Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2015.2
| Attachment | Size |
|---|---|
| 502.79 KB |
1. Introduction
Pancreatic cancer and diabetes have a complex bidirectional relationship. A large proportion (varied between 50%–80% by studies) of pancreatic cancer patients have concurrent diabetes or impaired glucose tolerance (15, 54). It has been debated for a long time whether diabetes is a predisposing risk factor for the development of pancreatic cancer or a consequence of disease onset (24, 37, 66). Because of the relatively low incidence of pancreatic cancer compared to other common cancers and the rapid fatality of this aggressive malignancy, epidemiological investigations faced many challenges. For example, early studies conducted by single institution often have limited study power. Population based case-control studies are easily subject to selection bias or information bias due to the rapid loss of patients with the most advanced disease. Misclassification bias seems unavoidable because there is no current clinical or laboratory methods to distinguish the type 2 diabetes from the pancreatic cancer caused type 3c diabetes (15). As a consequence of the obesity epidemic, the incidence of diabetes is increasing globally, which may have a significant impact on the pancreatic cancer burden. A better understanding of the association between diabetes and pancreatic cancer and the mechanism underlying this association would aid the development of novel strategies for the prevention and treatment of this cancer.
2. Epidemiological evidence
Up to date, more than 20 case-control studies (9, 11, 16, 20, 23, 28, 31, 37, 38, 44, 46, 48, 50, 56, 59, 65, 67, 93, 101) and the same number of cohort or nested case-control studies (1, 3, 5, 6, 13, 14, 27, 39, 43, 47, 55, 58, 63, 73, 76, 77, 78, 84, 94, 95) with information on the association between diabetes and pancreatic cancer have been reported. Findings from these individual studies were inconsistent. However, a consistent association of long-term diabetes and risk of pancreatic cancer was reported from three large scale meta-analyses (7, 22, 42) and three pooled data analyses (10 21, 53) with each involved more than 1500 pancreatic cancer cases (Table 1). The first meta-analysis (22), conducted in 1995, included 20 of the 40 published case-control and cohort studies and reported an overall estimated relative risk (RR) of 2.1 and a 95% confidence interval (CI) 1.6-2.8. This value was relatively unchanged when analyses were restricted to diabetes with a duration of at least 5 years (RR, 2.0, 95% CI, 1.2-3.2) (22). The second, conducted in 2005, included 17 case-control and 19 cohort or nested case-control studies published from 1996 to 2005 and reported an overall RR of 1.8 and 95% CI (1.7-1.9) (42). Individuals diagnosed with diabetes within 4 years of their pancreatic cancer had a 50% greater risk than those with diabetes for ≥5 years (RR, 2.1, 95% CI, 1.9-2.3 vs. RR, 1.5, 95%CI,1.3-1.8; P = 0.005). The third meta-analysis of 35 cohort studies reported a summary RR (95% CI) of 1.94 (1.66-2.27) (7). Subgroup analyses revealed that the
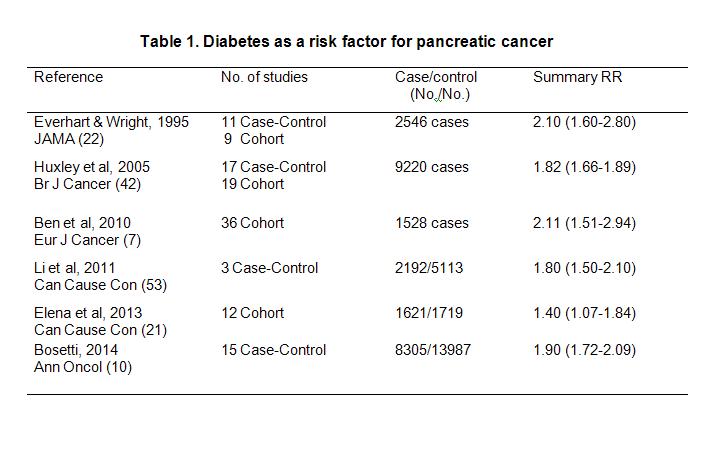
increased risk of pancreatic cancer was independent of geographic locations, sex, study design, alcohol consumption, body mass index (BMI) and smoking status. Similarly, a negative association was observed for diabetes duration and risk of pancreatic cancer.
In a pooled analysis of 2,192 pancreatic cancer patients and 5,113 cancer-free controls from three large case-control studies conducted in the United States, diabetes was associated with a 1.8-fold risk of pancreatic cancer (95% CI,1.5-2.1) (53). Risk estimates decreased with increasing years with diabetes. Individuals with diabetes for ≤2, 3-5, 6-10, 11-15 and >15 years had an RR (95% CI) of 2.9 (2.1-3.9), 1.9 (1.3-2.6), 1.6 (1.2-2.3), 1.3 (0.9-2.0), and 1.4 (1.0-2.0), respectively, (P <0.0001 for trend) (53). A pooled data analysis conducted by the Pancreatic Cancer Cohort Consortium in 1621 pancreatic cases and 1719 matched controls from 12 prospective cohorts reported that self-reported diabetes was associated with a 40% increased risk of pancreatic cancer (95% CI, 1.07-1.84) (21). The highest risk was for those with diabetes duration 2-8 years (RR,1.79, 95% CI, 1.25-2.55), but no association was found for those with >9 years of diabetes duration. The latest study reported by the Pancreatic Cancer Case Control Consortium involved 8305 cases and 13,987 controls pooled from 15 case-control studies (10). Overall, 1155 (15%) cases and 1087 (8%) controls reported a diagnosis of diabetes two or more years before cancer diagnosis (or interview, for controls), corresponding to a RR of 1.90 (95% CI, 1.72-2.09). Pancreatic cancer risk decreased with duration of diabetes, but a significant excess risk was still evident 20 or more years after diabetes diagnosis (RR, 1.30, 95% CI, 1.17-2.03).
One of the major concerns regarding the diabetes and pancreatic cancer associations is the reverse causality issue because pancreatic cancer could cause diabetes or diabetic state. The associations observed between risk of pancreatic cancer and long-term diabetes (> 5 years of duration) could not be explained by reverse causality because of the known rapid progression feature of pancreatic cancer. Another concern is whether diabetes is an independent risk factor for pancreatic cancer since 80% of the type 2 diabetes patients are obese and smoking is an established risk factor for both diabetes and pancreatic cancer. In the three pooled analyses, all risk estimates were adjusted for other known risk factors for pancreatic cancer, e.g. smoking, obesity and family history, in addition to age, race and sex. Consistent risk estimates were also observed across strata of body mass index and tobacco smoking (10). Therefore, there is sufficient epidemiological evidence supporting the conclusion that long-term type 2 diabetes is an independent risk factor for pancreatic cancer.
Information on the association of type 1 diabetes and pancreatic cancer is quite limited. Most studies have either been restricted to people with type 2 diabetes or have made no distinction between types of diabetes. A systematic review and meta-analysis conducted in 2007 identified 3 cohort studies and 6 case–control studies with information on type 1 diabetes (82). Based on 39 pancreatic cancer cases, the summary RR for pancreatic cancer in “young-onset” or type I diabetes versus no diabetes was 2.00 (95% CI, 1.37-3.01). Because the number of diabetes diagnosed in younger age (<45 years of age) is increasing (90), the impact of early diabetes onset on risk of pancreatic cancer deserve further investigations.
3. Evidence from biomarker studies
The hypothesis that long-standing diabetes is a risk factor for pancreatic cancer is also supported by results from biomarker studies. A number of studies have examined the serum or plasma levels of biomarkers for hyperglycemia, b-cell function and insulin resistance in relation to risk of pancreatic cancer using prediagnostic blood samples (Table 2).

In three prospective cohort studies with follow-up durations of more than 20 years, an increased risk of pancreatic cancer among subjects with high postload plasma glucose levels was observed (6, 29, 79). In the last of these studies, the risk of pancreatic cancer was 2.2-fold higher in individuals with a postload plasma glucose level greater than 200 mg/dL at baseline than in those with a level of no more than 119 mg/dL (29). Consistent with these findings, two additional studies have also shown a positive association of elevated HbA1C level and increased risk of pancreatic cancer (32, 97). Two studies directly examined the relationship between prediagnostic serum insulin levels and pancreatic cancer risk (83, 97). The first study demonstrated a 2-fold increase in risk after excluding cases diagnosed in the first 5 years of follow-up (RR, 2.01, 95% CI, 1.03–3.93) for the highest versus lowest quartile insulin level (83). The second one showed a RR (95% CI) of 1.57 (1.08-2.30) (97). Also, two studies showed an excessively high risk of pancreatic cancer associated with high levels of circulating proinsulin (C-peptide), a marker for b-cell function (62, 97). The RR (95% CI) was 1.52 (0.87-2.64) and 2.22 (1.50-3.29), respectively. However, this association was not observed in two other studies (32, 100). In the most recent study (97), it was found that the associations with insulin and proinsulin was stronger (highest vs lowest quintile, RR, 2.77, 95% CI,1.28 to 5.99 for insulin and RR, 3.60, 95%CI, 1.68 to 7.72 for proinsulin) in cancers diagnosed 10 or more years after blood collection. In mutually adjusted models including HbA1c, insulin, and proinsulin, only proinsulin remained statistically significant (highest vs lowest quintile, RR, 2.55; 95% CI, 1.54 to 4.21; P < .001 for trend), which suggest that markers of peripheral insulin resistance, rather than hyperglycemia or pancreatic beta-cell dysfunction, were independently associated with pancreatic cancer risk. Furthermore, at least three studies (4, 35, 84) have found a significant association of increased risk of pancreatic cancer with lower plasma level of adiponectin, a key regulator of glucose and fat metabolism and insulin sensitivity. These results indicate that both hyperglycemia markers and insulin resistance markers are associated with increased risk of pancreatic cancer. Although impaired glucose tolerance may precede the onset of pancreatic cancer rather than just be a consequence of this cancer, it is unlikely the altered glucose metabolism was caused by the cancer because all these studies used prediagnostic blood samples that were collected many years before the cancer onset. Findings from the biomarker studies not only provide supporting evidence for the association of diabetes and risk of pancreatic cancer, but also provide clues on the mechanisms underlying the association.
4. Mechanisms of diabetes-associated pancreatic cancer
The mechanism of the association between diabetes and pancreatic cancer is elusive but is known to include metabolic, hormonal, and immunological alterations that influence tumor growth (30). Insulin resistance and compensatory hyperinsulinemia as well as elevated levels of circulating insulin-like growth factors (IGFs) are perhaps the most hypothesized mechanisms underlying the association between type 2 diabetes and pancreatic cancer. Insulin per se is not a carcinogen, but insulin response via insulin receptor-mediated signaling transduction has a known mitogenic and cell proliferation stimulating effect. Insulin could also promote carcinogenesis through its effects on IGF-1. Insulin reduces the hepatic production of IGF binding proteins, which resulting in a higher level of circulating bioactive IGF1. IGF1 receptor-mediated initiation of signal transduction activates important intracellular signal pathways, including the Ras/Raf/mitogen-activated protein kinase and phosphoinositide-3 kinase/Akt/mammalian target of rapamycin (mTOR) pathways (71), both of which are frequently deregulated in pancreatic cancer. Several epidemiological studies failed to demonstrate a significant association of plasma IGFI, IGF2, or IGFBP3 levels and the development of pancreatic cancer (57, 75, 85, 98). However, lower plasma IGFBP1 levels or combination of a higher level of IGF1 and lower level of IGFBP3 were associated with increased risk of pancreatic cancer(75, 100).
In addition to the direct growth-promoting effect of insulin and IGFs, type 2 diabetes may increase the risk of pancreatic cancer by the mechanism of inflammation. The adipose tissues regulate the release of inflammatory cytokines such as TNF-a, IL-6 and anti-inflammatory cytokine, such as IL10 and adiponectin. The pro-inflammatory cytokines not only contribute to insulin resistance, but also play an etiologic role in cell transformation and tumor progression. Furthermore, altered levels or functions of several molecules that are implicated in obesity and/or diabetes, such as NF-κB, leptin (49), IGF1 (80), and peroxisome proliferator-activated receptor γ (PPARG) (96), may contribute to pancreatic cancer development by impairing immune function. Epidemiological studies did not find a significant association of risk of pancreatic cancer with plasma level of C-reactive protein (18, 33) but with an elevated level of TNF-α (33). Two studies have found that the circulating level of advanced glycation end products (AGE) was not but their soluble receptors (sRAGE) were significantly associated with reduced risk of pancreatic cancer (34, 45). AGEs are formed by nonenzymatic reactions of reduced sugars, such as glucose, with amino groups in proteins, lipids, and nucleic acids. Endogenous AGE formation is enhanced under hyperglycemia and AGE triggers rapid generation of intracellular reactive oxygen species and activates an array of key cell-signaling pathways that have been implicated in oncogenesis. AGEs exert their proinflammatory effects by binding to receptors RAGE. sRAGE has AGEs-binding properties but in the absence of intracellular domain that is essential for RAGE signaling, therefore, may neutralize circulating AGEs and protect against RAGE-mediated cascades. The inverse associations of sRAGE and risk of pancreatic cancer support the hypothesis on inflammatory mechanisms.
A recent study has shown that that both obesity and type 2 diabetes are associated with increased pancreatic duct cell replication in humans (12). Using tissues obtained from surgical resection or autopsies the study found a 10-fold higher ductal cell replication in obese patients and 4-fold higher in lean diabetic patients compared with lean non-diabetic controls. Although it is not fully understood how the pancreatic ductal cell replication was regulated, gastrointestinal hormones, with incretin activity in response to food intake may play a role (61). Another hypothesis is that progenitor cells in the pancreas can be activated upon pancreatic injury to form new islet or acinar cells. So it is conceivable that type 2 diabetes may promote increased formation of cells from pancreatic progenitors due to the damage to pancreatic tissues.
5. Genetic factors
Genetic factors that modify the risk of diabetes-associated pancreatic cancer have been investigated in several studies using the candidate gene approach with a focus on genes involved in glucose metabolism (17), obesity or diabetes (26, 69, 72, 88), insulin resistance (87) and inflammatory pathways (19, 74). Some modest effects on risk of pancreatic cancer and possible interaction with diabetes were reported for a number of variants but no replication study has been conducted.
Recent genome wide association studies have identified several pancreas development genes (2), i.e. NR5A2, PDX1, and HNF1A as susceptibility factor for pancreatic cancer (52, 68, 70, 99). Among these genes, heterozygous mutations in PDX1 and HNF1A genes are responsible for type 4 and type 3 of the maturity onset diabetes of the young (MODY), respectively. NR5A2 is known to play a critical role in phosphatidylcholine signalling pathway regulating fatty acid and glucose homeostasis (51). Common variants of these genes have also been associated with risk of obesity (81) and type II diabetes (40, 91) or fasting glucose level (60). On the other hand, emerging evidence suggest that these genes also act as an oncogene or tumor suppressor gene in pancreatic carcinogenesis (8, 25, 41, 92). It is conceivable that genes involved in organ development and differentiation may contribute to the ability of tumor cells to proliferate and evade cell death as well as reprogram progenitor cells to a state that give rise to a tumor (64). Although initial data analysis in a limited sample did not find significant interaction of the pancreas development genes with diabetes in modifying the risk of pancreatic cancer (89), further examination of the GWAS data in adequately powered studies may reveal the genetic factors that predispose diabetics to pancreatic cancer.
6. References
- Adami HO, McLaughlin J, Ekbom A, Berne C, Silverman D, Hacker D, Persson I. Cancer risk in patients with diabetes mellitus. Cancer Causes Control 2:307-14, 1991. PMID: 1932543
- Arda HE, Benitez CM, Kim SK. Gene regulatory networks governing pancreas development. Dev Cell 25:5-13, 2013. PMID: 23597482
- Balkau B, Barrett-Connor E, Eschwege E, Richard JL, Claude JR, Ducimetiere P. Diabetes and pancreatic carcinoma. Diabete Metab 19:458-62, 1993. PMID: 8056126
- Bao Y, Giovannucci EL, Kraft P, Stampfer MJ, Ogino S, Ma J, Buring JE, Sesso HD, Lee IM, Gaziano JM, Rifai N, Pollak MN, Cochrane BB, Kaklamani V, Lin JH, Manson JE, Fuchs CS, Wolpin BM. A prospective study of plasma adiponectin and pancreatic cancer risk in five US cohorts. J Natl Cancer Institute 105:95-103, 2013. PMID: 23243202
- Barone BB, Yeh HC, Snyder CF, Peairs KS, Stein KB, Derr RL, Wolff AC, Brancati FL. Long-term all-cause mortality in cancer patients with preexisting diabetes mellitus: a systematic review and meta-analysis. JAMA 300:2754-64, 2008. PMID: 19088353
- Batty GD, Shipley MJ, Marmot M, Smith GD. Diabetes status and post-load plasma glucose concentration in relation to site-specific cancer mortality: findings from the original Whitehall study. Cancer Causes Control 15:873-81, 2004. PMID: 15577289
- Ben Q, Xu M, Ning X, Liu J, Hong S, Huang W, Zhang H, Li Z. Diabetes mellitus and risk of pancreatic cancer: A meta-analysis of cohort studies. Eur J Cancer 47:1928-37, 2011. PMID: 21458985
- Benod C, Vinogradova MV, Jouravel N, Kim GE, Fletterick RJ, Sablin EP. Nuclear receptor liver receptor homologue 1 (LRH-1) regulates pancreatic cancer cell growth and proliferation. Proc Natl Acad Sci USA 108:16927-31, 2011. PMID: 21949357
- Bonelli L, Aste H, Bovo P, Cavallini G, Felder M, Gusmaroli R, Morandini E, Ravelli P, Briglia R, Lombardo L, De Micheli A, Pugliese V. Exocrine pancreatic cancer, cigarette smoking, and diabetes mellitus: a case-control study in northern Italy. Pancreas 27:143-9, 2003. PMID: 12883263
- Bosetti C, Rosato V, Li D, Silverman D, Petersen GM, Bracci PM, Neale RE, Muscat J, Anderson K, Gallinger S, Olson SH, Miller AB, Bueno-de-Mesquita HB, Scelo G, Janout V, Holcatova I, Lagiou P, Serraino D, Lucenteforte E, Fabianova E, Ghadirian P, Baghurst PA, Zatonski W, Foretova L, Fontham E, Bamlet WR, Holly EA, Negri E, Hassan M, Prizment A, Cotterchio M, Cleary S, Kurtz RC, Maisonneuve P, Trichopoulos D, Polesel J, Duell EJ, Boffetta P, Vecchia CL. Diabetes, antidiabetic medications and pancreatic cancer risk: an analysis from the international pancreatic cancer case-control consortium. Ann Oncol, 2014. PMID: 25057164
- Bueno de Mesquita HB, Maisonneuve P, Moerman CJ, Walker AM. Aspects of medical history and exocrine carcinoma of the pancreas: a population-based case-control study in The Netherlands. Int J Cancer 52:17-23, 1992. PMID: 1500222
- Butler AE, Galasso R, Matveyenko A, Rizza RA, Dry S, Butler PC. Pancreatic duct replication is increased with obesity and type 2 diabetes in humans. Diabetologia 53:21-6, 2010. PMID: 19844672
- Chow WH, Gridley G, Nyren O, Linet MS, Ekbom A, Fraumeni JF, Jr., Adami HO. Risk of pancreatic cancer following diabetes mellitus: a nationwide cohort study in Sweden. J Natl Cancer Inst 87:930-1, 1995. PMID: 7666483
- Coughlin SS, Calle EE, Teras LR, Petrelli J, Thun MJ. Diabetes mellitus as a predictor of cancer mortality in a large cohort of US adults. Am J Epidemiol 159:1160-7, 2004. PMID: 15191933
- Cui Y, Andersen DK. Diabetes and pancreatic cancer. Endocr Relat Cancer 19:F9-F26, 2012. PMID: 22843556
- Cuzick J, Babiker AG. Pancreatic cancer, alcohol, diabetes mellitus and gall-bladder disease. Int J Cancer 43:415-21, 1989. PMID: 2925272
- Dong X, Li Y, Chang P, Tang H, Hess KR, Abbruzzese JL, Li D. Glucose metabolism gene variants modulate the risk of pancreatic cancer. Cancer Prev Res (Phila) 4:758-66, 2011. PMID: 21411499
- Douglas JB, Silverman DT, Weinstein SJ, Graubard BI, Pollak MN, Tao Y, Virtamo J, Albanes D, Stolzenberg-Solomon RZ. Serum C-reactive protein and risk of pancreatic cancer in two nested, case-control studies. Cancer Epidemiol Biomarkers Prev 20:359-69, 2011. PMID: 21173171
- Duell EJ, Casella DP, Burk RD, Kelsey KT, Holly EA. Inflammation, genetic polymorphisms in proinflammatory genes TNF-A, RANTES, and CCR5, and risk of pancreatic adenocarcinoma. Cancer Epidemiol Biomarkers Prev 15:726-31, 2006. PMID: 16614115
- Ekoe JM, Ghadirian P, Simard A, Baillargeon J, Perret C. [Diabetes mellitus and pancreatic cancer: a case-control study in greater Montreal, Quebec, Canada]. Rev Epidemiol Sante Publique 40:447-53, 1992. PMID: 1287744
- Elena JW, Steplowski E, Yu K, Hartge P, Tobias GS, Brotzman MJ, Chanock SJ, Stolzenberg-Solomon RZ, Arslan AA, Bueno-de-Mesquita HB, Helzlsouer K, Jacobs EJ, LaCroix A, Petersen G, Zheng W, Albanes D, Allen NE, Amundadottir L, Bao Y, Boeing H, Boutron-Ruault MC, Buring JE, Gaziano JM, Giovannucci EL, Duell EJ, Hallmans G, Howard BV, Hunter DJ, Hutchinson A, Jacobs KB, Kooperberg C, Kraft P, Mendelsohn JB, Michaud DS, Palli D, Phillips LS, Overvad K, Patel AV, Sansbury L, Shu XO, Simon MS, Slimani N, Trichopoulos D, Visvanathan K, Virtamo J, Wolpin BM, Zeleniuch-Jacquotte A, Fuchs CS, Hoover RN, Gross M. Diabetes and risk of pancreatic cancer: a pooled analysis from the pancreatic cancer cohort consortium. Cancer Causes Control 24:13-25, 2013. PMID: 23112111
- Everhart J, Wright D. Diabetes mellitus as a risk factor for pancreatic cancer. A meta-analysis. JAMA 273:1605-9, 1995. PMID: 7745774
- Farrow DC, Davis S. Risk of pancreatic cancer in relation to medical history and the use of tobacco, alcohol and coffee. Int J Cancer 45:816-20, 1990. PMID: 2335385
- Fisher WE. Diabetes: risk factor for the development of pancreatic cancer or manifestation of the disease? World J Surg 25:503-8, 2001. PMID: 11396427
- Flandez M, Cendrowski J, Canamero M, Salas A, Del Pozo N, Schoonjans K, Real FX. Nr5a2 heterozygosity sensitises to, and cooperates with, inflammation in KRasG12V-driven pancreatic tumourigenesis. Gut, 2013. PMID: 23598351
- Fong P-y, Fesinmeyer MD, White E, Farin FM, Srinouanprachanh S, Afsharinejad Z, Mandelson MT, Brentnall TA, Barnett MJ, Goodman GE, Austin MA. Association of diabetes susceptibility gene calpain-10 with pancreatic cancer among smokers. J Gastrointestinal Cancer 41:203-8, 2010. PMID: 20178008
- Friedman GD, van den Eeden SK. Risk factors for pancreatic cancer: an exploratory study. Int J Epidemiol 22:30-7, 1993. PMID: 8449644
- Frye JN, Inder WJ, Dobbs BR, Frizelle FA. Pancreatic cancer and diabetes: is there a relationship? A case-controlled study. Aust NZ J Surg 70:722-4, 2000. PMID: 11021485
- Gapstur SM, Gann PH, Lowe W, Liu K, Colangelo L, Dyer A. Abnormal glucose metabolism and pancreatic cancer mortality. Jama 283:2552-2558, 2000. PMID: 10815119
- Giovannucci E, Harlan DM, Archer MC, Bergenstal RM, Gapstur SM, Habel LA, Pollak M, Regensteiner JG, Yee D. Diabetes and cancer: a consensus report. CA Cancer J Clin 2010;60:207-21. PMID: 20554718
- Gold EB, Gordis L, Diener MD, Seltser R, Boitnott JK, Bynum TE, Hutcheon DF. Diet and other risk factors for cancer of the pancreas. Cancer 55:460-7, 1985. PMID: 3965101
- Grote VA, Rohrmann S, Nieters A, Dossus L, Tjonneland A, Halkjaer J, Overvad K, Fagherazzi G, Boutron-Ruault MC, Morois S, Teucher B, Becker S, Sluik D, Boeing H, Trichopoulou A, Lagiou P, Trichopoulos D, Palli D, Pala V, Tumino R, Vineis P, Panico S, Rodriguez L, Duell EJ, Molina-Montes E, Dorronsoro M, Huerta JM, Ardanaz E, Jeurnink SM, Beulens JWJ, Peeters PHM, Sund M, Ye W, Lindkvist B, Johansen D, Khaw KT, Wareham N, Allen N, Crowe F, Jenab M, Romieu I, Michaud DS, Riboli E, Romaguera D, Bueno-de-Mesquita HB, Kaaks R. Diabetes mellitus, glycated haemoglobin and C-peptide levels in relation to pancreatic cancer risk: a study within the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Diabetologia 54:3037-46, 2011. PMID: 21953276
- Grote VA, Kaaks R, Nieters A, Tjonneland A, Halkjaer J, Overvad K, Skjelbo Nielsen MR, Boutron-Ruault MC, Clavel-Chapelon F, Racine A, Teucher B, Becker S, Pischon T, Boeing H, Trichopoulou A, Cassapa C, Stratigakou V, Palli D, Krogh V, Tumino R, Vineis P, Panico S, Rodriguez L, Duell EJ, Sanchez MJ, Dorronsoro M, Navarro C, Gurrea AB, Siersema PD, Peeters PH, Ye W, Sund M, Lindkvist B, Johansen D, Khaw KT, Wareham N, Allen NE, Travis RC, Fedirko V, Jenab M, Michaud DS, Chuang SC, Romaguera D, Bueno-de-Mesquita HB, Rohrmann S. Inflammation marker and risk of pancreatic cancer: a nested case-control study within the EPIC cohort. Br J Cancer 106:1866-74, 2012. PMID: 22617158
- Grote VA, Rohrmann S, Dossus L, Nieters A, Halkjaer J, Tjonneland A, Overvad K, Stegger J, Chabbert-Buffet N, Boutron-Ruault M-C, Clavel-Chapelon F, Teucher B, Becker S, Montonen J, Boeing H, Trichopoulou A, Lagiou P, Trichopoulos D, Palli D, Sieri S, Tumino R, Vineis P, Mattiello A, Arguelles M, Duell EJ, Molina-Montes E, Larranaga N, Chirlaque M-D, Gurrea AB, Jeurnink SM, Peeters PH, Ye W, Sund M, Lindkvist B, Johansen D, Khaw K-T, Wareham N, Crowe FL, Romieu I, Rinaldi S, Jenab M, Romaguera D, Michaud DS, Riboli E, Bas Bueno-de-Mesquita H, Kaaks R. The association of circulating adiponectin levels with pancreatic cancer risk: a study within the prospective EPIC cohort. Int J Cancer 130:2428-37, 2012. PMID: 21681743
- Grote VA, Nieters A, Kaaks R, Tjonneland A, Roswall N, Overvad K, Nielsen MR, Clavel-Chapelon F, Boutron-Ruault MC, Racine A, Teucher B, Lukanova A, Boeing H, Drogan D, Trichopoulou A, Trichopoulos D, Lagiou P, Palli D, Sieri S, Tumino R, Vineis P, Mattiello A, Arguelles Suarez MV, Duell EJ, Sanchez MJ, Dorronsoro M, Huerta Castano JM, Barricarte A, Jeurnink SM, Peeters PH, Sund M, Ye W, Regner S, Lindkvist B, Khaw KT, Wareham N, Allen NE, Crowe FL, Fedirko V, Jenab M, Romaguera D, Siddiq A, Bueno-de-Mesquita HB, Rohrmann S. The associations of advanced glycation end products and its soluble receptor with pancreatic cancer risk: a case-control study within the prospective EPIC Cohort. Cancer Epidemiol Biomarkers Prev 21:619-28, 2012. PMID: 22301828
- Gullo L, Pezzilli R, Morselli-Labate AM. Diabetes and the risk of pancreatic cancer. N Engl J Med 331:81-4, 1994. PMID: 8208269
- Gullo L, Pezzilli R. Diabetes and pancreatic cancer. Pancreas 28:451, 2004; author reply 451-2. PMID: 15097867
- Hassan MM, Bondy ML, Wolff RA, Abbruzzese JL, Vauthey JN, Pisters PW, Evans DB, Khan R, Chou TH, Lenzi R, Jiao L, Li D. Risk factors for pancreatic cancer: case-control study. Am J Gastroenterol 102:2696-707, 2007. PMID: 17764494
- Hiatt RA, Klatsky AL, Armstrong MA. Pancreatic cancer, blood glucose and beverage consumption. Int J Cancer 41:794-7, 1988. PMID: 3372055
- Holmkvist J, Cervin C, Lyssenko V, Winckler W, Anevski D, Cilio C, Almgren P, Berglund G, Nilsson P, Tuomi T, Lindgren CM, Altshuler D, Groop L. Common variants in HNF-1 alpha and risk of type 2 diabetes. Diabetologia 49:2882-91, 2006. PMID: 17033837
- Hoskins JW, Jia J, Flandez M, Parikh H, Xiao W, Collins I, Emmanuel MA, Ibrahim A, Powell J, Zhang L, Malets N, Bamlet WR, Peterson GM, Real FX, Amundadottir LT. Transcriptome analysis of pancreatic cancer reveals a tumor suppressor function for HNF1A. Carcinogenesis, 2014. PMID: 25233928
- Huxley R, Ansary-Moghaddam A, Berrington de Gonzalez A, Barzi F, Woodward M. Type-II diabetes and pancreatic cancer: a meta-analysis of 36 studies. Br J Cancer 92:2076-83, 2005. PMID: 15886696
- Inoue M, Tajima K, Takezaki T, Hamajima N, Hirose K, Ito H, Tominaga S. Epidemiology of pancreatic cancer in Japan: a nested case-control study from the Hospital-based Epidemiologic Research Program at Aichi Cancer Center (HERPACC). Int J Epidemiol 32:257-62, 2003. PMID: 12714546
- Jain M, Howe GR, St Louis P, Miller AB. Coffee and alcohol as determinants of risk of pancreas cancer: a case-control study from Toronto. Int J Cancer 47:384-9, 1991. PMID: 1993545
- Jiao L, Weinstein SJ, Albanes D, Taylor PR, Graubard BI, Virtamo J, Stolzenberg-Solomon RZ. Evidence that serum levels of the soluble receptor for advanced glycation end products are inversely associated with pancreatic cancer risk: a prospective study. Cancer Res 71:3582-9, 2011. PMID: 21540233
- Kalapothaki V, Tzonou A, Hsieh CC, Toupadaki N, Karakatsani A, Trichopoulos D. Tobacco, ethanol, coffee, pancreatitis, diabetes mellitus, and cholelithiasis as risk factors for pancreatic carcinoma. Cancer Causes Control 4:375-82, 1993. PMID: 8347787
- Kessler, II. Cancer mortality among diabetics. J Natl Cancer Inst 44:673-86, 1970. PMID: 11515436
- La Vecchia C, Negri E, Franceschi S, D'Avanzo B, Boyle P. A case-control study of diabetes mellitus and cancer risk. Br J Cancer 70:950-3, 1994. PMID: 7947103
- Lago R, Gomez R, Lago F, Gomez-Reino J, Gualillo O. Leptin beyond body weight regulation--current concepts concerning its role in immune function and inflammation. Cell Immunol 252:139-45, 2008. PMID: 18289518
- Lee CT, Chang FY, Lee SD. Risk factors for pancreatic cancer in orientals. J Gastroenterol Hepatol 11:491-5, 1996. PMID: 8743923
- Lee JM, Lee YK, Mamrosh JL, Busby SA, Griffin PR, Pathak MC, Ortlund EA, Moore DD. A nuclear-receptor-dependent phosphatidylcholine pathway with antidiabetic effects. Nature 474:506-10, 2011. PMID: 21614002
- Li D, Duell EJ, Yu K, Risch HA, Olson SH, Kooperberg C, Wolpin BM, Jiao L, Dong X, Wheeler B, Arslan AA, Bueno-de-Mesquita HB, Fuchs CS, Gallinger S, Gross M, Hartge P, Hoover RN, Holly EA, Jacobs EJ, Klein AP, Lacroix A, Mandelson MT, Petersen G, Zheng W, Agalliu I, Albanes D, Boutron-Ruault MC, Bracci PM, Buring JE, Canzian F, Chang K, Chanock SJ, Cotterchio M, Gaziano JM, Giovannucci EL, Goggins M, Hallmans G, Hankinson SE, Hoffman Bolton JA, Hunter DJ, Hutchinson A, Jacobs KB, Jenab M, Khaw KT, Kraft P, Krogh V, Kurtz RC, McWilliams RR, Mendelsohn JB, Patel AV, Rabe KG, Riboli E, Shu XO, Tjonneland A, Tobias GS, Trichopoulos D, Virtamo J, Visvanathan K, Watters J, Yu H, Zeleniuch-Jacquotte A, Amundadottir L, Stolzenberg-Solomon RZ. Pathway analysis of genome-wide association study data highlights pancreatic development genes as susceptibility factors for pancreatic cancer. Carcinogenesis 33:1384-90, 2012. PMID: 22523087
- Li D, Tang H, Hassan MM, Holly EA, Bracci PM, Silverman DT. Diabetes and risk of pancreatic cancer: a pooled analysis of three large case-control studies. Cancer Causes Control 22:189-97, 2011. PMID: 21104117
- Li D. Diabetes and pancreatic cancer. Mol Carcinog 51:64-74, 2012. PMID: 22162232
- Lin YS, Tamakoshi A, Kikuchi S, Yagyu K, Obata Y, Ishibashi T, Kawamura T, Inaba Y, Kurosawa M, Motohashi Y, Ohno Y, Group JS. Serum insulin-like growth factor-I, insulin-like growth factor binding protein-3, and the risk of pancreatic cancer death. Int J Cancer 110:584-588, 2004. PMID: 15122592
- Lin RS, Kessler, II. A multifactorial model for pancreatic cancer in man. Epidemiologic evidence. JAMA 245:147-52, 1981. PMID: 7452829
- Lin Y, Tamakoshi A, Kawamura T, Inaba Y, Kikuchi S, Motohashi Y, Kurosawa M, Ohno Y. Risk of pancreatic cancer in relation to alcohol drinking, coffee consumption and medical history: findings from the Japan collaborative cohort study for evaluation of cancer risk. Int J Cancer 99:742-6, 2002. PMID: 12115510
- Lund Nilsen TI, Johnsen R, Vatten LJ. Socio-economic and lifestyle factors associated with the risk of prostate cancer. Br J Cancer 82:1358-63, 2000. PMID: 10755415
- MacMahon B, Yen S, Trichopoulos D, Warren K, Nardi G. Coffee and cancer of the pancreas. N Engl J Med 304:630-3, 1981. PMID: 7453739
- Manning AK, Hivert MF, Scott RA, Grimsby JL, Bouatia-Naji N, Chen H, Rybin D, Liu CT, Bielak LF, Prokopenko I, Amin N, Barnes D, Cadby G, Hottenga JJ, Ingelsson E, Jackson AU, Johnson T, Kanoni S, Ladenvall C, Lagou V, Lahti J, Lecoeur C, Liu Y, Martinez-Larrad MT, Montasser ME, Navarro P, Perry JR, Rasmussen-Torvik LJ, Salo P, Sattar N, Shungin D, Strawbridge RJ, Tanaka T, van Duijn CM, An P, de Andrade M, Andrews JS, Aspelund T, Atalay M, Aulchenko Y, Balkau B, Bandinelli S, Beckmann JS, Beilby JP, Bellis C, Bergman RN, Blangero J, Boban M, Boehnke M, Boerwinkle E, Bonnycastle LL, Boomsma DI, Borecki IB, Bottcher Y, Bouchard C, Brunner E, Budimir D, Campbell H, Carlson O, Chines PS, Clarke R, Collins FS, Corbaton-Anchuelo A, Couper D, de Faire U, Dedoussis GV, Deloukas P, Dimitriou M, Egan JM, Eiriksdottir G, Erdos MR, Eriksson JG, Eury E, Ferrucci L, Ford I, Forouhi NG, Fox CS, Franzosi MG, Franks PW, Frayling TM, Froguel P, Galan P, de Geus E, Gigante B, Glazer NL, Goel A, Groop L, Gudnason V, Hallmans G, Hamsten A, Hansson O, Harris TB, Hayward C, Heath S, Hercberg S, Hicks AA, Hingorani A, Hofman A, Hui J, Hung J, et al. A genome-wide approach accounting for body mass index identifies genetic variants influencing fasting glycemic traits and insulin resistance. Nat Genet 44:659-69, 2012. PMID: 22581228
- Meier JJ, Nauck MA. Incretins and the development of type 2 diabetes. Curr Diab Rep 6:194-201, 2006. PMID: 16898571
- Michaud DS, Wolpin B, Giovannucci E, Liu S, Cochrane B, Manson JE, Pollak MN, Ma J, Fuchs CS. Prediagnostic plasma C-peptide and pancreatic cancer risk in men and women. Cancer Epidemiol Biomarkers Prev 16:2101-9, 2007. PMID: 17905943
- Mills PK, Beeson WL, Abbey DE, Fraser GE, Phillips RL. Dietary habits and past medical history as related to fatal pancreas cancer risk among Adventists. Cancer 61:2578-85, 1988. PMID: 3365678
- Morris JPt, Wang SC, Hebrok M. KRAS, Hedgehog, Wnt and the twisted developmental biology of pancreatic ductal adenocarcinoma. Nat Rev Cancer 10:683-95, 2010. PMID: 20814421
- Norell S, Ahlbom A, Erwald R, Jacobson G, Lindberg-Navier I, Olin R, Wiechel KL. Diabetes, gall stone disease, and pancreatic cancer. Br J Cancer 54:377-8, 1986. PMID: 3741772
- Noy A, Bilezikian JP. Clinical review 63: Diabetes and pancreatic cancer: clues to the early diagnosis of pancreatic malignancy. J Clin Endocrinol Metab 79:1223-31, 1994. PMID: 7962312
- O'Mara BA, Byers T, Schoenfeld E. Diabetes mellitus and cancer risk: a multisite case-control study. J Chronic Dis 38:435-41, 1985. PMID: 3998058
- Petersen GM, Amundadottir L, Fuchs CS, Kraft P, Stolzenberg-Solomon RZ, Jacobs KB, Arslan AA, Bueno-de-Mesquita HB, Gallinger S, Gross M, Helzlsouer K, Holly EA, Jacobs EJ, Klein AP, LaCroix A, Li D, Mandelson MT, Olson SH, Risch HA, Zheng W, Albanes D, Bamlet WR, Berg CD, Boutron-Ruault MC, Buring JE, Bracci PM, Canzian F, Clipp S, Cotterchio M, de Andrade M, Duell EJ, Gaziano JM, Giovannucci EL, Goggins M, Hallmans G, Hankinson SE, Hassan M, Howard B, Hunter DJ, Hutchinson A, Jenab M, Kaaks R, Kooperberg C, Krogh V, Kurtz RC, Lynch SM, McWilliams RR, Mendelsohn JB, Michaud DS, Parikh H, Patel AV, Peeters PH, Rajkovic A, Riboli E, Rodriguez L, Seminara D, Shu XO, Thomas G, Tjonneland A, Tobias GS, Trichopoulos D, Van Den Eeden SK, Virtamo J, Wactawski-Wende J, Wang Z, Wolpin BM, Yu H, Yu K, Zeleniuch-Jacquotte A, Fraumeni JF, Jr., Hoover RN, Hartge P, Chanock SJ. A genome-wide association study identifies pancreatic cancer susceptibility loci on chromosomes 13q22.1, 1q32.1 and 5p15.33. Nat Genet 42:224-8, 2010. PMID: 20101243
- Pierce BL, Ahsan H. Genome-wide "pleiotropy scan" identifies HNF1A region as a novel pancreatic cancer susceptibility locus. Cancer Res 71:4352-8, 2011. PMID: 21498636
- Pierce BL, Austin MA, Ahsan H. Association study of type 2 diabetes genetic susceptibility variants and risk of pancreatic cancer: an analysis of PanScan-I data. Cancer Causes Control 22:877-83, 2011. PMID: 21445555
- Pollak M. Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer 8:915-28, 2008. PMID: 19029956
- Prizment AE, Gross M, Rasmussen-Torvik L, Peacock JM, Anderson KE. Genes related to diabetes may be associated with pancreatic cancer in a population-based case-control study in Minnesota. Pancreas 41:50-3, 2012. PMID: 22015968
- Ragozzino M, Melton LJ, 3rd, Chu CP, Palumbo PJ. Subsequent cancer risk in the incidence cohort of Rochester, Minnesota, residents with diabetes mellitus. J Chronic Dis 35:13-9, 1982. PMID: 7068798
- Reid-Lombardo KM, Fridley BL, Bamlet WR, Cunningham JM, Sarr MG, Petersen GM. Inflammation-related gene variants as risk factors for pancreatic cancer. Cancer Epidemiol Biomarkers Prev 20:1251-4, 2011. PMID: 21467233
- Rohrmann S, Grote VA, Becker S, Rinaldi S, Tjonneland A, Roswall N, Gronbaek H, Overvad K, Boutron-Ruault MC, Clavel-Chapelon F, Racine A, Teucher B, Boeing H, Drogan D, Dilis V, Lagiou P, Trichopoulou A, Palli D, Tagliabue G, Tumino R, Vineis P, Mattiello A, Rodriguez L, Duell EJ, Molina-Montes E, Dorronsoro M, Huerta JM, Ardanaz E, Jeurnink S, Peeters PH, Lindkvist B, Johansen D, Sund M, Ye W, Khaw KT, Wareham NJ, Allen NE, Crowe FL, Fedirko V, Jenab M, Michaud DS, Norat T, Riboli E, Bueno-de-Mesquita HB, Kaaks R. Concentrations of IGF-I and IGFBP-3 and pancreatic cancer risk in the European Prospective Investigation into Cancer and Nutrition. Br J Cancer 106:1004-10, 2012. PMID: 22315049
- Rulyak SJ, Lowenfels AB, Maisonneuve P, Brentnall TA. Risk factors for the development of pancreatic cancer in familial pancreatic cancer kindreds. Gastroenterology 124:1292-9, 2003. PMID: 12730869
- Shibata A, Mack TM, Paganini-Hill A, Ross RK, Henderson BE. A prospective study of pancreatic cancer in the elderly. Int J Cancer 58:46-9, 1994. PMID: 8014014
- Silverman DT, Schiffman M, Everhart J, Goldstein A, Lillemoe KD, Swanson GM, Schwartz AG, Brown LM, Greenberg RS, Schoenberg JB, Pottern LM, Hoover RN, Fraumeni JF, Jr. Diabetes mellitus, other medical conditions and familial history of cancer as risk factors for pancreatic cancer. Br J Cancer 80:1830-7, 1999. PMID: 10468306
- Smith GD, Egger M, Shipley MJ, Marmot MG. Post-challenge glucose concentration, impaired glucose tolerance, diabetes, and cancer mortality in men. Am J Epidemiol 136:1110-4, 1992. PMID: 1462971
- Smith TJ. Insulin-like growth factor-I regulation of immune function: a potential therapeutic target in autoimmune diseases? Pharmacol Rev 62:199-236, 2010. PMID: 20392809
- Speliotes EK, Willer CJ, Berndt SI, Monda KL, Thorleifsson G, Jackson AU, Lango Allen H, Lindgren CM, Luan J, Magi R, Randall JC, Vedantam S, Winkler TW, Qi L, Workalemahu T, Heid IM, Steinthorsdottir V, Stringham HM, Weedon MN, Wheeler E, Wood AR, Ferreira T, Weyant RJ, Segre AV, Estrada K, Liang L, Nemesh J, Park JH, Gustafsson S, Kilpelainen TO, Yang J, Bouatia-Naji N, Esko T, Feitosa MF, Kutalik Z, Mangino M, Raychaudhuri S, Scherag A, Smith AV, Welch R, Zhao JH, Aben KK, Absher DM, Amin N, Dixon AL, Fisher E, Glazer NL, Goddard ME, Heard-Costa NL, Hoesel V, Hottenga JJ, Johansson A, Johnson T, Ketkar S, Lamina C, Li S, Moffatt MF, Myers RH, Narisu N, Perry JR, Peters MJ, Preuss M, Ripatti S, Rivadeneira F, Sandholt C, Scott LJ, Timpson NJ, Tyrer JP, van Wingerden S, Watanabe RM, White CC, Wiklund F, Barlassina C, Chasman DI, Cooper MN, Jansson JO, Lawrence RW, Pellikka N, Prokopenko I, Shi J, Thiering E, Alavere H, Alibrandi MT, Almgren P, Arnold AM, Aspelund T, Atwood LD, Balkau B, Balmforth AJ, Bennett AJ, Ben-Shlomo Y, Bergman RN, Bergmann S, Biebermann H, Blakemore AI, Boes T, Bonnycastle LL, Bornstein SR, Brown MJ, Buchanan TA, et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet 42:937-48, 2010. PMID: 20935630
- Stevens RJ, Roddam AW, Beral V. Pancreatic cancer in type 1 and young-onset diabetes: systematic review and meta-analysis. Br J Cancer 96:507-9, 2007. PMID: 17224924
- Stolzenberg-Solomon RZ, Limburg P, Pollak M, Taylor PR, Virtamo J, Albanes D. Insulin-like growth factor (IGF)-1, IGF-binding protein-3, and pancreatic cancer in male smokers. Cancer Epidemiol Biomarkers Prev 13:438-4, 2004. PMID: 15006921
- Stolzenberg-Solomon RZ, Weinstein S, Pollak M, Tao Y, Taylor PR, Virtamo J, Albanes D. Prediagnostic adiponectin concentrations and pancreatic cancer risk in male smokers. Am J Epidemiol 168:1047-55, 2008. PMID: 18801887
- Stolzenberg-Solomon RZ, Graubard BI, Chari S, Limburg P, Taylor PR, Virtamo J, Albanes D. Insulin, glucose, insulin resistance, and pancreatic cancer in male smokers. JAMA 294:2872-8, 2005. PMID: 16352795
- Stolzenberg-Solomon RZ, Pietinen P, Taylor PR, Virtamo J, Albanes D. A prospective study of medical conditions, anthropometry, physical activity, and pancreatic cancer in male smokers (Finland). Cancer Causes Control 13:417-26, 2002. PMID: 12146846
- Suzuki H, Li Y, Dong X, Hassan MM, Abbruzzese JL, Li D. Effect of insulin-like growth factor gene polymorphisms alone or in interaction with diabetes on the risk of pancreatic cancer. Cancer Epidemiol Biomarkers Prev 17:3467-73, 2008. PMID: 19064563
- Tang H, Dong X, Hassan M, Abbruzzese JL, Li D. Body mass index and obesity- and diabetes-associated genotypes and risk for pancreatic cancer. Cancer Epidemiol Biomarkers Prev 20:779-92, 2011. PMID: 21357378
- Tang H, Wei P, Duell EJ, Risch HA, Olson SH, Bueno-de-Mesquita HB, Gallinger S, Holly EA, Petersen GM, Bracci PM, McWilliams RR, Jenab M, Riboli E, Tjonneland A, Boutron-Ruault MC, Kaaks R, Trichopoulos D, Panico S, Sund M, Peeters PH, Khaw KT, Amos CI, Li D. Genes-environment interactions in obesity- and diabetes-associated pancreatic cancer: a GWAS data analysis. Cancer Epidemiol Biomarkers Prev 23:98-106, 2014. PMID: 24136929
- Tuomi T, Santoro N, Caprio S, Cai M, Weng J, Groop L. The many faces of diabetes: a disease with increasing heterogeneity. Lancet 383:1084-94, 2014.
- Voight BF, Scott LJ, Steinthorsdottir V, Morris AP, Dina C, Welch RP, Zeggini E, Huth C, Aulchenko YS, Thorleifsson G, McCulloch LJ, Ferreira T, Grallert H, Amin N, Wu G, Willer CJ, Raychaudhuri S, McCarroll SA, Langenberg C, Hofmann OM, Dupuis J, Qi L, Segre AV, van Hoek M, Navarro P, Ardlie K, Balkau B, Benediktsson R, Bennett AJ, Blagieva R, Boerwinkle E, Bonnycastle LL, Bengtsson Bostrom K, Bravenboer B, Bumpstead S, Burtt NP, Charpentier G, Chines PS, Cornelis M, Couper DJ, Crawford G, Doney AS, Elliott KS, Elliott AL, Erdos MR, Fox CS, Franklin CS, Ganser M, Gieger C, Grarup N, Green T, Griffin S, Groves CJ, Guiducci C, Hadjadj S, Hassanali N, Herder C, Isomaa B, Jackson AU, Johnson PR, Jorgensen T, Kao WH, Klopp N, Kong A, Kraft P, Kuusisto J, Lauritzen T, Li M, Lieverse A, Lindgren CM, Lyssenko V, Marre M, Meitinger T, Midthjell K, Morken MA, Narisu N, Nilsson P, Owen KR, Payne F, Perry JR, Petersen AK, Platou C, Proenca C, Prokopenko I, Rathmann W, Rayner NW, Robertson NR, Rocheleau G, Roden M, Sampson MJ, Saxena R, Shields BM, Shrader P, Sigurdsson G, Sparso T, Strassburger K, Stringham HM, Sun Q, Swift AJ, Thorand B, et al. Twelve type 2 diabetes susceptibility loci identified through large-scale association analysis. Nat Genet 42:579-89, 2010. PMID: 20581827
- von Figura G, Morris JPt, Wright CV, Hebrok M. Nr5a2 maintains acinar cell differentiation and constrains oncogenic Kras-mediated pancreatic neoplastic initiation. Gut, 2013. PMID: 23645620
- Wang F, Gupta S, Holly EA. Diabetes mellitus and pancreatic cancer in a population-based case-control study in the San Francisco Bay Area, California. Cancer Epidemiol Biomarkers Prev 15:1458-63, 2006. PMID: 16896032
- Whittemore AS, Paffenbarger RS, Jr., Anderson K, Halpern J. Early precursors of pancreatic cancer in college men. J Chronic Dis 36:251-6, 1983. PMID: 6826689
- Wideroff L, Gridley G, Mellemkjaer L, Chow WH, Linet M, Keehn S, Borch-Johnsen K, Olsen JH. Cancer incidence in a population-based cohort of patients hospitalized with diabetes mellitus in Denmark. J Natl Cancer Inst 89:1360-5, 1997. PMID: 9308706
- Wohlfert EA, Nichols FC, Nevius E, Clark RB. Peroxisome proliferator-activated receptor gamma (PPARgamma) and immunoregulation: enhancement of regulatory T cells through PPARgamma-dependent and -independent mechanisms. J Immunol 178:4129-35, 2007. PMID: 17371968
- Wolpin BM, Bao Y, Qian ZR, Wu C, Kraft P, Ogino S, Stampfer MJ, Sato K, Ma J, Buring JE, Sesso HD, Lee IM, Gaziano JM, McTiernan A, Phillips LS, Cochrane BB, Pollak MN, Manson JE, Giovannucci EL, Fuchs CS. Hyperglycemia, insulin resistance, impaired pancreatic beta-cell function, and risk of pancreatic cancer. J Natl Cancer Inst 105:1027-35, 2013. PMID: 23847240
- Wolpin BM, Rizzato C, Kraft P, Kooperberg C, Petersen GM, Wang Z, Arslan AA, Beane-Freeman L, Bracci PM, Buring J, Canzian F, Duell EJ, Gallinger S, Giles GG, Goodman GE, Goodman PJ, Jacobs EJ, Kamineni A, Klein AP, Kolonel LN, Kulke MH, Li D, Malats N, Olson SH, Risch HA, Sesso HD, Visvanathan K, White E, Zheng W, Abnet CC, Albanes D, Andreotti G, Austin MA, Barfield R, Basso D, Berndt SI, Boutron-Ruault MC, Brotzman M, Buchler MW, Bueno-de-Mesquita HB, Bugert P, Burdette L, Campa D, Caporaso NE, Capurso G, Chung C, Cotterchio M, Costello E, Elena J, Funel N, Gaziano JM, Giese NA, Giovannucci EL, Goggins M, Gorman MJ, Gross M, Haiman CA, Hassan M, Helzlsouer KJ, Henderson BE, Holly EA, Hu N, Hunter DJ, Innocenti F, Jenab M, Kaaks R, Key TJ, Khaw KT, Klein EA, Kogevinas M, Krogh V, Kupcinskas J, Kurtz RC, LaCroix A, Landi MT, Landi S, Le Marchand L, Mambrini A, Mannisto S, Milne RL, Nakamura Y, Oberg AL, Owzar K, Patel AV, Peeters PH, Peters U, Pezzilli R, Piepoli A, Porta M, Real FX, Riboli E, Rothman N, Scarpa A, Shu XO, Silverman DT, Soucek P, Sund M, Talar-Wojnarowska R, Taylor PR, Theodoropoulos GE, et al. Genome-wide association study identifies multiple susceptibility loci for pancreatic cancer. Nat Genet, 2014. PMID: 25086665
- Wolpin BM, Michaud DS, Giovannucci EL, Schernhammer ES, Stampfer MJ, Manson JE, Cochrane BB, Rohan TE, Ma J, Pollak MN, Fuchs CS. Circulating insulin-like growth factor binding protein-1 and the risk of pancreatic cancer. Cancer Res 67:7923-8, 2007. PMID: 17699799
- Wolpin BM, Michaud DS, Giovannucci EL, Schernhammer ES, Stampfer MJ, Manson JE, Cochrane BB, Rohan TE, Ma J, Pollak MN, Fuchs CS. Circulating insulin-like growth factor axis and the risk of pancreatic cancer in four prospective cohorts. Br J Cancer 97:98-104, 2007. PMID: 17533398
- Wynder EL, Mabuchi K, Maruchi N, Fortner JG. A case control study of cancer of the pancreas. Cancer 31:641-8, 1973. PMID: 4693593


