal pancreas, chronic pancreatitis and pancreatic cancer. Toxicol Pathol 31: 500-513, 2003. PMID: 14692619
- Starek K, Kaminski M. [Polish] Toxicity of certain petrochemical derivatives used as dielectrics in electromachining. V. Morphological, cytoenzymic, and biochemical changes in the liver of rats chronically exposed to kerosene hydrocarbons. Med Pr 33: 9-53, 1982.
- Strubelt O. Interactions between ethanol and other hepatotoxic agents. Biochem Pharmacol 29: 1445-1449, 1980. PMID: 6994745
- Syrota A, Dop-Ngassa M, Cerf M, Paraf A. 11C-L-methionine for evaluation of pancreatic exocrine function. Gut 22: 907-915, 1981. PMID: 6171485
- Szuster-Ciesielska A, Daniluk J, Kandefer-Szerszeńn M. Oxidative stress in blood of patients with alcohol related pancreatitis. Pancreas 22: 261-266, 2001. PMID: 11291927
- Takahashi T, Akagi R, Shimizu H, Hirakawa M, Sassa S. Heme oxygenase-1. A major player in the defence against the oxidative tissue injury. In: Abraham NG, Alam J, Nath K (eds) Heme oxygenase in biology and medicine pp 387-396, 2002. Kluwer Academic / Plenum Publishers, New York.
- Takasu A, Shimosegawa E, Hatazawa J, Nagasaki Y, Kimura K, Fujita M, Toyota T. [11C]Methionine positron emission tomography for the evaluation of pancreatic exocrine function in chronic pancreatitis. Pancreas 22:203-209, 2001. PMID: 11249078
- Tandon R.K, Garg P.K. Oxidative stress in chronic pancreatitis: pathophysiological relevance and management. Antiox Redox Signal 15: 2757-2766, 2011. PMID: 21902596
- Telek G, Regõly-Mérei J, Kovács G.C, Simon L, Hamar J, Jakab F. The first histological demonstration of pancreatic oxidative stress in human acute pancreatitis. Hepato-gastroenterology 48: 1252-1258, 2001. PMID: 11677940
- Uboh F.E, Akpanabiatu M.I, Eyong E.U, Ebong P.E, Eka O.O. Evaluation of toxicological implications of inhalation exposure to kerosene fumes and petrol fumes in rats. Acta Biol Szeged 49: 19-22, 2005.
- Uboh F.E, Ebong P.E, Akpan H.D, Usoh I.F. Hepatoprotective effect of vitamins C and E against gasoline vapour-induced liver injury in male rats. Turk J Biol 36: 217-223, 2012.
- Uden S, Acheson D.W.K, Reeves J, Worthington H.V, Hunt L.P, Brown S, Braganza J.M. Antioxidants, enzyme induction, and chronic pancreatitis, A reappraisal following studies in patients on anticonvulsants. Eur J Clin Nutr 42; 561-569, 1988. PMID: 3224602
- Uden S, Bilton D, Nathan L. Hunt L.P, Main C, Braganza J.M. Antioxidant therapy for recurrent pancreatitis: placebo-controlled trial. Aliment Pharmacol Ther 4: 357-371, 1990. PMID: 2103755
- Uden S, Schofield D, Miller P.F, Bottiglieri T, Braganza J.M. Antioxidant therapy for recurrent pancreatitis: biochemical profiles in a placebo-controlled trial. Aliment Pharmacol Ther 6: 229-240, 1992. PMID: 160043
- Ulrich A.B, Schmeid B.M, Matsuzaki H, Lawson T.A, Freiss HY, Andrén-Sandberg A, Büchler M.W, Pour P.M. Increased expression of glutathione S-transferase- π in the islets of patients with primary chronic pancreatitis but not secondary chronic pancreatitis. Pancreas 22: 388-394, 2001. PMID: 11345140
- Uomo G, Talamini G, Rabitti P.G. Antioxidant treatment in hereditary pancreatitis. A pilot study on three young patients. Dig Liver Dis 33: 58-62, 2001. PMID: 11303976
- Van Gossum A, Closset P, Noel E, Cremer M, Neve J. Deficiency in antioxidant factors in patients with alcohol-related chronic pancreatitis. Dig Dis Sci 41: 1225-1231, 1996. PMID: 8654156
- Vaona B, Stanzial A.M, Talamini G, Bovo P, Corrocher R, Cavallini G. Serum selenium concentrations in chronic pancreatitis and controls. DIg Liver Dis 37: 522-525, 2005. PMID: 15975540
- Veghelyi P.V, Kemeny T.T, Pozsonyi J, Sos J. Toxic lesions of the pancreas. Am J Dis Child 80: 390-403, 1950. PMID: 14770454
- Veghelyi P.V, Kemeny T.T, Pozsonyi J, Sos J. Dietary lesions of the pancreas.Am J Dis Child 79: 658-665, 1950.
- Verlaan R, Roelofs H.M, van-Schaik A, Wanten G.J, Jansen J.B, Peters W.H, Drenth J.P. Assessment of oxidative stress in chronic pancreatitis patients. World J Gastroenterol 12: 5705-5710, 2006. PMID: 17007026
- Wacke R, Kirchner A, Prall F, Nizze H, Schmidt W, Fischer U, Nitschke F-P, Adam U, Fritz P, Belloc C, Drewelow B. Up-regulaton of cytochrome P4501A2 2C9 and 2E1 in chronic pancreatitis. Pancreas 16: 521-528, 1998. PMID: 9598815
- Wagner C. Some more comments on ‘folate deficiency in chronic pancreatitis’. JOP 11: 646-647, 2010.
- Wallig M. Xenobiotic metabolism, oxidant stress and chronic pancreatitis. Digestion 59 (suppl 4):13-24, 1998. PMID: 9832632
- Whitcomb D.C. Framework for interpretation of genetic variations in pancreatitis patients. Front Physiol 3: 440, 2012.
- Whiteman M, Le Trionnaire S, Chopra M, Fox B, Whatmore J. Emerging role of hydrogen sulphide in health and disease: critical appraisal of biomarkers and pharmacological tools. Clin Sci (Lond)121: 459-488, 2011. PMID: 21843150
- Winklhofer-Roob B, Tiran B, Tuchschmid P.E, Van’t Hof M.A, Shmerling D.H. Effects of pancreatic enzyme preparations on erythrocyte glutathione peroxidase activities and plasma selenium concentrations in cystic fibrosis. Free Radic Biol Med 25: 242-249, 1998. PMID: 9667502
- Xu Y, Szép S, Lu Z. The antioxidant role of thiocyanate in the pathogenesis of cystic fibrosis and other inflammation-related diseases. PNAS 106: 20515-20519, 2009. PMID: 19918082
- Yadav S, Day J.P, Mohan V, Snehalatha C, Braganza J.M. Selenium and diabetes in the tropics. Pancreas 5: 528-533, 1991. PMID: 1946309
- Yadav D, O’Connell M, Papachristou G.I. Natural history following the first attack of acute pancreatitis. Am J Gastroenterol 107: 1096-1103, 2012. PMID: 22613906
- Zhang K. Integration of ER stress, oxidative stress and the inflammatory response in health and disease. Int J Clin Exp Med 3: 33-40, 2010. PMID: 20369038
- Zhou D, Wang W, Cheng X, Wei J, Zheng S. Antioxidant therapy for patients with chronic pancreatitis: A systematic review and meta-analysis. Clin Nutr 2014. PMID: 25035087
- Zimniak P. Relationship of electrophilic stress to aging. Free Radic Biol Med 51: 1087-1105, 2011. PMID: 21708248


