Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2013.17
| Attachment | Size |
|---|---|
| 667.35 KB |
Introduction
Autoimmune pancreatitis (AIP) has historically been considered a rare disorder, but is increasingly recognized due to an improved understanding of its diverse nature and proper means of diagnosis. The current international consensus diagnostic criteria (ICDC) for the diagnosis of AIP incorporate 5 cardinal features: imaging characteristics of the pancreas (parenchyma and duct), serology, other organ involvement, pancreatic histology, and response to steroids (46). Imaging techniques recognized in the guidelines include computed tomography (CT), magnetic resonance imaging (MRI), magnetic resonance cholangiopancreatography (MRCP), and endoscopic retrograde cholangiopancreatography (ERCP). Endoscopic ultrasound (EUS) is notably absent from the diagnostic algorithms.
Even when the diagnosis of AIP is strongly considered, the diagnosis often remains elusive (26, 49, 54). Despite the use of existing diagnostic algorithms, there is often a significant delay in the diagnosis and use of unnecessary interventions, including pancreatic resection. In addition, some patients remain undiagnosed, leading to diagnostic steroid trials that often contribute to further diagnostic confusion and risk patient safety. With these uncertainties, further refinement of the current consensus diagnostic criteria may be beneficial for some cases.
There are emerging data suggesting the potential utility of EUS in the diagnosis of AIP (14, 15, 34, 38, 41). EUS not only has the ability to provide high-definition imaging of the pancreas, but it also has the ability to acquire tissue through either fine needle aspiration (FNA) or trucut biopsy (TCB). These characteristics help to make it one of the most useful techniques in the diagnosis of pancreatic cancer and chronic pancreatitis (4, 5, 25, 28). Therefore, EUS has the potential to play a role in the both diagnosis of AIP and exclusion of other pancreatic diseases.
Endoscopic ultrasound imaging features
Standard EUS Imaging
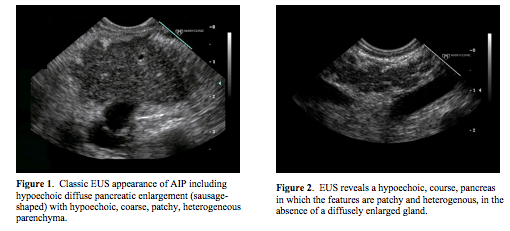
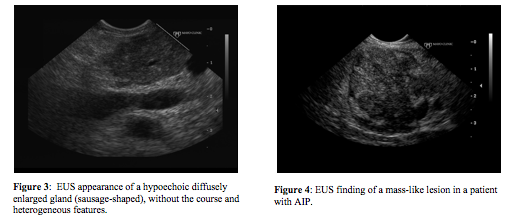
There are no pathognomonic EUS imaging findings of AIP. The “classic” appearance is a diffusely enlarged gland (“sausage-shaped”) with hypoechoic, patchy, heterogenous appearing parenchyma (Figure 1) (3, 9, 14). In our experience, when a patient has all of these classic features, which may be found in up to 57% of patients, there is a high probability of AIP (9, 14). However, patients often are not found to have all of the features, limiting the diagnosis of AIP using EUS (Figures 2 & 3). Another pancreatic finding on EUS is a focal solitary mass (Figure 4). The hypoechoic lesion is commonly located in the head of the pancreas resulting in obstructive jaundice. The mass may appear to invade adjacent vessels, cause upstream dilation of the main pancreatic duct (MPD), and be associated with enlarged peripancreatic lymph nodes, mimicking locally advanced pancreatic cancer (Figure 5) (3, 9, 14). In areas of pancreatic involvement, the MPD may be narrowed with duct wall thickening (3). Also, EUS features of the pancreatic parenchyma may overlap with some characteristics seen in chronic pancreatitis including the presence of hyperechoic foci, hyperechoic strands, and lobularity (Figure 6). In a case series with patients given steroid therapy, the parenchymal enlargement, lobularity, and lobular outer margins improved with steroid treatment while the hyperechoic foci and strands remained (43). Finally, EUS may demonstrate a normal appearing pancreas.

As the biliary tree is the most common extrapancreatic organ involvement in AIP, the extrahepatic duct may be abnormal on EUS. In a study of 37 patients with AIP, ultrasonographic findings of extrahepatic bile duct and gallbladder wall thickening was seen in 38%. There were 2 types of bile duct wall thickening including a “3-layer type” with a high-low-high echo appearance and a “parenchymal-echo type” with a thickened wall throughout the entire bile lumen and a parenchymal echo present within the bile duct itself (30). A similar appearance to the “3-layer type” with a regular homogenous thickening with a hyper-hypo-hyperechoic series of layers of the ductal wall (termed “sandwich pattern”) was seen on EUS in addition to bile duct dilatation in one series (9). This EUS appearance is different than what is often seen with pancreaticobiliary malignancies, which may be more irregular.
It is important to distinguish focal AIP from the dreaded pancreatic cancer. Hoki et al compared EUS findings in patients who were diagnosed with AIP and resected pancreatic cancer (21). This study found that diffuse hypoechoic areas, diffuse enlargement of the pancreas, bile duct wall thickening, and peripancreatic hypoechoic margins were more commonly seen in patients ultimately diagnosed with AIP as compared to those with pancreatic cancer. On the other hand, focal hyperechoic areas and focal enlargement were more common in patients with pancreatic cancer. Although all comparisons reached statistical significance, each characteristic (other than peripancreatic hypoechoic margins) was seen in both diseases. In addition, lymph node enlargement was seen with similar frequencies in AIP and pancreatic cancer.
To our knowledge, there are no studies that have directly compared EUS to other imaging modalities such as CT scan, MRI, or ERCP for the diagnosis of AIP. Therefore, it is unclear as to the additive value of EUS imaging to the other imaging techniques. However, a cohort of 48 patients seen at Mayo Clinic Rochester with a diagnosis of AIP using HISORt criteria (Histology, Imaging, Serology, Other organ involvement, and Response to steroid therapy) underwent EUS with TCB (7, 34, 36). The diagnosis of AIP was strongly suspected in 14 patients prior to EUS based on their clinical, laboratory, and imaging findings. In 22 patients, the diagnosis was considered as a part of a broader differential prior to EUS and in remaining 12 patients the EUS appearance alone led to the initial suspicion of AIP. Therefore, this suggests that EUS imaging alone may increase the diagnostic accuracy of AIP in patients who underwent other imaging modalities without a definitive diagnosis.
Image-Enhancing Techniques in EUS
With the lack of pathognomonic features and diverse spectrum of EUS imaging findings in patients with AIP, several image-enhancing techniques have been evaluated to improve diagnostic accuracy. Each of these complementary imaging methods is in the experimental phase and cannot be recommended to be routinely used in the diagnostic algorithm for AIP until further studies determine its role.
One image-enhancing technique is EUS elastography, which distinguishes tissues based on their stiffness by measuring tissue strain while slightly compressing an area that encompasses both the abnormal and normal tissue (17). Five patients with focal AIP were found to have a homogenous stiff (blue) pattern in the mass and throughout the entire pancreas, which differed from pancreatic cancer or normal pancreas in which the pancreatic parenchyma was predominately of intermediate stiffness (green) (12).
Contrast-enhanced EUS uses intravenously administered ultrasound contrast agents [e.g., Sonovue (sulfur hexafluoride MBs; Bracco Interventional BV, Amsterdam), Levovist (Bayer AG, Leverkusen, Germany), or Sonazoid (perfluorobutane; GE Healthcare, Little Chalfont, Buckinghamsire, UK)] to produce microbubbles that allow visualization of the vascular pattern within the pancreatic mass lesion (17). In an cohort of 10 patients who received Sonovue contrast and EUS imaging in the bicolor Doppler mode, AIP was associated with hypervascularity within the mass-like lesion and the surrounding pancreatic parenchyma as compared to pancreatic cancer where the mass was hypovascular in comparison to the surrounding pancreatic tissue (20).
Contrast-enhanced harmonic EUS is similar to the technique described above but uses a dedicated contrast harmonic mode rather than Doppler imaging. The use of contrast harmonic-enhanced imaging allows for decreased artifact produced by the Doppler, including ballooning and overpainting (17). In one study, 8 patients with focal AIP and 22 patients with pancreatic cancer were given Sonazoid ultrasonographic contrast and analyzed using a radial echoendoscope with the conventional tissue harmonic echo (for standard harmonic imaging) and extended pure harmonic detection (for contrast-enhanced harmonic imaging) (22). The ultrasonographic contrast uptake and distribution was isoehanced and homogenous in all patients with AIP compared to only 1 patient with pancreatic cancer. The majority of patients with pancreatic cancer had hypoenhanced uptake in a heterogenous pattern. Furthermore, the optimal maximum intensity gain (MIG) cutoff value to differentiate between AIP and pancreatic cancer with a 100% specificity and sensitivity using a receiver operator characteristic (ROC) curve was 12.5. All of the results from the studies above must be interpreted with caution as additional studies are required to confirm the utility of the image-enhancing techniques in differentiating between AIP and pancreatic cancer.
Endoscopic ultrasound-guided tissue acquisition
Fine Needle Aspiration
EUS imaging itself has not proven to be useful when used in isolation to diagnose AIP. Although the role of EUS-guided tissue acquisition has not been extensively studied, pancreatic histology is recognized as an important diagnostic criterion in the ICDC. Despite a few reports on the ability to diagnose AIP using FNA only, there are no broadly accepted consensus cytological diagnostic criteria for AIP and most pathologists are reluctant to rely solely on FNA specimens (6, 10, 27, 40). FNA commonly yields small specimen samples and results in a loss of tissue architecture, making its interpretation challenging. Even EUS-guided FNA using a 19-gauge needle for histological review was only able to achieve a diagnosis of AIP in 43% of patients (24).
Due to the inability to obtain adequate core specimens using standard FNA needles, some advocate for the use of less rigorous or incomplete pathology criteria for the cytologic diagnosis of AIP. For example, the less stringent criteria may rely on the presence of a lymphoplasmacytic infiltrate alone without the requirement to find the infiltrate positioned in a periductal location or the degree of preservation of ductules, venules or arterioles required within the specimen (6, 10, 40). Although lowering the requirements of the pathologic criteria may improve the diagnostic sensitivity, it is at the expense of decreasing the specificity of FNA for AIP. This is particularly problematic for differentiating AIP from pancreatic cancer, which is often associated with a lymphoplasmacytic infiltration.
Some suggest that the benefit of EUS-guided FNA lies on its ability to exclude pancreatic cancer rather than diagnose AIP (41, 42, 48). However, assuming that a negative EUS FNA of a pancreas mass equates to exclusion of an underlying malignancy can be dangerous given the 10-40% false negative FNA rate for cancer (8, 13, 39, 50, 51).
Trucut Biopsy
To overcome the limitations of FNA needles, larger caliber cutting biopsy needles have been developed that acquire samples with preserved tissue architecture, allowing for histological examination (1, 2, 19, 23, 29, 31, 44, 52). An EUS TCB device (Quick-Core, Wilson-Cook, Winston-Salem, NC) uses a 19-gauge needle with a tissue tray and sliding sheath that is designed for the capture of a core tissue sample. This device has been shown to be useful for the diagnosis of neoplasms that are often difficult to diagnose based on cytopathology alone including stromal tumors and lymphoma when immunohistochemical analysis is useful, or well-differentiated desmoplastic tumors that make aspiration difficult (11, 18, 32, 33, 35, 37, 45, 53). Furthermore, with the larger specimen size and the ability to preserve tissue architecture, TCB has been shown to help differentiate between AIP, usual chronic pancreatitis, and pancreatic cancer (47, 54).
We looked at the previous Mayo Clinic experience regarding the diagnostic sensitivity and safety of EUS TCB in patients with final diagnosis of AIP based on the HISORt criteria (unpublished data). Forty-eight patients (38 male, mean age 59.7 years) in whom a mean of 2.9 EUS TCB (range 1-7) were performed. Histologic examination of the EUS TCB specimens provided a diagnosis in 35 patients (73%). The diagnostic sensitivity varied among the 5 endosonographers from 33-90%. Non-diagnostic cases were found to have chronic pancreatitis (n=8), non-specific histology (n=2), or a failed tissue acquisition (n=3). Complications included mild transient abdominal pain (n=3) and self-limited intra-procedural bleeding (n=1). It is unclear if TCB and/or FNA can be attributed to these complications. No patient required hospitalization or therapeutic intervention. Of note, the serum IgG4 was >2x the upper limit of normal in only 23% of patients. None of the patients with EUS TCB diagnosis of AIP required surgical intervention for diagnosis. Over a mean follow-up of 2.6 years, no false negative diagnoses of pancreatic cancer were identified. Prior to EUS, the diagnosis of AIP was strongly suspected in 14 patients as a result of their clinical, laboratory, or imaging findings. For 22 patients, the diagnosis was considered pre-EUS as part of a broader differential. Our data suggest the potential utility of EUS imaging to the initial suspicion of AIP in 12 patients, thereby initiating pancreatic TCB and subsequent clinical evaluation of AIP. More recently, we looked at the use of EUS TCB in pediatric patients with a suspected diagnosis of AIP (16). The diagnostic yield of EUS TBC in this patient population was 87%.
EUS TCB appears to be safe and may provide sufficient material to aid in the diagnosis of AIP, thereby guiding treatment and avoiding surgical intervention. Some suggest the use of EUS TCB as a “rescue” technique to obtain adequate tissue samples if EUS FNA failed (34, 40). The current ICDC guidelines recommend a pancreatic core biopsy in patients presenting with a focal mass and/or obstructive jaundice if cancer has been excluded and the diagnosis remains elusive (46).
ProCore biopsy
The ProCore needle has a lateral bevel (Cook Medical Inc., Bloomington, IN) that may occasionally provide a sufficient specimen to allow histologic analysis and diagnosis of AIP. However, studies on the use of the ProCore needle in the diagnosis of AIP are lacking and in our experience has been inferior to EUS TCB in this setting.
Summary
Although personal opinion and limited data suggest that EUS imaging alone may improve the diagnosis of AIP, there are few studies to substantiate this view. Despite early promise, the utility and role of elastography, contrast-enhanced EUS, and harmonic imaging in patients with AIP remains to be determined. The lack of pathognomonic EUS imaging characteristics and diverse spectrum of both the clinical presentation and pancreatic findings of AIP emphasize the need for a safe and reliable way to acquire tissue specimens, particularly in cases with atypical features.
While FNA cytologic samples can be examined for the presence of lymphocytes and plasma cells, other disorders may also have a similar appearance. This limits the specificity of FNA and risks inappropriate management of patients who may have unrecognized pancreatic cancer. Therefore, until data suggests otherwise, it is not recommended to rely on FNA to diagnose AIP. Instead, core biopsies using EUS TCB should be used for histologic examination and IgG4 immunostaining. We perform EUS TCB in patients with a compatible clinical presentation but the diagnosis remains uncertain and when the findings are likely to alter management. By performing EUS TCB, pancreatic cancer may be excluded and unnecessary surgical intervention may be averted. Unfortunately, it may not be possible to obtain pancreatic core biopsies in all patients with an indeterminate diagnosis due to technical, anatomical, or personnel limitations. In such patients, it is even more critical to consider all possible diagnostic components of the ICDC to attempt to establish a diagnosis without histological evaluation. Further study is needed to determine the diagnostic yield of EUS imaging alone, newer imaging-enhancing techniques, and FNA or TCB for AIP.
References:
- Ball AB, Fisher C, Pittam M, Watkins RM, and Westbury G. Diagnosis of soft tissue tumours by Tru-Cut biopsy. Br J Surg 77: 756-758, 1990. PMID: 2383749
- Brandt KR, Charboneau JW, Stephens DH, Welch TJ, and Goellner JR. CT- and US-guided biopsy of the pancreas. Radiology 187: 99-104, 1993. PMID: 8451443
- Buscarini E, Lisi SD, Arcidiacono PG, Petrone MC, Fuini A, Conigliaro R, Manfredi G, Manta R, Reggio D, and Angelis CD. Endoscopic ultrasonography findings in autoimmune pancreatitis. World J Gastroenterol 17: 2080-2085, 2011. PMID: 21547126
- Catanzaro A, Richardson S, Veloso H, Isenberg GA, Wong RC, Sivak MV, Jr., and Chak A. Long-term follow-up of patients with clinically indeterminate suspicion of pancreatic cancer and normal EUS. Gastrointest Endosc 58: 836-840, 2003. PMID: 14652549
- Chang DK, Nguyen NQ, Merrett ND, Dixson H, Leong RW, and Biankin AV. Role of endoscopic ultrasound in pancreatic cancer. Expert Rev Gastroenterol Hepatol 3: 293-303, 2009. PMID: 19485810
- Chari ST, Kloeppel G, Zhang L, Notohara K, Lerch MM, and Shimosegawa T. Histopathologic and clinical subtypes of autoimmune pancreatitis: the Honolulu consensus document. Pancreas 39: 549-554, 2010. PMID: 21242705
- Chari ST, Smyrk TC, Levy MJ, Topazian MD, Takahashi N, Zhang L, Clain JE, Pearson RK, Petersen BT, Vege SS, and Farnell MB. Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin Gastroenterol Hepatol 4: 1010-1016; quiz 1934, 2006. PMID: 16843735
- Chen J, Yang R, Lu Y, Xia Y, and Zhou H. Diagnostic accuracy of endoscopic ultrasound-guided fine-needle aspiration for solid pancreatic lesion: a systematic review. J Cancer Res Clin Oncol 138: 1433-1441, 2012. PMID: 22752601
- De Lisi S, Buscarini E, Arcidiacono PG, Petrone M, Menozzi F, Testoni PA, and Zambelli A. Endoscopic ultrasonography findings in autoimmune pancreatitis: be aware of the ambiguous features and look for the pivotal ones. Jop 11: 78-84, 2010. PMID: 20065561
- Deshpande V, Mino-Kenudson M, Brugge WR, Pitman MB, Fernandez-del Castillo C, Warshaw AL, and Lauwers GY. Endoscopic ultrasound guided fine needle aspiration biopsy of autoimmune pancreatitis: diagnostic criteria and pitfalls. Am J Surg Pathol 29: 1464-1471, 2005. PMID: 16224213
- DeWitt J, Emerson RE, Sherman S, Al-Haddad M, McHenry L, Cote GA, and Leblanc JK. Endoscopic ultrasound-guided Trucut biopsy of gastrointestinal mesenchymal tumor. Surg Endosc 25: 2192-2202, 2011. PMID: 21184105
- Dietrich CF, Hirche TO, Ott M, and Ignee A. Real-time tissue elastography in the diagnosis of autoimmune pancreatitis. Endoscopy 41: 718-720, 2009. PMID: 19618344
- Eloubeidi MA and Tamhane A. EUS-guided FNA of solid pancreatic masses: a learning curve with 300 consecutive procedures. Gastrointest Endosc 61: 700-708, 2005. PMID: 15855975
- Farrell JJ, Garber J, Sahani D, and Brugge WR. EUS findings in patients with autoimmune pancreatitis. Gastrointest Endosc 60: 927-936, 2004. PMID: 15605008
- Finkelberg DL, Sahani D, Deshpande V, and Brugge WR. Autoimmune pancreatitis. N Engl J Med 355: 2670-2676, 2006. PMID: 17182992
- Fujii LL, Chari ST, El-Youssef M, Takahashi N, Topazian MD, Zhang L, and Levy MJ. Pediatric pancreatic EUS-guided trucut biopsy for evaluation of autoimmune pancreatitis. Gastrointest Endosc 77: 824-828, 2013. PMID: 23433594
- Fusaroli P, Saftoiu A, Mancino MG, Caletti G, and Eloubeidi MA. Techniques of image enhancement in EUS (with videos). Gastrointest Endosc 74: 645-655, 2011. PMID: 21679945
- Gines A, Wiersema MJ, Clain JE, Pochron NL, Rajan E, and Levy MJ. Prospective study of a Trucut needle for performing EUS-guided biopsy with EUS-guided FNA rescue. Gastrointest Endosc 62: 597-601, 2005. PMID: 16185976
- Harrison BD, Thorpe RS, Kitchener PG, McCann BG, and Pilling JR. Percutaneous Trucut lung biopsy in the diagnosis of localised pulmonary lesions. Thorax 39: 493-499, 1984. PMID: 6463928
- Hocke M, Ignee A, and Dietrich CF. Contrast-enhanced endoscopic ultrasound in the diagnosis of autoimmune pancreatitis. Endoscopy 43: 163-165, 2011. PMID: 22139794
- Hoki N, Mizuno N, Sawaki A, Tajika M, Takayama R, Shimizu Y, Bhatia V, and Yamao K. Diagnosis of autoimmune pancreatitis using endoscopic ultrasonography. J Gastroenterol 44: 154-159, 2009. PMID: 19214678
- Imazu H, Kanazawa K, Mori N, Ikeda K, Kakutani H, Sumiyama K, Hino S, Ang TL, Omar S, and Tajiri H. Novel quantitative perfusion analysis with contrast-enhanced harmonic EUS for differentiation of autoimmune pancreatitis from pancreatic carcinoma. Scand J Gastroenterol 47: 853-860, 2012. PMID: 22507131
- Ingram DM, Sheiner HJ, and Shilkin KB. Operative biopsy of the pancreas using the Trucut needle. Aust N Z J Surg 48: 203-206, 1978. PMID: 280329
- Iwashita T, Yasuda I, Doi S, Ando N, Nakashima M, Adachi S, Hirose Y, Mukai T, Iwata K, Tomita E, Itoi T, and Moriwaki H. Use of samples from endoscopic ultrasound-guided 19-gauge fine-needle aspiration in diagnosis of autoimmune pancreatitis. Clin Gastroenterol Hepatol 10: 316-322, 2012. PMID: 22019795
- Kahl S, Glasbrenner B, Leodolter A, Pross M, Schulz HU, and Malfertheiner P. EUS in the diagnosis of early chronic pancreatitis: a prospective follow-up study. Gastrointest Endosc 55: 507-511, 2002. PMID: 11923762
- Kamisawa T, Egawa N, Nakajima H, Tsuruta K, Okamoto A, and Kamata N. Clinical difficulties in the differentiation of autoimmune pancreatitis and pancreatic carcinoma. Am J Gastroenterol 98: 2694-2699, 2003. PMID: 14687819
- Kanno A, Ishida K, Hamada S, Fujishima F, Unno J, Kume K, Kikuta K, Hirota M, Masamune A, Satoh K, Notohara K, and Shimosegawa T. Diagnosis of autoimmune pancreatitis by EUS-FNA by using a 22-gauge needle based on the International Consensus Diagnostic Criteria. Gastrointest Endosc 76: 594-602, 2012. PMID: 22898417
- Klapman JB, Chang KJ, Lee JG, and Nguyen P. Negative predictive value of endoscopic ultrasound in a large series of patients with a clinical suspicion of pancreatic cancer. Am J Gastroenterol 100: 2658-2661, 2005. PMID: 16393216
- Kovalik EC, Schwab SJ, Gunnells JC, Bowie D, and Smith SR. No change in complication rate using spring-loaded gun compared to traditional percutaneous renal allograft biopsy techniques. Clin Nephrol 45: 383-385, 1996. PMID: 8793230
- Koyama R, Imamura T, Okuda C, Sakamoto N, Honjo H, and Takeuchi K. Ultrasonographic imaging of bile duct lesions in autoimmune pancreatitis. Pancreas 37: 259-264, 2008. PMID: 18815546
- Lavelle MA and O'Toole A. Trucut biopsy of the prostate. Br J Urol 73: 600, 1994. PMID: 8012797
- Lee JH, Choi KD, Kim MY, Choi KS, Kim do H, Park YS, Kim KC, Song HJ, Lee GH, Jung HY, Yook JH, Kim BS, Kang YK, and Kim JH. Clinical impact of EUS-guided Trucut biopsy results on decision making for patients with gastric subepithelial tumors >/= 2 cm in diameter. Gastrointest Endosc 74: 1010-1018, 2011. PMID: 21889136
- Levy MJ, Jondal ML, Clain J, and Wiersema MJ. Preliminary experience with an EUS-guided trucut biopsy needle compared with EUS-guided FNA. Gastrointest Endosc 57: 101-106, 2003. PMID: 12518144
- Levy MJ, Reddy RP, Wiersema MJ, Smyrk TC, Clain JE, Harewood GC, Pearson RK, Rajan E, Topazian MD, Yusuf TE, Chari ST, and Petersen BT. EUS-guided trucut biopsy in establishing autoimmune pancreatitis as the cause of obstructive jaundice. Gastrointest Endosc 61: 467-472, 2005. PMID: 15758927
- Levy MJ, Smyrk TC, Reddy RP, Clain JE, Harewood GC, Kendrick ML, Pearson RK, Petersen BT, Rajan E, Topazian MD, Wang KK, Wiersema MJ, Yusuf TE, and Chari ST. Endoscopic ultrasound-guided trucut biopsy of the cyst wall for diagnosing cystic pancreatic tumors. Clin Gastroenterol Hepatol 3: 974-979, 2005. PMID: 16234042
- Levy MJ, Smyrk TC, Takahashi N, Zhang L, and Chari ST. Idiopathic duct-centric pancreatitis: disease description and endoscopic ultrasonography-guided trucut biopsy diagnosis. Pancreatology 11: 76-80, 2011. PMID: 21525775
- Levy MJ and Wiersema MJ. EUS-guided Trucut biopsy. Gastrointest Endosc 62: 417-426, 2005. PMID: 16111962
- Levy MJ, Wiersema MJ, and Chari ST. Chronic pancreatitis: focal pancreatitis or cancer? Is there a role for FNA/biopsy? Autoimmune pancreatitis. Endoscopy 38 Suppl 1: S30-35, 2006. PMID: 16802220
- Mitsuhashi T, Ghafari S, Chang CY, and Gu M. Endoscopic ultrasound-guided fine needle aspiration of the pancreas: cytomorphological evaluation with emphasis on adequacy assessment, diagnostic criteria and contamination from the gastrointestinal tract. Cytopathology 17: 34-41, 2006. PMID: 16417563
- Mizuno N, Bhatia V, Hosoda W, Sawaki A, Hoki N, Hara K, Takagi T, Ko SB, Yatabe Y, Goto H, and Yamao K. Histological diagnosis of autoimmune pancreatitis using EUS-guided trucut biopsy: a comparison study with EUS-FNA. J Gastroenterol 44: 742-750, 2009. PMID: 19434362
- Moon SH and Kim MH. The role of endoscopy in the diagnosis of autoimmune pancreatitis. Gastrointest Endosc 76: 645-656, 2012. PMID: 24079796
- Naitoh I, Nakazawa T, Hayashi K, Okumura F, Miyabe K, Shimizu S, Kondo H, Yoshida M, Yamashita H, Ohara H, and Joh T. Clinical differences between mass-forming autoimmune pancreatitis and pancreatic cancer. Scand J Gastroenterol 47: 607-613, 2012. PMID: 22416894
- Okabe Y, Ishida Y, Kaji R, Sugiyama G, Yasumoto M, Naito Y, Toyonaga A, Tsuruta O, and Sata M. Endoscopic ultrasonographic study of autoimmune pancreatitis and the effect of steroid therapy. J Hepatobiliary Pancreat Sci 19: 266-273, 2012. PMID: 21671062
- Piccinino F, Sagnelli E, Pasquale G, and Giusti G. Complications following percutaneous liver biopsy. A multicentre retrospective study on 68,276 biopsies. J Hepatol 2: 165-173, 1986. PMID: 3958472
- Saftoiu A, Vilmann P, Guldhammer Skov B, and Georgescu CV. Endoscopic ultrasound (EUS)-guided Trucut biopsy adds significant information to EUS-guided fine-needle aspiration in selected patients: a prospective study. Scand J Gastroenterol 42: 117-125, 2007. PMID: 17190771
- Shimosegawa T, Chari ST, Frulloni L, Kamisawa T, Kawa S, Mino-Kenudson M, Kim MH, Kloppel G, Lerch MM, Lohr M, Notohara K, Okazaki K, Schneider A, and Zhang L. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas 40: 352-358, 2011. PMID: 21412117
- Suda K, Takase M, Fukumura Y, Ogura K, Ueda A, Matsuda T, and Suzuki F. Histopathologic characteristics of autoimmune pancreatitis based on comparison with chronic pancreatitis. Pancreas 30: 355-358, 2005. PMID: 15841047
- Takuma K, Kamisawa T, Gopalakrishna R, Hara S, Tabata T, Inaba Y, Egawa N, and Igarashi Y. Strategy to differentiate autoimmune pancreatitis from pancreas cancer. World J Gastroenterol 18: 1015-1020, 2012. PMID: 22416175
- Taniguchi T, Tanio H, Seko S, Nishida O, Inoue F, Okamoto M, Ishigami S, and Kobayashi H. Autoimmune pancreatitis detected as a mass in the head of the pancreas without hypergammaglobulinemia, which relapsed after surgery: case report and review of the literature. Dig Dis Sci 48: 1465-1471, 2003. PMID: 12924637
- Turner BG, Cizginer S, Agarwal D, Yang J, Pitman MB, and Brugge WR. Diagnosis of pancreatic neoplasia with EUS and FNA: a report of accuracy. Gastrointest Endosc 71: 91-98, 2010. PMID: 19846087
- Voss M, Hammel P, Molas G, Palazzo L, Dancour A, O'Toole D, Terris B, Degott C, Bernades P, and Ruszniewski P. Value of endoscopic ultrasound guided fine needle aspiration biopsy in the diagnosis of solid pancreatic masses. Gut 46: 244-249, 2000. PMID: 10644320
- Welch TJ, Sheedy PF, 2nd, Johnson CD, Johnson CM, and Stephens DH. CT-guided biopsy: prospective analysis of 1,000 procedures. Radiology 171: 493-496, 1989. PMID: 2704815
- Wiersema MJ, Levy MJ, Harewood GC, Vazquez-Sequeiros E, Jondal ML, and Wiersema LM. Initial experience with EUS-guided trucut needle biopsies of perigastric organs. Gastrointest Endosc 56: 275-278, 2002. PMID: 12145612
- Yadav D, Notahara K, Smyrk TC, Clain JE, Pearson RK, Farnell MB, and Chari ST. Idiopathic tumefactive chronic pancreatitis: clinical profile, histology, and natural history after resection. Clin Gastroenterol Hepatol 1: 129-135, 2003. PMID: 15017505


