Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2020.09
Abstract:
Zymogen granules (ZG) are located at the apex of pancreatic acinar cells. Secretagogues bind to receptors at the basolateral membrane of the cells and trigger the activation of intracellular signaling pathways, which elicit the fusion of ZG with the apical plasma membrane and exocytosis of digestive (pro-)enzymes (the “zymogens”) into the lumen. The regulated discharge of stored macromolecules is accompanied by secretion of solutes and water to the cell exterior to fluidize these protein-rich secretory products. Previous functional and pharmacological studies in pancreatic acinar cells and isolated ZG had indicated that ion channels and transporters are expressed in the membrane of ZG where they may contribute to maturation, fusion, exocytosis and/or fluidization of zymogens. This review provides a current update of studies that have been mainly published in the postgenomic era and identified cloned transporters and ion channels in the membrane of ZG. Available experimental evidence indicates the presence of several ion channel and transporter proteins in ZG membranes (aquaporins, vacuolar-type H+-ATPase, K+ channels Kv7.1 and Kir6.1, zinc influx transporter SLC30A2, P2X4 receptor). Evidence for ClC Cl- channels and for the vesicular nucleotide transporter SLC17A9 in ZG is less strong. For a better understanding of the function of these proteins in the secretory pathway, further studies are needed.
I. INTRODUCTION
An update of this review on pancreatic zymogen granule (ZG) channels and transporters and their function is timely since its first publication in 2015 (137). The advent of the post-genomic era has led – similar to other areas of cell biology – to the discovery of a number of ZG transport proteins and the characterization of their function in pancreatic acinar secretion. Recent studies have combined functional and molecular approaches to identify ZG channels and transporters and their contribution to pancreas physiology and pathology. A better understanding of the physiology of pancreatic acinar cell secretion and the role played by ZG in this process is prerequisite to comprehend the pathogenesis of major pancreatic disorders, such as pancreatitis, cystic fibrosis or cancer (86). Yet the fact that only a very limited number of studies have been published in this area of research is surprising, despite tremendous progress of knowledge and methodologies to investigate the molecular and cellular biology and physiology of the pancreas (155).
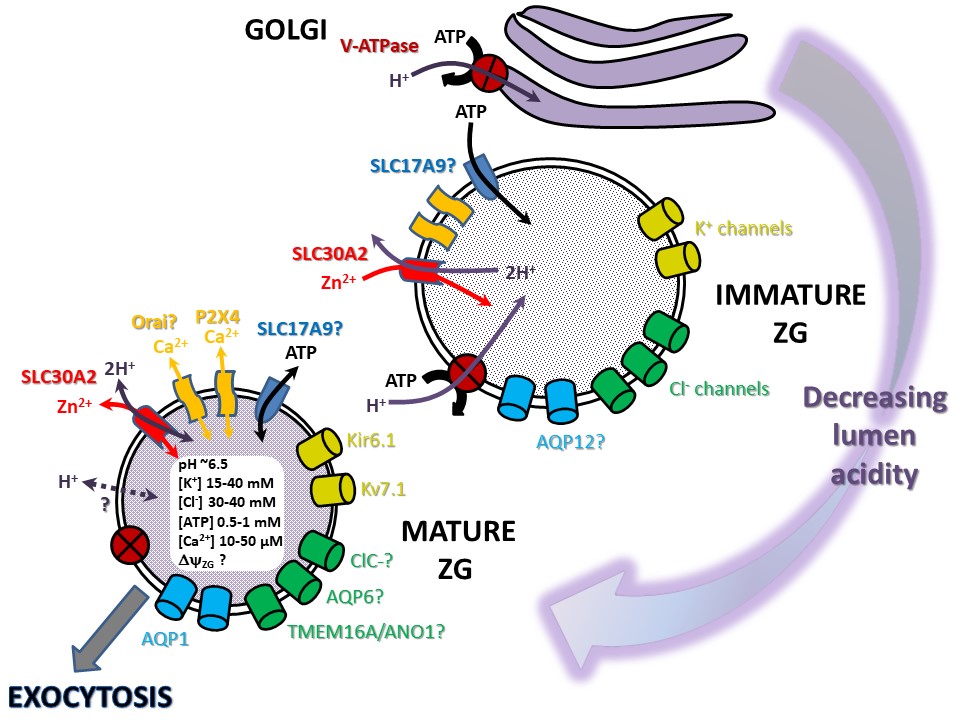
In the following paragraphs, I review significant advances in the characterization of ZG channels and transporters in the last two decades and discuss their putative role in pancreatic acinar secretion (Figure 1). The review ends with an outlook on unsolved issues that require clarification to move the field forward.
Figure 1. The model summarizes current evidence (strong or weak) for the presence of ion channels and transporters in zymogen granules (ZG) of pancreatic acinar cells. The Figure also describes relevant ion concentrations in mature ZG and emphasizes the decreasing lumen acidity during maturation along the compartments of the secretory pathway that participate in ZG maturation (for further details, see text).
II. PROTEOMICS
In the past, ion pathways in ZG were either characterized in permeabilized acini in which the effect of the ionic composition of the “extended cytosol” on stimulated secretion was investigated, and/or by recording macroscopic ion fluxes using an osmotic swelling assay (and lysis as an end-point) in suspensions of isolated ZG, and has been previously exhaustively reviewed (138). That piece of work has provided important 'background' knowledge for the interpretation of the post-genomic work (see below).
A key advance in the last decade has been the widespread availability of proteomics that allow to simultaneously identify large numbers of expressed proteins. The use of proteomics for analysis of ZG and their membranes has propelled the identification of channels and transporters of pancreatic ZG although various caveats must be contemplated. Several proteomic studies have been published that used highly purified ZG membranes (ZGM) to identify cloned transporters and channel proteins (10, 11, 22-26, 124, 135). In most of these studies, granules were disrupted by different techniques, and membranes were subjected to carbonate and/or bromide extraction, which is a standardized and reliable procedure to obtain pure membranes without peripheral proteins (16). A critical step in this isolation procedure represents the initial “purification” of ZG by either differential or continuous Percoll gradient centrifugation. These methods yield fractions that are enriched 4-8-times in α-amylase compared to homogenate, which is close to the theoretical limit of purity (98). Yet, it is practically impossible to avoid contaminations by lysosomes (124), plasma membranes (141) or membranes of other disrupted organelles (124, 135); see also electron micrographs in references (16, 141, 163). These contaminants are, however, a major drawback for proteomic analyses because even minimal contaminations by membranes originating from other organelles or plasma membranes may be prominent in a proteomic analysis of ZGM because of proteins that are highly expressed in these contaminating membranes. This issue needs to be considered, particularly for “mitochondrial” proteins such as voltage-dependent anion channels (VDACs) and subunits of ATP-synthase that have been detected in ZGM in several independent studies (10, 22, 23, 26), or for “plasma membrane” proteins, such as the α-subunit of Na+/K+-ATPase (124). Hence, these particular observations need to be taken with caution. In addition, detection of a particular channel or transporter in ZGM may not infer any physiological/functional significance of that identified protein in mature ZG (see the paragraph on Vacuolar-type H+-ATPase). Finally, it should be born in mind that several cloned channel proteins and transporters that have been identified by functional or immunological approaches have not been detected by proteomic analyses. Some studies even claim that no transmembrane proteins or classical membrane-anchored proteins are expressed in ZGM (11). But this conclusion may be due to the relative insensitivity of the proteomic approaches used because of low expression levels of transport proteins in ZGM.
Interestingly, among a variety of putative novel transporters and channels identified in ZGM by Rindler and coworkers (124), the ATP-gated receptor P2X4 deserves special attention. Indeed, P2X4 has been recently identified in lysosomes and lamellar bodies where it operates as a nonselective cation channel that is inhibited by luminal acidity, while increasing luminal pH in the presence of ATP elicits channel activation (39, 64). These functional properties of a P2X4 ATP receptor located in ZGM - if not due to lysosomal contamination - are attractive for ZG exocytosis, considering the fact that maturing ZG lose their acidity (see the paragraph on Vacuolar-type H+-ATPase). Moreover, the P2X4 cation channel is activated by ATP concentrations that have been measured in ZG (see the paragraph on Vesicular nucleotide transporter SLC17A9 for a discussion of this concept). Nonetheless, the detection of the P2X4 ATP receptor offers an example of a potentially novel aspect of ZG physiology that is derived from proteomic approaches. Hence, P2X4 is a promising candidate for future studies on ZG exocytosis.
III. CHANNELS
A. H2O Channels
Digestive macromolecules are packaged and condensed together with various osmotically active ions and small organic molecules in the lumen of ZG during maturation, where granular Cl- and K+ concentrations display lower concentrations than in the cytosol (106, 125). It is a long-standing observation that isolated ZG and other secretory granules suspended in isotonic sugar or salt solutions, such as KCl, remain stable for hours (8, 33). Consequently, the stability of secretory granules in isotonic solutions indicates that the intragranular osmolarity does not exceed cytosolic osmolarity and/or that granules exhibit a low basal permeability for H2O, K+ and Cl- in situ and in vitro. Reduction of extragranular osmolarity below 245 mOsmol/l (8), however, or addition of cation or anion ionophores to isolated ZG suspended in iso-osmotic KCl buffers (33) elicits granular swelling and lysis. In the latter case, ionophores generate an electrochemical membrane potential that activates endogenous conductive pathways for counterions, thus allowing entry of osmotically active KCl into the granular lumen, which attract H2O and induce swelling (and lysis) of secretory granules (33, 139). Thus, these observations imply that H2O permeable pathways, e.g. aquaporins, are present in ZGM.
The expression and function of aquaporins in pancreas has been recently reviewed (36, 99). Aquaporin 1 (AQP1) has been identified in rat pancreatic ZGM by a full range of immunodetection methods, namely immunoblotting, immunofluorescence confocal microscopy and immunogold labeling (28). Furthermore, swelling of ZG suspended in hypo-osmotic KCl solution was detected by atomic force microscopy (AFM) when 40 µM GTP was added and blocked by Hg2+, an inhibitor of AQP1, as well as by a functional AQP1 antibody that was raised against the carboxyl-terminal domain of AQP1. These changes of ZG swelling were paralleled by corresponding effects on fluxes of 3H2O (28). The effect of GTP on AQP1 activation is assumed to be mediated by interaction of GTP with a complex of several proteins that were co-immunoprecipitated with an antibody against AQP1 and identified by immunoblotting. They included the GTP-binding protein subunit Gαi3, the inwardly rectifying K+ channel IRK8 (Kir6.1/KCNJ8), the voltage-gated Cl- channel ClC-2 and phospholipase A2 (PLA2) (2). Furthermore, exposure of ZG to either the K+ channel blocker glyburide, or the PLA2 inhibitor ONO-RS-082, blocked GTP-induced ZG swelling measured by AFM, whereas the non-specific Cl- channel inhibitor 4,4-diisothiocyanatostilbene-2,2-disulfonate (DIDS) had no effect.
Interestingly, proteomic analyses have identified AQP8, but not other AQPs, in rat ZGM (124), thus confirming previous immunohistochemical studies in rat pancreas tissues (136). AQP8 may be a genuine ZG membrane protein, however a contamination by mitochondria cannot be ruled out, which express AQP8 as well (18, 82).
An intriguing observation has been made by Ohta et al. (110) who found AQP12 expressed in the rough endoplasmic reticulum (rER) of rat pancreatic acinar cells and extends a previous study demonstrating expression of AQP12 in rat pancreatic acinar cells by in situ hybridization and reverse transcription-polymerase chain reaction (RT-PCR) (68). In rats treated with the secretagogue cholecystokinin octapeptide (CCK-8), Otah et al. (110) detected AQP12 in the rER and also on the membranes of ZG near the rER. Furthermore, AQP12 knockout mice were more prone to caerulein-induced pancreatitis (110). The authors suggested that AQP12 may be involved in the mechanisms underlying the proper generation, maturation, and trafficking of ZG in the secretory pathway and that adequate H2O flux may be necessary for this process to function adequately.
Altogether, it seems highly likely that AQP H2O channels are expressed in pancreatic ZGM because ZG swell and lyse in hypotonic solutions, although the AQP isoform(s) involved remain(s) uncertain. Moreover, the physiological role of AQPs in the complex process of secretory granule maturation, trafficking and exocytosis required for secretagogue-induced pancreatic acinar enzyme and fluid secretion are poorly understood.
B. K+ Channels
In pancreatic acinar cells, ZG are about 4-8-fold lower in K+ compared to the cytosol, as measured by X-ray microanalysis (106) or using a potassium-sensitive fluorescence dye (156). Hence, any K+ conductive pathway should allow flux of K+ into the ZG lumen and increase its osmotic load. Based on the work by Hopfer and coworkers (41), I proposed in 1992 that (ATP- and glibenclamide-sensitive) K+ channels are expressed in ZGM where they contribute to osmotic swelling of isolated pancreatic ZG (139). This observation was soon confirmed by others (42). Subsequent studies have suggested that glibenclamide may modulate ZG K+ permeability indirectly, possibly by binding to a 65-kDa multidrug resistance P-glycoprotein (ABCB1)-like regulatory protein (13), and/or the ZG membrane-associated protein ZG-16p (14). Additional support for the presence of K+ channels in secretory granules came from studies with mucin granules from goblet cells and suggested a Ca2+/K+ ion-exchange mechanism via parallel operation of a Ca2+ channel and a Ca2+-activated K+ channel (104). Ca2+/K+ ion-exchange has been subsequently proposed to play a critical role in the premature activation of trypsin in ZG and development of acute pancreatitis (156).
Jena and coworkers (2, 78) have identified an inwardly rectifying K+ channel IRK8 (Kir6.1/KCNJ8) in ZGM by immunoblotting and measured whole vesicle currents of patched single ZG with KCl as permeating ions that were reduced by ~15% by either 40 µM quinidine or 20 µM glibenclamide. Proteomic analyses of purified rat ZGM have detected the K+ channel TWIK-2/KCNK6 (124), a member of a two-pore-domain K+ channel family that produces constitutive inward rectifying K+ currents of weak amplitude and that is highly expressed in the pancreas (88, 97). However, their functional properties and their wide distribution suggest that these channels are mainly involved in the control of background K+ conductive pathways in the plasma membranes of many cell types. Hence, it cannot be excluded that plasma membranes may have contaminated ZGM in the proteomic study reporting TWIK-2/KCNK6 expression in ZGM (124) (see also the paragraph on Proteomics).
Kv7.1 (KvLQT1/KCNQ1) is a very low-conductance, voltage-gated six-membrane-spanning K+ channel distributed widely in epithelial and non-epithelial tissues, including pancreatic acinar cells (71, 147). KCNQ1 channels associate with all five members of the KCNE β-subunit family, resulting in a β-subunit-specific change of the current characteristics (71, 147). Kv7.1 K+ channels are blocked by the chromanol 293B (87) and by the more potent and selective derivative HMR1556 (146). Indeed, we showed that 293B selectively blocks osmotic swelling of ZG induced by activation of the K+ permeability (IC50 ~10 µM) (84). Upon incorporation of ZGM into planar bilayer membranes, K+ selective channels were detected with linear current-voltage relationships. Single channel analysis identified several K+ channel groups with distinct channel behaviors. K+ channels were inhibited by 100µM 293B or HMR1556, but not by the maxiKCa channel inhibitor, charybdotoxin (5nM). Kv7.1 protein was demonstrated by immunoperoxidase labelling of rat pancreatic tissue, immunogold labelling of ZG and immunoblotting of ZGM. Moreover, 293B also inhibited cholecystokinin (CCK)-induced amylase secretion of permeabilized acini (IC50 ~10µM) when applied together with the ZG nonselective cation conductance blocker, flufenamate. Thus these data indicated that Kv7.1 accounts at least partially for ZG K+ conductance and contributes to pancreatic secretagogue-stimulated enzyme and fluid secretion (84).
A question of some significance for the discussion of ZG K+ channels is whether the presence of several K+ channels with distinct biophysical properties in our bilayer experiments (84) argues against the purity of the ZGM used for single channel studies with the planar bilayer technique. However, the observation by others that single patched ZG also display a mixture of channel types (78) supports our model that ZG carry several different cation and anion conductive pathways (reviewed in (138). To further exemplify the concept that several conductive pathways are localized in ZGM, a study should be cited that investigated the effect of 293B and HMR-1556 on fluid and enzyme secretion induced by acetylcholine in the perfused rat pancreas (81). Interestingly, the authors were unable to observe any inhibitory effect of Kv7.1 channel blockers on secretion and concluded that Kv7.1 is not essential for secretagogue-mediated secretion of pancreatic acini. These results appear to question a role of Kv7.1 in enzyme secretion of rat pancreatic acini, as described in our study (84). However, we showed that CCK-stimulated enzyme secretion in permeabilized rat pancreatic acini was only abolished if flufenamate was applied together with 293B (see above), which is a strong indication that several ZG cation channels with functional redundancy are involved in enzyme secretion (84).
In a more recent study, we followed up the role of the ZG K+ conductance in CCK-induced enzyme secretion and showed that the immunosuppressive drug cyclosporin A (CsA) (but not FK506, another commonly used immunosuppressant) activates ZG K+ conductance and selectively increases the open probability of ZG K+ channels incorporated into planar bilayers (83). The electrophysiological (and other) data let us conclude that CsA has a direct effect on the underlying K+ channel (83). CsA also increased basal enzyme release of permeabilized rat pancreatic acini, but did not enhance CCK-induced enzyme secretion (83). These results are concordant with previous studies that had investigated the role of CsA on exocrine pancreas function (53, 61), although these authors suggested a different mode of action of CsA. We speculated that selective activation of ZG K+ channels by the immunosuppressant CsA may cause an increased intracellular release of digestive enzymes due to osmotic swelling and lysis of ZG in situ, and that this pathological process may account for the increased incidence of post-transplant allograft pancreatitis following hypoxia-reperfusion injury in patients treated with CsA (but not with FK506) (83).
In summary, although contamination by other organelles and membranes cannot be completely excluded, it seems likely that several K+ channels with different biophysical properties and pharmacological profiles are expressed in ZGM. This conclusion is supported by different electrophysiological techniques (incorporation of ZGM in planar bilayers and single ZG patch techniques), as well as by the detection of several different K+ channel proteins by immunological or proteomic approaches. However, as for AQPs, the significance of ZG K+ channels for the physiology of secretion by pancreatic acinar cells remains unclear.
C. Cl- Channels
ClC Cl- channels and transporters
X-ray microanalysis studies of pancreatic acinar cells have demonstrated that the ZG Cl- concentration is about half the cytosolic Cl- concentration (106). When measured with a Cl- sensitive fluorescent dye, cytosolic Cl- concentrations vary between 63 and 83 mM in resting cells, (81, 164), suggesting that ZG Cl- concentrations are well below those values. Early osmotic swelling studies with isolated ZG postulated the presence of regulated Cl- channels (reviewed in reference (138)). Subsequently, several candidates for ClC ion channels (70) have been identified in ZGM that have been linked to ZG Cl- conductive pathways. The earliest report suggested the presence of the Cl- channel ClC-2 in ZGM (20). Using the whole-cell patch-clamp technique, the authors identified a hyperpolarization-activated Cl- current in isolated pig pancreatic acinar cells. This current had the characteristic biophysical properties of ClC-2 (145) and was activated by extracellular hypotonicity, similar to ClC-2 (54), suggesting a role in volume regulation. An antiserum raised against the C-terminus of ClC-2 localized the channel to secretory granules containing amylase by immunofluorescence microscopy of acinar cells, and the authors suggested that the channel protein incorporates into the apical plasma membrane following granule exocytosis (20). By measuring whole vesicle currents of patched single ZG and using KCl as permeating ions, Jena and coworkers (78) detected a conductance that could be reduced by the non-specific anion transport blocker DIDS (40 µM) by ~50% and concluded that Cl- channels/transporters were present in ZGM. Immunoblots of ZG with ClC-2 and ClC-3 antibodies showed several bands, including immunoreactive bands of ~100 kDa, as expected for ClC-2 and ClC-3 (12, 134). However, ClC-2 (as well as ClC-1) is a plasma membrane Cl- channel, whereas CIC-3 through ClC-7 2Cl-/H+ exchangers are predominantly localized in intracellular vesicles of the endosomal/lysosomal system (70), further arguing against a significant role of ClC proteins in the secretory pathway. Yet proteomic analyses of rat ZGM identified ClC-3 and ClC-5, thus supporting the notion that ClC channel proteins are expressed in ZGM (52, 124). Although the lack of pancreas or salivary phenotype of ClC-2 (12) and ClC-3 knockout mice (6) does not exclude that ZG express these channel proteins - also because of the assumed functional redundancy of ZG ion channel proteins (138) - the evidence provided for ClC-2 and ClC-3 expression in secretory granules largely depends on immunological studies. However, these studies have to be taken with caution as long as negative control experiments with ZG from knockout tissues have not been performed, thus proving specificity of the antibodies (see (69) for a critical discussion). Hence, the decisive experiments proving expression of ClC proteins in ZG are still missing.
Other candidate Cl- channels
Another possible candidate for ZG anion channels may be AQP6 that is unique among the AQPs because it is activated by Hg2+ rather than being inhibited as for other AQPs; and it does not operate as a water channel, but as an anion permeable channel with the halide permeability sequence NO3- I- Br- Cl- F- (66) (reviewed in (158)). Analysis of sequence alignments led to the identification of a critical amino acid residue responsible for anion permeability: An asparagine residue (Asn-60) at the position corresponding to Gly-57 in other AQP H2O channels is responsible for anion permeation (66, 89, 159). Interestingly, AQP6 is almost exclusively localized in acidic intracellular organelles where it co-localizes with vacuolar-type H+-ATPases (V-ATPases) (160) and is activated by acidic pH (66, 159) (reviewed in (158)). Although no evidence is available for AQP6 localization in pancreatic ZGM, AQP6 has been detected in secretory granules of parotid acinar cells by immunological methods (96). Furthermore, osmotic swelling and lysis of secretory granules suspended in iso-osmotic KCl buffer displayed the same conductive properties as the native AQP6 channel when induced by the AQP6 activator Hg2+ (95). Interestingly, osmotic swelling showed a Hg2+-independent component (about 33%) and Hg2+-dependent swelling was about 50% DIDS-sensitive, again suggesting at least three different Cl- conductive pathways (however without functional redundancy) in these granules.
A family of putative anion channel proteins predominantly expressed in epithelial tissues that are gated by Ca2+ were named CLCA (chloride channel, Ca2+-activated), but rather represent chloride channel accessory modulatory proteins (see below and (92)). Mouse CLCA1/2 (mCLCA1/2) was detected in ZGM with a variety of immunological techniques using several antibodies that were thoroughly characterized (128, 143). Nevertheless, the gold standard of antibody characterization (namely to test the specificity of antibodies in knockout tissues used as negative controls) was not performed, because mCLCA1/2 knockout mice were not available at the time of those studies. In one study, an anion permeability was demonstrated using a ZG osmotic swelling assay whose permeability sequence, Ca2+ dependence and inhibitor sensitivity was reminiscent of CLCA associated anion currents (143). It is now established that hCLCA1, the human homolog of mCLCA3 that is expressed in mucous cells of the airway epithelium is a secreted protein rather than being a Cl- channel (49) although it seems to modulate the conductance of endogenous Ca2+-activated Cl− channels (58). It is likely that all the CLCA proteins cannot act in solo as ion channels but instead interact with other proteins in a possible signaling or regulatory capacity (reviewed in (117)). Their role in ZG function is unclear and awaits further elucidation.
Whether a member of the TMEM16/anoctamin (ANO) protein family of Ca2+-activated Cl- channels and phospholipid scramblases (119, 154) contributes to ZG anion permeability remains an interesting question (79), especially because the Ca2+-activated Cl- channel TMEM16A/ANO1 is expressed in the area of pancreatic acinar ZG (157). Interestingly, TMEM16A/ANO1 is permeated by HCO3-, a selectivity process which is thought to be regulated by the Ca2+/calmodulin complex (76). Hence, TMEM16A/ANO1-dependent HCO3- secretion through ZGM fused with the apical plasma membrane may also contribute to solubilization of exocytosed secretory products (123) during secretion of pancreatic acinar cells stimulated by Ca2+-dependent secretagogues (59).
In summary, although there is some suggestive evidence for the expression of intracellular ClC Cl- channel proteins, AQP6 and TMEM16A/ANO1 in ZGM, no strong candidate for a ZG Cl- channel has been identified.
D. Ca2+ Channels
Pancreatic ZG are a significant Ca2+ store (30, 106, 125), although the majority of ZG Ca2+ appears to be bound to proteins (30, 47, 156). Depending on the Ca2+ sensor used, the free Ca2+ concentration in the ZG lumen varies between 9 µM (156) and 55 µM (47). How Ca2+ accumulates in ZG is unclear, but Ca2+ uptake could occur in compartments of the secretory pathway upstream of secretory granules, e.g. the rER or the Golgi apparatus, and involve active transport mediated by different Ca2+-ATPases, such as SERCA (Sarcoplasmic/endoplasmic Reticulum Calcium ATPase) and SPCA (Secretory Pathway Calcium ATPase) pumps (152). Ca2+ released from ZG into the pancreatic juice may have a paracrine function on pancreatic duct cells that express Ca2+-sensing receptors on their apical surface, as proposed by Bruce et al. (15).
Secretagogues have been shown to evoke local increases of cytosolic Ca2+ in the secretory pole region of pancreatic acinar cells (148). Following studies demonstrating IP3-induced Ca2+ release from acidic neuroendocrine chromaffin granules induced by the second messenger inositol 1,4,5-trisphosphate (IP3) (162), experimental evidence has suggested that secretory granules from both endocrine and exocrine cells, including pancreatic acinar and airway goblet cells, are IP3-sensitive Ca2+ stores that play a significant role in Ca2+-dependent secretion (reviewed in (121, 161). In particular, using ZG isolated by differential centrifugation or intact pancreatic acinar cells, Petersen and colleagues have demonstrated release of Ca2+ from an acidic store in the apical pole of acinar cells induced by IP3, cyclic-ADP ribose (cADPR) or nicotinic acid adenine dinucleotide phosphate (NAADP) (i.e. suggestive of functional IP3- and ryanodine receptors) that they associated with ZG (46, 47, 149). Moreover, Gerasimenko and coworkers (44, 45) (reviewed in (121)) hinted at the importance of this acidic ZG store for the pathogenesis of alcohol-induced pancreatitis. However, two independent studies have challenged these observations in pancreatic (163) and parotid acinar cells (103). These authors provided functional and biochemical evidence indicating that ZGM do not express IP3- or ryanodine receptors, provided that ZG are not contaminated by other organellar structures, especially ER or mitochondria. Although Gerasimenko et al. (47) had shown a three-fold enrichment of the ZG marker α-amylase in the ZG preparation, the assay used to exclude ER contamination relied on the fluorescent dye DiOC6 (3) that is generally applied to assess ΔΨ of energized mitochondria [sic!] (77). Using the ZG isolation procedure of Gerasimenko et al. (47), Yule et al. (163) showed disrupted mitochondria and ER membranes by electron microscopy that contaminated the ZG preparation. Interestingly, Gerasimenko et al. (43) revisited this issue and proposed that the main apical Ca2+ pools that are responsible for release of Ca2+ by all three intracellular Ca2+ messengers IP3, cADPR and NAADP are the ER and ill-defined “acidic stores”, where these second messengers elicit Ca2+ release by activation of the Ca2+ channels IP3- and ryanodine receptors. The two stores interact with each other via Ca2+-induced Ca2+ release and can be separated using pharmacological tools. The ER relies on SERCA pumps that can be blocked by the specific inhibitor, thapsigargin; Ca2+ uptake in the acidic store relies on a bafilomycin-sensitive V-ATPase and probably a Ca2+/H+ exchanger, as originally proposed by Thévenod & coworkers for an IP3-sensitive ER Ca2+ pool (140, 142). Gerasimenko et al. suggested that a large part of the acidic store is “co-localized” with ZG, however, other acidic organelles like endosomes and lysosomes may also participate in intracellular Ca2+ signaling (43). This conclusion is more cautious and accurate than the earlier proposal of ZG as acidic Ca2+ stores but is still not satisfying. Three lines of evidence argue against ZG being an acidic Ca2+ store involved in secretagogue-induced apical Ca2+ release. Firstly, the majority of ZG are not acidic (111) (see also the paragraph on Vacuolar-type H+-ATPase); secondly, ER also co-localizes with ZG (48, 151); thirdly, an acidic IP3-sensitive ER Ca2+ pool has also been described that takes up Ca2+ via parallel activity of a V-type H+ pump and a Ca2+/H+ exchanger, both in pancreatic (140, 142) and parotid acinar cells (144). It also appears unlikely that ZG are an IP3 sensitive Ca2+ pool because all proteomic studies of ZGM so far did not detect any IP3 receptors (10, 11, 22-26, 124, 135). Hence, it remains an open question whether other mechanisms of Ca2+ release are operative in ZG.
The ATP-gated cation channel P2X4 is Ca2+ permeable (107) and has been identified in ZGM (124) (see the paragraphs on Proteomics and Vesicular nucleotide transporter SLC17A9). This receptor could play a role in Ca2+ release from weakly acidic Ca2+ stores (118), such as ZG, because P2X4 receptors are normally inhibited at acidic pH and are activated when intraluminal pH increases (39, 64, 107).
Finally, Hille and coworkers (37) have shown a release of Ca2+ via “store-operated” Orai channels in the membrane of secretory granules from neuroendocrine PC12 cells (interestingly, they did not detect IP3 receptors in these secretory granules, in contrast to previous studies (161)). The granule Orai channels are activated by regulator stromal interaction molecule 1 (STIM1) on the ER and may raise local cytoplasmic Ca2+ concentrations for refilling of Ca2+ stores of the ER and promotion of exocytosis (37). A subsequent study in RBL-2H3 mast cells identified Orai-2 on secretory granules (67). Ca2+ release from a Ca2+ store induced by antigen stimulation was attenuated by Orai-2 silencing, while that induced by thapsigargin was not affected. Furthermore, exocytotic release induced by antigen stimulation was inhibited in Orai-2 silenced cells (67).
IV. TRANSPORTERS
A. Vacuolar-type H+-ATPase
One of the hallmarks of many vesicular compartments is the expression of a V-ATPase that operates to acidify the vesicular lumen (40). Subunits of the V-ATPase have been clearly identified in the membrane of rat pancreas ZG by immunological methods (127) and by proteomic analyses (22, 23, 26, 124). Nevertheless, conflicting results were obtained in several early studies when measuring luminal pH of secretory granules in freshly isolated acini or purified granules. Using fluorescence microscopy and the weak base acridine orange (AO) that accumulates in acidic organelles and changes its fluorescence from green to orange when concentrated, ZG were found to be acidic whereas parotid secretory granules showed a neutral pH (7, 8, 35, 105). To resolve this issue, in a landmark study Orci et al. (111) used the probe 3-(2,4-dinitroanilino)-3-amino-N-methyldipro-pylamine (DAMP), a weak base that accumulates in the lumen of acidic organelles, where it can be fixed (5). Quantitative immunostaining of DAMP in conjunction with electron microscopy was used to measure pH in the secretory pathway of fixed isolated pancreatic acini. The data revealed that condensing vacuoles are acidic but lose their acidity during maturation (111). To account for these results, the authors speculated that the loss of acidity of maturing ZG may be the consequence of either removal of H+-pump subunits, inactivation of the V-ATPase, or closing of an anion conductance. It is noteworthy that it took more than 20 years to confirm the observation that the acidity of ZG decreases during maturation, using freshly isolated pancreas acini with AO and live-cell two-photon imaging (9), thus indicating that earlier AO measurements with fluorescence microscopic techniques were not accurate. Of note, the study by Thorn and coworkers (9) also showed that the H+ concentration of mature granules is not affected by pre-incubation with a blocker of the V-ATPase. However, in contrast to the data by Orci et al. (111), labeling of the cells with the ratiometric pH indicator, Lysosensor Yellow/ Blue, suggested that mature secretory granules have a pH of less than 6.0 (yet without proof that the labelled acidic structures are indeed mature ZG) (9). Furthermore, upon secretagogue-stimulated secretion, the pH in the acinar lumen never dropped below 7.0, which suggests that ZG pH is not or weakly acidic. To account for this discrepancy, Thorn and coworkers have subsequently described a luminal secretagogue-stimulated HCO3- conductance, mediated by TMEM16A/ANO1 that is activated coincidently with ZG fusion and exocytosis (59).
An increase of pH in the secretory pathway during maturation is anomalous because along the secretory pathway luminal pH generally drops from a near-cytosolic value of 7.2 within the ER to 6.7–6.0 along the Golgi complex to approximately 5.2–5.7 in secretory granules and is often associated with a reciprocal decrease of a H+ permeability (see (21, 116) for review). This is particularly the case in neuroendocrine cells where the increasing electrochemical gradient H+ gradient generated by the V-ATPase is also used to store cargo (e.g. neurotransmitters or metal ions) above equilibrium with the cytosol (40, 74). In contrast, mature pancreatic ZG do not store ATP (57) or Zn2+ (106) above cytosolic concentrations, which is in line with a reduced acidity of mature ZG. How ZG (as well as parotid secretory granules (see (7, 8)) lose their acidity during maturation, remains to be investigated. This could be the consequence of a differential activity or expression of V-ATPases and/or differences in H+ permeability, as suggested by Orci et al. (111). Moreover, activity of exchange pathways for counter-ions, e.g. Na+/H+-, Ca2+/H+- or Cl-/H+-exchangers (3, 4, 112, 133) along the secretory pathway of pancreatic (and parotid) acinar cells could also result in a dissipation of the pH gradient during maturation.
B. Vesicular nucleotide transporter SLC17A9
SLC17A9 (also named VNUT for Vesicular Nucleotide Transporter) is a vesicular ATP transporter widely expressed in various organs, but predominantly in the adrenal gland, brain, and thyroid gland (the pancreas was not tested) (129) (reviewed in (60, 101)). SLC17A9 mediates concentrative accumulation of ATP in secretory vesicles, such as chromaffin or synaptic vesicles, to a luminal concentration of 100 mM or more (38); reviewed in references (17, 73)). SLC17A9 protein shows a relative mobility varying between 61 and 68 kDa in SDS-PAGE, depending on the origin of the protein (native or recombinant). When reconstituted into proteoliposomes, SLC17A9 transports ATP (as well as ADP, GTP and UTP as shown by cis inhibition experiments) and uses a ΔΨ (positive inside) but not a ΔpH (the vesicle lumen being acidic) as driving force for uptake. ATP transport depends on external Cl- and saturates at ~4 mM external Cl-. ATP uptake is inhibited by DIDS) with an IC50 of ~1.5 µM. Evans blue is even more potent (IC50 ~40 nM) whereas 200 µM atractyloside inhibits SLC17A9 only in the presence of Mg2+. All these properties are similar to those that have been reported for ATP uptake in chromaffin granules and granule membrane ghosts (73). Although ATP transport rate appeared to be independent of Ca2+ or Mg2+, subsequent studies showed that SLC17A9 also transports both divalent cations in an ATP- and Cl-- dependent manner, which is driven by an inside-positive ΔΨ, but not by an inside-positive ΔpH, and with a similar inhibitor sensitivity as ATP transport (100). Hence, divalent cation transport probably occurs by complexation with ATP and may contribute to vesicular accumulation of divalent cations in secretory vesicles.
It has been suggested that during pancreas secretion evoked by secretagogues, ATP is released from ZG into the primary fluid to mediate paracrine signaling between acinar and duct cells and thereby coordinates ductal and acinar fluid secretion (108). This may occur via purinergic receptors expressed on the apical membrane of duct cells (93). Indeed, pancreatic ZG do contain ATP. From measurements with freshly isolated ZG, it has been estimated that the in vivo concentration of ATP in ZG is 0.5-1 mM ATP (57), a more than 100-times lower concentration than in neuroendocrine secretory vesicles expressing SLC17A9 and a concentration that is also below the average cytosolic concentration of 2-5 mM ATP (17), thus questioning concentrative ATP transport in ZG. Nevertheless, Novak and coworkers have suggested that SLC17A9 is expressed in the membrane of ZG where it mediates ATP uptake (56, 57). Their experimental evidence supports expression of SLC17A9 in ZGM, e.g. by detection of a ca. 65-kDa protein with a SLC17A9-specific antiserum, co-localization of immunolabeling for SLC17A9 with the ZG membrane marker Rab3D, or inhibition of ATP uptake into ZG by 100 µM DIDS and Evans blue (56, 57). Furthermore, induction of rat AR42J acinar cells into a secretory phenotype by dexamethasone administration increases carbachol-induced ATP release and SLC17A9 expression. In Haanes’ and Novak’s work on isolated ZG, ATP uptake depends on external Mg2+ and high Cl- (56, 57). Moreover, ATP uptake is driven by an inside-positive ΔpH (either generated by an artificial pH gradient or as demonstrated by bafilomycin A1-sensitive uptake) and by an inside-positive K+ diffusion potential. Furthermore, ATP uptake shows no cis inhibition by ADP or UTP (57). As a matter of fact, most of these properties differ from the transport properties of SLC17A9 (129) (reviewed in (60, 101)). It may be argued that results obtained in intact ZG may differ from transport of a protein reconstituted into proteoliposomes (57). However, transport of reconstituted SLC17A9 matches transport in intact chromaffin granules (73, 129). Hence, ATP transport in ZG may occur independently from SLC17A9 expression.
On the other hand, these observations in ZG have gained recent support from studies in lysosomes (19, 64) and lamellar bodies (LB) (39). SLC17A9 is expressed in these acidic intracellular organelles, which contain ATP concentrations between <0.5 mM (in lysosomes) and about 2 mM in lysosomal bodies. Interestingly, SLC17A9 expression was functionally linked to the ATP-gated cation channel P2X4 (39, 166) (reviewed in (102)), which is also expressed in ZGM (124) (see the paragraphs on Proteomics and Ca2+ channels). The study by Fois et al. (39) demonstrates a physiological role of lysosomal P2X4 receptors during the secretion of surfactant from alveolar type II epithelial cells. Apparently, LB fusion triggers P2X4 receptors within the LB membrane to generate a highly localized, cytosolic Ca2+ signal in the vicinity of the fused LB, which drives expansion of the fusion pore and facilitates surfactant release (reviewed in (102)). LB are likely lysosome-derived but less acidic than juxtanuclear lysosomes by virtue of reduced V-ATPase activity and an increase in proton leak (72), once more emphasizing the importance of electrogenic conductive pathways for ions, in particular H+, for concentrative ATP uptake in intracellular vesicles.
In conclusion, experiments providing evidence for a causal relationship between ZG ATP accumulation and SLC17A9 are needed to confirm the postulated role of SLC17A9 in (concentrative?) ATP uptake into ZG. This requires, for instance, silencing of the transporter and measurements of ZG ATP concentration and/or secretagogue-induced ATP release from acinar cells, as well as measurements of the driving force for ATP accumulation, namely Ψ, in ZG. Moreover, it remains to be tested whether SLC17A9 - if it is indeed expressed in ZGM - may also contribute to the accumulation of Ca2+ as nucleotide complex in ZG that, apart from the endoplasmic reticulum represent a significant intracellular Ca2+ pool (30, 125, 126) (see also the paragraph on Ca2+ channels). In summary, the available evidence that SLC17A9 is responsible for concentrative ATP uptake into pancreatic ZG awaits further experimental confirmation.
C. Zinc transporter SLC30A2
The SLC30A2 gene encodes a zinc (Zn2+) transporter (also named ZnT2 for Zinc Transporter 2) that was cloned from a rat kidney cDNA expression library by complementation of a Zn2+-sensitive Baby Hamster Kidney cell line (114) and is highly expressed in pancreas (reviewed in (63, 91, 130)). Using ZnT2-GFP fusion proteins and immunofluorescence microscopy, the transporter was localized in Zn2+ accumulating acidic intracellular vesicles (as opposed to the Zn2+ efflux transporter SLC30A1/ZnT1 that is expressed in the plasma membrane (115)). The authors suggested that ZnT2 contributes to protection against Zn2+ toxicity by facilitating Zn2+ influx into an endosomal/lysosomal compartment thereby lowering cytosolic Zn2+ jointly with plasma membrane ZnT1 (114). Subsequent studies have revealed that, in addition to the pancreas, SLC30A2 is also expressed in mammary gland, prostate, retina, small intestine and kidney. In these organs, it is localized on the membrane of intracellular organelles (e.g. endosomal/lysosomal and secretory vesicles) and sequesters cytoplasmic Zn2+ for secretion, storage, or for use in proteins that require Zn2+ for their activities (reviewed in (63, 91, 130)). Interestingly, a loss-of-function mutation or silencing of SLC30A2 results in reduced Zn2+ secretion from mammary gland epithelial cells in vivo and in vitro (29). Subsequently, transient neonatal Zn2+ deficiency (TNZD) in infants was found to be caused by a variety of loss-of-function mutations in the SLC30A2 gene in women, resulting in poor secretion of Zn2+ into the breast milk. Consequently, infants exclusively breastfed with Zn2+-deficient breast milk develop severe Zn2+ deficiency (reviewed in (51)). The transport mechanism of SLC30A2 has been recently elucidated (50): Similarly to vesicular Zn2+ transport mediated by other mammalian SLC30 transporters (SLC30A1 and SLC30A5) (109, 131), SLC30A2 is an electroneutral proton-coupled vesicular antiporter displaying an apparent stoichiometry of two H+ per Zn2+ ion and is driven by the vesicular H+ gradient generated by V-ATPases (50).
In pancreatic endocrine β-cells, secretory granules accumulate Zn2+ almost 40-fold above cytosolic levels, whereas no difference between cytosolic and acinar ZG Zn2+ concentration has been found, as measured by X-ray microanalysis (106). In β-cells granules (and other islet secretory granules), SLC30A8 is the Zn2+ transporter that provides Zn2+ for maturation of stored macromolecules, such as insulin, and their crystallization before secretion (27), and its possible importance in the etiology of diabetes has been highlighted (85). Because insulin containing granules are acidic (1, 65, 113), SLC30A8-mediated Zn2+ accumulation against a concentration gradient is likely driven by the vesicular H+ gradient generated by V-ATPases and could occur via H+/Zn2+ exchange, as demonstrated for SLC30A1, SLC30A2 and SLC30A5 (50, 109, 131).
The situation in ZG is much less clear. In rats, Zn2+ deficiency caused Zn2+ depletion in acinar but not in β-cells (132), which is in line with the ability of β-cells to store Zn2+ in insulin granules in a slowly-exchanging intra-granule compartment and also with the apparent inability of pancreatic ZG to accumulate Zn2+ (106). Liuzzi et al. (90) showed that Zn2+ depletion in mice is associated with a reduction of pancreatic acinar SLC30A1 and SLC30A2, and the authors speculated that SLC30A1/2 may mediate lowering of cytosolic Zn2+ in pancreatic acinar cells of Zn2+ replete animals. Furthermore, De Lisle and coworkers showed secretion of the cytosolic Zn2+-binding protein metallothionein into pancreatic juice (34). All these studies led to the hypothesis by Cousins and colleagues that exocytotic secretion of Zn2+ by pancreatic acinar cells may be modulated by Zn2+ transporter expression, which would thus affect entero-pancreatic Zn2+ trafficking and also regulate physiological Zn2+ body homeostasis by controlling Zn2+ losses into the pancreatic fluid and intestinal excretion of Zn2+ (31, 90). More recently, Cousins and coworkers demonstrated SLC30A2 expression in ZG, and reduction of the Zn2+ diet of mice was paralleled by decreases of Zn2+ concentrations and SLC30A2 expression in ZG (55>). Rat AR42J acinar cells, when induced into a secretory phenotype with dexamethasone, exhibited increased SLC30A2 expression that was associated with a reduction of cytosolic 65Zn2+ content and an increase of the 65Zn2+ content of ZG. SLC30A2 silencing of AR42J cells by RNAi (~70% mRNA reduction) increased cytoplasmic 65Zn2+ by 36% and decreased ZG 65Zn2+ by 15%, suggesting that SLC30A2 mediates the sequestration of Zn2+ into ZG (55). The relatively low impact of SLC30A2 silencing on ZG Zn2+ content may be explained by low ZG Zn2+ below cytosolic concentrations, which is likely caused by the loss of acidity of ZG during maturation (see the paragraph on Vacuolar-type H+-ATPase). Alternatively, other Zn2+ transporters may also contribute to Zn2+ fluxes across the ZG membrane.
What could be the role of Zn2+ in ZG? For instance, it is known that ZG contain Zn2+-metalloenzymes, such as carboxypeptidases that require Zn2+ for their function (62, 150), but also for inhibition of their function at higher (micromolar) Zn2+ concentrations (80>). Although SLC30A2 is expressed in ZG, Zn2+ uptake via SLC30A2 (or other members of the SLC30 protein family) is more likely to be operative in precursor acidic organelles of the secretory pathway, e.g. the Golgi apparatus or immature granules after protein synthesis (120). Whether secretagogue-evoked exocytotic secretion of Zn2+ from ZG (55) and/or secretion of Zn2+-metallothionein (34) are effective means to regulate Zn2+ body homeostasis remains to be shown. In fact, regulation of the expression of transporters for Zn2+ uptake (from dietary sources and/or the pancreatic juice) in the intestinal mucosa may be a more effective and sensitive means to control Zn2+ body homeostasis (32, 75, 94). However, a divergent view, based on zebrafish studies, concluded that the responses of epithelial tissues to Zn2+ deficiency and excess are best explained by local epithelial homeostasis, with no evidence of systemic control (165).
V. CONCLUSION AND PERSPECTIVES
From a scientific perspective, the major unresolved issue is not so much the presence/absence of specific pancreatic ZG channels and transporters - there is no doubt that channels and transporters are expressed in the membrane of ZG - but rather a clarification of their physiological role in pancreatic acinar cells during the process of maturation, fusion and exocytosis of ZG. Here the development of organ-specific channel/transporter knockout animal models is mandatory to move the field forward. In this context the anomalous property of the pancreatic acinar cell, which displays a decreasing ZG acidity during maturation (see Figure 1,and the paragraph on Vacuolar-type H+-ATPase) remains an enigma because other secretory cells, in particular neuroendocrine cells, rather exhibit increasing acidity along the secretory pathway (21, 116). This is particularly puzzling because studies in neuroendocrine cells have highlighted the important role of the V0 domain of the V-ATPase as a sensor of intragranular pH that identifies and regulates the ability of granules to undergo exocytosis (122, 153). Hence, the functional implication of a decreasing acidity of maturing ZG for exocytosis needs to be addressed. Moreover, more studies investigating differences in H+ permeability (116) and/ or activity of exchange pathways for counter-ions, e.g. Na+/H+-, Ca2+/H+- or Cl-/H+-exchangers (3, 4, 112, 133) along the secretory pathway of pancreatic acinar cells and their contribution to dissipation of the pH gradient during maturation are required, to better understand their significance in the process of regulated exocytotic secretion.
As this review has sought to demonstrate, many unsolved – and neglected – problems remain in ZG physiology. They need to be addressed and require clarification before we can frame accurate concepts of efficient preventive and therapeutic strategies for diseases of the pancreas, such as acute and chronic pancreatitis.
VI. ACKNOWLEDGEMENTS
I thank Prof. John A. Williams for contacting and encouraging me to write this review. I am also grateful to my collaborator, Prof. Wing-Kee Lee, for continuous backing. Financial support for my studies on zymogen granules was obtained from the Deutsche Forschungsgemeinschaft, Deutsche Mukoviszidose e.V. and the University of Witten/Herdecke (ZBAF).
VII. REFERENCES
- Abrahamsson H, and Gylfe E. Demonstration of a proton gradient across the insulin granule membrane. Acta Physiol Scand 109: 113-114, 1980. PMID: 6255750.
- Abu-Hamdah R, Cho WJ, Cho SJ, Jeremic A, Kelly M, Ilie AE, and Jena BP. Regulation of the water channel aquaporin-1: isolation and reconstitution of the regulatory complex. Cell Biol Int 28: 7-17, 2004. PMID: 14759764.
- Anderie I, Blum R, Haase W, Grinstein S, and Thévenod F. Expression of NHE1 and NHE4 in rat pancreatic zymogen granule membranes. Biochem Biophys Res Commun 246: 330-336, 1998. PMID: 9610358.
- Anderie I, and Thévenod F. Evidence for involvement of a zymogen granule Na+/H+ exchanger in enzyme secretion from rat pancreatic acinar cells. J Membr Biol 152: 195-205, 1996. PMID: 8672081.
- Anderson RG, and Pathak RK. Vesicles and cisternae in the trans Golgi apparatus of human fibroblasts are acidic compartments. Cell 40: 635-643, 1985. PMID: 3882239.
- Arreola J, Begenisich T, Nehrke K, Nguyen HV, Park K, Richardson L, Yang B, Schutte BC, Lamb FS, and Melvin JE. Secretion and cell volume regulation by salivary acinar cells from mice lacking expression of the Clcn3 Cl- channel gene. J Physiol 545: 207-216, 2002. PMID: 12433961.
- Arvan P, and Castle JD. Isolated secretion granules from parotid glands of chronically stimulated rats possess an alkaline internal pH and inward-directed H+ pump activity. J Cell Biol 103: 1257-1267, 1986. PMID: 3021780.
- Arvan P, Rudnick G, and Castle JD. Osmotic properties and internal pH of isolated rat parotid secretory granules. J Biol Chem 259: 13567-13572, 1984. PMID: 6490664.
- Behrendorff N, Floetenmeyer M, Schwiening C, and Thorn P. Protons released during pancreatic acinar cell secretion acidify the lumen and contribute to pancreatitis in mice. Gastroenterology 139: 1711-1720, 1720 e1711-1715, 2010. PMID: 20691184.
- Berkane AA, Nguyen HT, Tranchida F, Waheed AA, Deyris V, Tchiakpe L, Fasano C, Nicoletti C, Desseaux V, Ajandouz el H, Comeau D, Comeau L, and Hiol A. Proteomic of lipid rafts in the exocrine pancreas from diet-induced obese rats. Biochem Biophys Res Commun 355: 813-819, 2007. PMID: 17320817.
- Borta H, Aroso M, Rinn C, Gomez-Lazaro M, Vitorino R, Zeuschner D, Grabenbauer M, Amado F, and Schrader M. Analysis of low abundance membrane-associated proteins from rat pancreatic zymogen granules. J Proteome Res 9: 4927-4939, 2010. PMID: 20807389.
- Bosl MR, Stein V, Hubner C, Zdebik AA, Jordt SE, Mukhopadhyay AK, Davidoff MS, Holstein AF, and Jentsch TJ. Male germ cells and photoreceptors, both dependent on close cell-cell interactions, degenerate upon ClC-2 Cl- channel disruption. EMBO J 20: 1289-1299, 2001. PMID: 11250895.
- Braun M, Anderie I, and Thévenod F. Evidence for a 65 kDa sulfonylurea receptor in rat pancreatic zymogen granule membranes. FEBS Lett 411: 255-259, 1997. PMID: 9271216.
- Braun M, and Thévenod F. Photoaffinity labeling and purification of ZG-16p, a high-affinity dihydropyridine binding protein of rat pancreatic zymogen granule membranes that regulates a K+-selective conductance. Mol Pharmacol 57: 308-316, 2000. PMID: 1064840.
- Bruce JI, Yang X, Ferguson CJ, Elliott AC, Steward MC, Case RM, and Riccardi D. Molecular and functional identification of a Ca2+ (polyvalent cation)-sensing receptor in rat pancreas. J Biol Chem 274: 20561-20568, 1999. PMID: 10400686.
- Burnham DB, Munowitz P, Thorn N, and Williams JA. Protein kinase activity associated with pancreatic zymogen granules. Biochem J 227: 743-751, 1985. PMID: 4004796.
- Burnstock G. Physiology and pathophysiology of purinergic neurotransmission. Physiol Rev 87: 659-797, 2007. PMID: 17429044.
- Calamita G, Ferri D, Gena P, Liquori GE, Cavalier A, Thomas D, and Svelto M. The inner mitochondrial membrane has aquaporin-8 water channels and is highly permeable to water. J Biol Chem 280: 17149-17153, 2005. PMID: 15749715.
- Cao Q, Zhao K, Zhong XZ, Zou Y, Yu H, Huang P, Xu TL, and Dong XP. SLC17A9 protein functions as a lysosomal ATP transporter and regulates cell viability. J Biol Chem 289: 23189-23199, 2014. PMID: 24962569.
- Carew MA, and Thorn P. Identification of ClC-2-like chloride currents in pig pancreatic acinar cells. Pflugers Arch 433: 84-90, 1996. PMID: 9019735.
- Casey JR, Grinstein S, and Orlowski J. Sensors and regulators of intracellular pH. Nat Rev Mol Cell Biol 11: 50-61, 2010. PMID: 19997129.
- Chen X, and Andrews PC. Purification and proteomics analysis of pancreatic zymogen granule membranes. Methods Mol Biol 432: 275-287, 2008. PMID: 18370025.
- Chen X, and Andrews PC. Quantitative proteomics analysis of pancreatic zymogen granule membrane proteins. Methods Mol Biol 528: 327-338, 2009. PMID: 19153703.
- Chen X, Li C, Izumi T, Ernst SA, Andrews PC, and Williams JA. Rab27b localizes to zymogen granules and regulates pancreatic acinar exocytosis. Biochem Biophys Res Commun 323: 1157-1162, 2004. PMID: 15451418.
- Chen X, Ulintz PJ, Simon ES, Williams JA, and Andrews PC. Global topology analysis of pancreatic zymogen granule membrane proteins. Mol Cell Proteomics 7: 2323-2336, 2008. PMID: 18682380.
- Chen X, Walker AK, Strahler JR, Simon ES, Tomanicek-Volk SL, Nelson BB, Hurley MC, Ernst SA, Williams JA, and Andrews PC. Organellar proteomics: analysis of pancreatic zymogen granule membranes. Mol Cell Proteomics 5: 306-312, 2006. PMID: 16278343.
- Chimienti F, Devergnas S, Favier A, and Seve M. Identification and cloning of a beta-cell-specific zinc transporter, ZnT-8, localized into insulin secretory granules. Diabetes 53: 2330-2337, 2004. PMID: 15331542.
- Cho SJ, Sattar AK, Jeong EH, Satchi M, Cho JA, Dash S, Mayes MS, Stromer MH, and Jena BP. Aquaporin 1 regulates GTP-induced rapid gating of water in secretory vesicles. Proc Natl Acad Sci U S A 99: 4720-4724, 2002. PMID: 11917120.
- Chowanadisai W, Lonnerdal B, and Kelleher SL. Identification of a mutation in SLC30A2 (ZnT-2) in women with low milk zinc concentration that results in transient neonatal zinc deficiency. J Biol Chem 281: 39699-39707, 2006. PMID: 17065149.
- Clemente F, and Meldolesi J. Calcium and pancreatic secretion. I. Subcellular distribution of calcium and magnesium in the exocrine pancreas of the guinea pig. J Cell Biol 65: 88-102, 1975. PMID: 165206.
- Cousins RJ. Gastrointestinal factors influencing zinc absorption and homeostasis. Int J Vitam Nutr Res 80: 243-248, 2010. PMID: 21462106.
- Cragg RA, Phillips SR, Piper JM, Varma JS, Campbell FC, Mathers JC, and Ford D. Homeostatic regulation of zinc transporters in the human small intestine by dietary zinc supplementation. Gut 54: 469-478, 2005. PMID: 15753530.
- De Lisle RC, and Hopfer U. Electrolyte permeabilities of pancreatic zymogen granules: implications for pancreatic secretion. Am J Physiol Gastrointest Liver Physiol 250: G489-G496, 1986. PMID: 3754390.
- De Lisle RC, Sarras MP, Jr., Hidalgo J, and Andrews GK. Metallothionein is a component of exocrine pancreas secretion: implications for zinc homeostasis. Am J Physiol Cell Physiol 271: C1103-1110, 1996. PMID: 8897816.
- De Lisle RC, and Williams JA. Zymogen granule acidity is not required for stimulated pancreatic protein secretion. Am J Physiol Gastrointest Liver Physiol 253: G711-719, 1987. PMID: 2447787.
- Delporte C. Aquaporins in salivary glands and pancreas. Biochim Biophys Acta 1840: 1524-1532, 2014. PMID: 23954206.
- Dickson EJ, Duman JG, Moody MW, Chen L, and Hille B. Orai-STIM-mediated Ca2+ release from secretory granules revealed by a targeted Ca2+ and pH probe. Proc Natl Acad Sci U S A 109: E3539-3548, 2012. PMID: 23184982.
- Estevez-Herrera J, Dominguez N, Pardo MR, Gonzalez-Santana A, Westhead EW, Borges R, and Machado JD. ATP: The crucial component of secretory vesicles. Proc Natl Acad Sci U S A 113: E4098-4106, 2016. PMID: 27342860.
- Fois G, Winkelmann VE, Bareis L, Staudenmaier L, Hecht E, Ziller C, Ehinger K, Schymeinsky J, Kranz C, and Frick M. ATP is stored in lamellar bodies to activate vesicular P2X4 in an autocrine fashion upon exocytosis. J Gen Physiol 150: 277-291, 2018. PMID: 29282210.
- Forgac M. Vacuolar ATPases: rotary proton pumps in physiology and pathophysiology. Nat Rev Mol Cell Biol 8: 917-929, 2007. PMID: 17912264.
- Gasser KW, DiDomenico J, and Hopfer U. Potassium transport by pancreatic and parotid zymogen granule membranes. Am J Physiol 255: C705-711, 1988. PMID: 3202144.
- Gasser KW, and Holda JR. ATP-sensitive potassium transport by pancreatic secretory granule membrane. Am J Physiol 264: G137-142, 1993. PMID: 8430796.
- Gerasimenko J, Peng S, and Gerasimenko O. Role of acidic stores in secretory epithelia. Cell Calcium 55: 346-354, 2014. PMID: 24832105.
- Gerasimenko JV, Lur G, Ferdek P, Sherwood MW, Ebisui E, Tepikin AV, Mikoshiba K, Petersen OH, and Gerasimenko OV. Calmodulin protects against alcohol-induced pancreatic trypsinogen activation elicited via Ca2+ release through IP3 receptors. Proc Natl Acad Sci U S A 108: 5873-5878, 2011. PMID: 21436055.
- Gerasimenko JV, Lur G, Sherwood MW, Ebisui E, Tepikin AV, Mikoshiba K, Gerasimenko OV, and Petersen OH. Pancreatic protease activation by alcohol metabolite depends on Ca2+ release via acid store IP3 receptors. Proc Natl Acad Sci U S A 106: 10758-10763, 2009. PMID: 19528657.
- Gerasimenko JV, Sherwood M, Tepikin AV, Petersen OH, and Gerasimenko OV. NAADP, cADPR and IP3 all release Ca2+ from the endoplasmic reticulum and an acidic store in the secretory granule area. J Cell Sci 119: 226-238, 2006. PMID: 16410548.
- Gerasimenko OV, Gerasimenko JV, Belan PV, and Petersen OH. Inositol trisphosphate and cyclic ADP-ribose-mediated release of Ca2+ from single isolated pancreatic zymogen granules. Cell 84: 473-480, 1996. PMID: 8608601.
- Gerasimenko OV, Gerasimenko JV, Rizzuto RR, Treiman M, Tepikin AV, and Petersen OH. The distribution of the endoplasmic reticulum in living pancreatic acinar cells. Cell Calcium 32: 261-268, 2002. PMID: 12543088.
- Gibson A, Lewis AP, Affleck K, Aitken AJ, Meldrum E, and Thompson N. hCLCA1 and mCLCA3 are secreted non-integral membrane proteins and therefore are not ion channels. J Biol Chem 280: 27205-27212, 2005. PMID: 15919655.
- Golan Y, Alhadeff R, Warshel A, and Assaraf YG. ZnT2 is an electroneutral proton-coupled vesicular antiporter displaying an apparent stoichiometry of two protons per zinc ion. PLoS Comput Biol 15: e1006882, 2019. PMID: 30893306.
- Golan Y, Kambe T, and Assaraf YG. The role of the zinc transporter SLC30A2/ZnT2 in transient neonatal zinc deficiency. Metallomics 9: 1352-1366, 2017. PMID: 28665435.
- Gomez-Lazaro M, Rinn C, Aroso M, Amado F, and Schrader M. Proteomic analysis of zymogen granules. Expert Rev Proteomics 7: 735-747, 2010. PMID: 20973645.
- Groblewski GE, Wagner AC, and Williams JA. Cyclosporin A inhibits Ca2+/calmodulin-dependent protein phosphatase and secretion in pancreatic acinar cells. J Biol Chem 269: 15111-15117, 1994. PMID: 7515049.
- Grunder S, Thiemann A, Pusch M, and Jentsch TJ. Regions involved in the opening of CIC-2 chloride channel by voltage and cell volume. Nature 360: 759-762, 1992. PMID: 1334533.
- Guo L, Lichten LA, Ryu MS, Liuzzi JP, Wang F, and Cousins RJ. STAT5-glucocorticoid receptor interaction and MTF-1 regulate the expression of ZnT2 (Slc30a2) in pancreatic acinar cells. Proc Natl Acad Sci U S A 107: 2818-2823, 2010. PMID: 20133611.
- Haanes KA, Kowal JM, Arpino G, Lange SC, Moriyama Y, Pedersen PA, and Novak I. Role of vesicular nucleotide transporter VNUT (SLC17A9) in release of ATP from AR42J cells and mouse pancreatic acinar cells. Purinergic Signal 10: 431-440, 2014. PMID: 24488439.
- Haanes KA, and Novak I. ATP storage and uptake by isolated pancreatic zymogen granules. Biochem J 429: 303-311, 2010. PMID: 20441565.
- Hamann M, Gibson A, Davies N, Jowett A, Walhin JP, Partington L, Affleck K, Trezise D, and Main M. Human ClCa1 modulates anionic conduction of calcium-dependent chloride currents. J Physiol 587: 2255-2274, 2009. PMID: 19307298.
- Han Y, Shewan AM, and Thorn P. HCO3- Transport through Anoctamin/Transmembrane Protein ANO1/TMEM16A in Pancreatic Acinar Cells Regulates Luminal pH. J Biol Chem 291: 20345-20352, 2016. PMID: 27510033.
- Hasuzawa N, Moriyama S, Moriyama Y, and Nomura M. Physiopathological roles of vesicular nucleotide transporter (VNUT), an essential component for vesicular ATP release. Biochim Biophys Acta Biomembr 1862: 183408, 2020. PMID: 32652056.
- Hirano Y, Hisatomi A, Ohara K, and Noguchi H. The effects of FK506 and cyclosporine on the exocrine function of the rat pancreas. Transplantation 54: 883-887, 1992. PMID: 1279850.
- Hsu JM, Anilane JK, and Scanlan DE. Pancreatic carboxypeptidases: activities in zinc-deficient rats. Science 153: 882-883, 1966. PMID: 5914752.
- Huang L, and Tepaamorndech S. The SLC30 family of zinc transporters - a review of current understanding of their biological and pathophysiological roles. Mol Aspects Med 34: 548-560, 2013. PMID: 23506888.
- Huang P, Zou Y, Zhong XZ, Cao Q, Zhao K, Zhu MX, Murrell-Lagnado R, and Dong XP. P2X4 forms functional ATP-activated cation channels on lysosomal membranes regulated by luminal pH. J Biol Chem 289: 17658-17667, 2014. PMID: 24817123.
- Hutton JC. The internal pH and membrane potential of the insulin-secretory granule. Biochem J 204: 171-178, 1982. PMID: 6126183.
- Ikeda M, Beitz E, Kozono D, Guggino WB, Agre P, and Yasui M. Characterization of aquaporin-6 as a nitrate channel in mammalian cells. Requirement of pore-lining residue threonine 63. J Biol Chem 277: 39873-39879, 2002. PMID: 12177001.
- Ikeya M, Yamanoue K, Mochizuki Y, Konishi H, Tadokoro S, Tanaka M, Suzuki R, and Hirashima N. Orai-2 is localized on secretory granules and regulates antigen-evoked Ca2+ mobilization and exocytosis in mast cells. Biochem Biophys Res Commun 451: 62-67, 2014. PMID: 25044118.
- Itoh T, Rai T, Kuwahara M, Ko SB, Uchida S, Sasaki S, and Ishibashi K. Identification of a novel aquaporin, AQP12, expressed in pancreatic acinar cells. Biochem Biophys Res Commun 330: 832-838, 2005. PMID: 15809071.
- Jentsch TJ, Maritzen T, Keating DJ, Zdebik AA, and Thévenod F. ClC-3--a granular anion transporter involved in insulin secretion? Cell Metab 12: 307-308; author reply 309-310, 2010. PMID: 20889118.
- Jentsch TJ, and Pusch M. CLC Chloride Channels and Transporters: Structure, Function, Physiology, and Disease. Physiol Rev 98: 1493-1590, 2018. PMID: 29845874.
- Jespersen T, Grunnet M, and Olesen SP. The KCNQ1 potassium channel: from gene to physiological function. Physiology (Bethesda) 20: 408-416, 2005. PMID: 16287990.
- Johnson DE, Ostrowski P, Jaumouille V, and Grinstein S. The position of lysosomes within the cell determines their luminal pH. J Cell Biol 212: 677-692, 2016. PMID: 26975849.
- Johnson RG, Jr. Accumulation of biological amines into chromaffin granules: a model for hormone and neurotransmitter transport. Physiol Rev 68: 232-307, 1988. PMID: 2892215.
- Johnson RG, Jr. Proton pumps and chemiosmotic coupling as a generalized mechanism for neurotransmitter and hormone transport. Ann N Y Acad Sci 493: 162-177, 1987. PMID: 2884918.
- Jou MY, Hall AG, Philipps AF, Kelleher SL, and Lonnerdal B. Tissue-specific alterations in zinc transporter expression in intestine and liver reflect a threshold for homeostatic compensation during dietary zinc deficiency in weanling rats. J Nutr 139: 835-841, 2009. PMID: 19297427.
- Jung J, Nam JH, Park HW, Oh U, Yoon JH, and Lee MG. Dynamic modulation of ANO1/TMEM16A HCO3(-) permeability by Ca2+/calmodulin. Proc Natl Acad Sci U S A 110: 360-365, 2013. PMID: 23248295.
- Kataoka M, Fukura Y, Shinohara Y, and Baba Y. Analysis of mitochondrial membrane potential in the cells by microchip flow cytometry. Electrophoresis 26: 3025-3031, 2005. PMID: 16078196.
- Kelly ML, Abu-Hamdah R, Jeremic A, Cho SJ, Ilie AE, and Jena BP. Patch clamped single pancreatic zymogen granules: direct measurements of ion channel activities at the granule membrane. Pancreatology 5: 443-449, 2005. PMID: 15985770.
- Kunzelmann K, Tian Y, Martins JR, Faria D, Kongsuphol P, Ousingsawat J, Thevenod F, Roussa E, Rock J, and Schreiber R. Anoctamins. Pflugers Arch 462: 195-208, 2011. PMID: 21607626.
- Larsen KS, and Auld DS. Characterization of an inhibitory metal binding site in carboxypeptidase A. Biochemistry 30: 2613-2618, 1991. PMID: 2001351.
- Lee JE, Park HS, Uhm DY, and Kim SJ. Effects of KCNQ1 channel blocker, 293B, on the acetylcholine-induced Cl- secretion of rat pancreatic acini. Pancreas 28: 435-442, 2004. PMID: 15097862.
- Lee WK, Bork U, Gholamrezaei F, and Thévenod F. Cd2+-induced cytochrome c release in apoptotic proximal tubule cells: role of mitochondrial permeability transition pore and Ca2+ uniporter. Am J Physiol Renal Physiol 288: F27-F39, 2005. PMID: 15339793.
- Lee WK, Braun M, Langeluddecke C, and Thévenod F. Cyclosporin a, but not FK506, induces osmotic lysis of pancreas zymogen granules, intra-acinar enzyme release, and lysosome instability by activating K+ channel. Pancreas 41: 596-604, 2012. PMID: 22158076.
- Lee WK, Torchalski B, Roussa E, and Thévenod F. Evidence for KCNQ1 K+ channel expression in rat zymogen granule membranes and involvement in cholecystokinin-induced pancreatic acinar secretion. Am J Physiol Cell Physiol 294: C879-C892, 2008. PMID: 18216164.
- Lemaire K, Chimienti F, and Schuit F. Zinc transporters and their role in the pancreatic beta-cell. J Diabetes Investig 3: 202-211, 2012. PMID: 24843567.
- Lerch MM, and Gorelick FS. Models of acute and chronic pancreatitis. Gastroenterology 144: 1180-1193, 2013. PMID: 23622127.
- Lerche C, Bruhova I, Lerche H, Steinmeyer K, Wei AD, Strutz-Seebohm N, Lang F, Busch AE, Zhorov BS, and Seebohm G. Chromanol 293B binding in KCNQ1 (Kv7.1) channels involves electrostatic interactions with a potassium ion in the selectivity filter. Mol Pharmacol 71: 1503-1511, 2007. PMID: 17347319.
- Lesage F, and Lazdunski M. Molecular and functional properties of two-pore-domain potassium channels. Am J Physiol Renal Physiol 279: F793-801, 2000. PMID: 11053038.
- Liu K, Kozono D, Kato Y, Agre P, Hazama A, and Yasui M. Conversion of aquaporin 6 from an anion channel to a water-selective channel by a single amino acid substitution. Proc Natl Acad Sci U S A 102: 2192-2197, 2005. PMID: 15671159.
- Liuzzi JP, Bobo JA, Lichten LA, Samuelson DA, and Cousins RJ. Responsive transporter genes within the murine intestinal-pancreatic axis form a basis of zinc homeostasis. Proc Natl Acad Sci U S A 101: 14355-14360, 2004. PMID: 15381762.
- Liuzzi JP, and Cousins RJ. Mammalian zinc transporters. Annu Rev Nutr 24: 151-172, 2004. PMID: 15189117.
- Loewen ME, and Forsyth GW. Structure and function of CLCA proteins. Physiol Rev 85: 1061-1092, 2005. PMID: PMID: 15987802.
- Luo X, Zheng W, Yan M, Lee MG, and Muallem S. Multiple functional P2X and P2Y receptors in the luminal and basolateral membranes of pancreatic duct cells. Am J Physiol 277: C205-215, 1999. PMID: 10444396.
- Martin L, Lodemann U, Bondzio A, Gefeller EM, Vahjen W, Aschenbach JR, Zentek J, and Pieper R. A high amount of dietary zinc changes the expression of zinc transporters and metallothionein in jejunal epithelial cells in vitro and in vivo but does not prevent zinc accumulation in jejunal tissue of piglets. J Nutr 143: 1205-1210, 2013. PMID: 23761649.
- Matsuki-Fukushima M, Fujita-Yoshigaki J, Murakami M, Katsumata-Kato O, Yokoyama M, and Sugiya H. Involvement of AQP6 in the Mercury-sensitive osmotic lysis of rat parotid secretory granules. J Membr Biol 246: 209-214, 2013. PMID: 23183829.
- Matsuki-Fukushima M, Hashimoto S, Shimono M, Satoh K, Fujita-Yoshigaki J, and Sugiya H. Presence and localization of aquaporin-6 in rat parotid acinar cells. Cell Tissue Res 332: 73-80, 2008. PMID: 18188600.
- Medhurst AD, Rennie G, Chapman CG, Meadows H, Duckworth MD, Kelsell RE, Gloger, II, and Pangalos MN. Distribution analysis of human two pore domain potassium channels in tissues of the central nervous system and periphery. Brain Res Mol Brain Res 86: 101-114, 2001. PMID: 11165377.
- Meldolesi J. Membranes of pancreatic zymogen granules. Methods Enzymol 98: 67-75, 1983. PMID: 6669071.
- Mendez-Gimenez L, Ezquerro S, da Silva IV, Soveral G, Fruhbeck G, and Rodriguez A. Pancreatic Aquaporin-7: A Novel Target for Anti-diabetic Drugs? Front Chem 6: 99, 2018. PMID: 29675407.
- Miyaji T, Sawada K, Omote H, and Moriyama Y. Divalent cation transport by vesicular nucleotide transporter. J Biol Chem 286: 42881-42887, 2011. PMID: 22052906.
- Moriyama Y, Hiasa M, Sakamoto S, Omote H, and Nomura M. Vesicular nucleotide transporter (VNUT): appearance of an actress on the stage of purinergic signaling. Purinergic Signal 13: 387-404, 2017. PMID: 28616712.
- Murrell-Lagnado RD. A role for P2X4 receptors in lysosome function. J Gen Physiol 150: 185-187, 2018. PMID: 29358186.
- Nezu A, Tanimura A, Morita T, Irie K, Yajima T, and Tojyo Y. Evidence that zymogen granules do not function as an intracellular Ca2+ store for the generation of the Ca2+ signal in rat parotid acinar cells. Biochem J 363: 59-66, 2002. PMID: 11903047.
- Nguyen T, Chin WC, and Verdugo P. Role of Ca2+/K+ ion exchange in intracellular storage and release of Ca2+. Nature 395: 908-912, 1998. PMID: 9804425.
- Niederau C, Van Dyke RW, Scharschmidt BF, and Grendell JH. Rat pancreatic zymogen granules. An actively acidified compartment. Gastroenterology 91: 1433-1442, 1986. PMID: 3021556.
- Norlund R, Roos N, and Taljedal IB. Quantitative energy dispersive X-ray microanalysis of eight elements in pancreatic endocrine and exocrine cells after cryo-fixation. Biosci Rep 7: 859-869, 1987. PMID: 3329534.
- North RA. Molecular physiology of P2X receptors. Physiol Rev 82: 1013-1067, 2002. PMID: 12270951.
- Novak I. ATP as a signaling molecule: the exocrine focus. News Physiol Sci 18: 12-17, 2003. PMID: 12531926.
- Ohana E, Hoch E, Keasar C, Kambe T, Yifrach O, Hershfinkel M, and Sekler I. Identification of the Zn2+ binding site and mode of operation of a mammalian Zn2+ transporter. J Biol Chem 284: 17677-17686, 2009. PMID: 19366695.
- Ohta E, Itoh T, Nemoto T, Kumagai J, Ko SB, Ishibashi K, Ohno M, Uchida K, Ohta A, Sohara E, Uchida S, Sasaki S, and Rai T. Pancreas-specific aquaporin 12 null mice showed increased susceptibility to caerulein-induced acute pancreatitis. Am J Physiol Cell Physiol 297: C1368-1378, 2009. PMID: 19726746.
- Orci L, Ravazzola M, and Anderson RG. The condensing vacuole of exocrine cells is more acidic than the mature secretory vesicle. Nature 326: 77-79, 1987. PMID: 2434862.
- Orlowski J, and Grinstein S. Emerging roles of alkali cation/proton exchangers in organellar homeostasis. Curr Opin Cell Biol 19: 483-492, 2007. PMID: 17646094.
- Pace CS, and Sachs G. Glucose-induced proton uptake in secretory granules of beta-cells in monolayer culture. Am J Physiol 242: C382-387, 1982. PMID: 6282137.
- Palmiter RD, Cole TB, and Findley SD. ZnT-2, a mammalian protein that confers resistance to zinc by facilitating vesicular sequestration. EMBO J 15: 1784-1791, 1996. PMID: 8617223.
- Palmiter RD, and Findley SD. Cloning and functional characterization of a mammalian zinc transporter that confers resistance to zinc. EMBO J 14: 639-649, 1995. PMID: 7882967.
- Paroutis P, Touret N, and Grinstein S. The pH of the secretory pathway: measurement, determinants, and regulation. Physiology (Bethesda) 19: 207-215, 2004. PMID: 15304635.
- Patel AC, Brett TJ, and Holtzman MJ. The role of CLCA proteins in inflammatory airway disease. Annu Rev Physiol 71: 425-449, 2009. PMID: 18954282.
- Patel S, and Cai X. Evolution of acidic Ca2+ stores and their resident Ca2+-permeable channels. Cell Calcium 57: 222-230, 2015. PMID: 25591931.
- Pedemonte N, and Galietta LJ. Structure and function of TMEM16 proteins (anoctamins). Physiol Rev 94: 419-459, 2014. PMID: 24692353.
- Pekas JC. Pancreatic incorporation of 65Zn and histidine-14C into secreted proteins of the pig. Am J Physiol 220: 799-803, 1971. PMID: 5545692.
- Petersen OH, Gerasimenko OV, Tepikin AV, and Gerasimenko JV. Aberrant Ca2+ signalling through acidic calcium stores in pancreatic acinar cells. Cell Calcium 50: 193-199, 2011. PMID: 21435718.
- Poea-Guyon S, Ammar MR, Erard M, Amar M, Moreau AW, Fossier P, Gleize V, Vitale N, and Morel N. The V-ATPase membrane domain is a sensor of granular pH that controls the exocytotic machinery. J Cell Biol 203: 283-298, 2013. PMID: 24165939.
- Quinton PM. Cystic fibrosis: impaired bicarbonate secretion and mucoviscidosis. Lancet 372: 415-417, 2008. PMID: 18675692.
- Rindler MJ, Xu CF, Gumper I, Smith NN, and Neubert TA. Proteomic analysis of pancreatic zymogen granules: identification of new granule proteins. J Proteome Res 6: 2978-2992, 2007. PMID: 17583932.
- Roomans GM, and Wei X. X-ray microanalysis of resting and stimulated rat pancreas. Acta Physiol Scand 124: 353-359, 1985. PMID: 4050472.
- Roos N. A possible site of calcium regulation in rat exocrine pancreas cells: an X-ray microanalytical study. Scanning Microsc 2: 323-329, 1988. PMID: 3368762.
- Roussa E, Alper SL, and Thévenod F. Immunolocalization of anion exchanger AE2, Na+/H+ exchangers NHE1 and NHE4, and vacuolar type H+-ATPase in rat pancreas. J Histochem Cytochem 49: 463-474, 2001. PMID: 11259449.
- Roussa E, Wittschen P, Wolff NA, Torchalski B, Gruber AD, and Thévenod F. Cellular distribution and subcellular localization of mCLCA1/2 in murine gastrointestinal epithelia. J Histochem Cytochem 58: 653-668, 2010. PMID: 20385786.
- Sawada K, Echigo N, Juge N, Miyaji T, Otsuka M, Omote H, Yamamoto A, and Moriyama Y. Identification of a vesicular nucleotide transporter. Proc Natl Acad Sci U S A 105: 5683-5686, 2008. PMID: 18375752.
- Seve M, Chimienti F, Devergnas S, and Favier A. In silico identification and expression of SLC30 family genes: an expressed sequence tag data mining strategy for the characterization of zinc transporters' tissue expression. BMC Genomics 5: 32, 2004. PMID: 15154973.
- Shusterman E, Beharier O, Shiri L, Zarivach R, Etzion Y, Campbell CR, Lee IH, Okabayashi K, Dinudom A, Cook DI, Katz A, and Moran A. ZnT-1 extrudes zinc from mammalian cells functioning as a Zn2+/H+ exchanger. Metallomics 6: 1656-1663, 2014. PMID: 24951051.
- Sondergaard LG, Stoltenberg M, Doering P, Flyvbjerg A, and Rungby J. Zinc ions in the endocrine and exocrine pancreas of zinc deficient rats. Histol Histopathol 21: 619-625, 2006. PMID: 16528672.
- Stauber T, and Jentsch TJ. Chloride in vesicular trafficking and function. Annu Rev Physiol 75: 453-477, 2013. PMID: 23092411.
- Stobrawa SM, Breiderhoff T, Takamori S, Engel D, Schweizer M, Zdebik AA, Bosl MR, Ruether K, Jahn H, Draguhn A, Jahn R, and Jentsch TJ. Disruption of ClC-3, a chloride channel expressed on synaptic vesicles, leads to a loss of the hippocampus. Neuron 29: 185-196, 2001. PMID: 11182090.
- Sun X, and Jiang X. Combination of FASP and fully automated 2D-LC-MS/MS allows in-depth proteomic characterization of mouse zymogen granules. Biomed Chromatogr 27: 407-408, 2013. PMID: 22949036.
- Tani T, Koyama Y, Nihei K, Hatakeyama S, Ohshiro K, Yoshida Y, Yaoita E, Sakai Y, Hatakeyama K, and Yamamoto T. Immunolocalization of aquaporin-8 in rat digestive organs and testis. Arch Histol Cytol 64: 159-168, 2001. PMID: 11436986.
- Thévenod F. Channels and Transporters in Zymogen Granule Membranes and their Role in Granule Function: Recent Progress and a Critical Assessment. In: The Pancreapedia: Exocrine Pancreas Knowledge Base. 2015. DOI: 10.3998/panc.2015.01.
- Thévenod F. Ion channels in secretory granules of the pancreas and their role in exocytosis and release of secretory proteins. Am J Physiol Cell Physiol 283: C651-C672, 2002. PMID: 12176723.
- Thévenod F, Chathadi KV, Jiang B, and Hopfer U. ATP-sensitive K+ conductance in pancreatic zymogen granules: block by glyburide and activation by diazoxide. J Membr Biol 129: 253-266, 1992. PMID: 1433277.
- Thévenod F, Dehlinger-Kremer M, Kemmer TP, Christian AL, Potter BV, and Schulz I. Characterization of inositol 1,4,5-trisphosphate-sensitive (IsCaP) and -insensitive (IisCaP) nonmitochondrial Ca2+ pools in rat pancreatic acinar cells. J Membr Biol 109: 173-186, 1989. PMID: 2527996.
- Thévenod F, Haase W, and Hopfer U. Large-scale purification of calf pancreatic zymogen granule membranes. Anal Biochem 202: 54-60, 1992. PMID: 1377890.
- Thévenod F, Kemmer TP, Christian AL, and Schulz I. Characterization of MgATP-driven H+ uptake into a microsomal vesicle fraction from rat pancreatic acinar cells. J Membr Biol 107: 263-275, 1989. PMID: 2469802.
- Thévenod F, Roussa E, Benos DJ, and Fuller CM. Relationship between a HCO3- -permeable conductance and a CLCA protein from rat pancreatic zymogen granules. Biochem Biophys Res Commun 300: 546-554, 2003. PMID: 12504118.
- Thévenod F, and Schulz I. H+-dependent calcium uptake into an IP3-sensitive calcium pool from rat parotid gland. Am J Physiol 255: G429-440, 1988. PMID: 3263053.
- Thiemann A, Grunder S, Pusch M, and Jentsch TJ. A chloride channel widely expressed in epithelial and non-epithelial cells. Nature 356: 57-60, 1992. PMID: 1311421.
- Thomas GP, Gerlach U, and Antzelevitch C. HMR 1556, a potent and selective blocker of slowly activating delayed rectifier potassium current. J Cardiovasc Pharmacol 41: 140-147, 2003. PMID: 12500032.
- Thompson E, Eldstrom J, and Fedida D. Hormonal Signaling Actions on Kv7.1 (KCNQ1) Channels. Annu Rev Pharmacol Toxicol 2020. PMID: 32667860.
- Thorn P, Lawrie AM, Smith PM, Gallacher DV, and Petersen OH. Ca2+ oscillations in pancreatic acinar cells: spatiotemporal relationships and functional implications. Cell Calcium 14: 746-757, 1993. PMID: 8131191.
- Titievsky AV, Takeo T, Tepikin AV, and Petersen OH. Decrease of acidity inside zymogen granules inhibits acetylcholine- or inositol trisphosphate-evoked cytosolic Ca2+ spiking in pancreatic acinar cells. Pflugers Arch 432: 938-940, 1996. PMID: 8772146.
- Vallee BL, and Neurath H. Carboxypeptidase, a zinc metalloenzyme. J Biol Chem 217: 253-261, 1955. PMID: 13271390.
- van de Put FH, and Elliott AC. The endoplasmic reticulum can act as a functional Ca2+ store in all subcellular regions of the pancreatic acinar cell. J Biol Chem 272: 27764-27770, 1997. PMID: 9346920.
- Vandecaetsbeek I, Vangheluwe P, Raeymaekers L, Wuytack F, and Vanoevelen J. The Ca2+ pumps of the endoplasmic reticulum and Golgi apparatus. Cold Spring Harb Perspect Biol 3: 2011. PMID: 21441596.
- Wang D, Epstein D, Khalaf O, Srinivasan S, Williamson WR, Fayyazuddin A, Quiocho FA, and Hiesinger PR. Ca2+-Calmodulin regulates SNARE assembly and spontaneous neurotransmitter release via v-ATPase subunit V0a1. J Cell Biol 205: 21-31, 2014. PMID: 24733584.
- Whitlock JM, and Hartzell HC. Anoctamins/TMEM16 Proteins: Chloride Channels Flirting with Lipids and Extracellular Vesicles. Annu Rev Physiol 79: 119-143, 2017. PMID: 27860832.
- Williams JA. The nobel pancreas: a historical perspective. Gastroenterology 144: 1166-1169, 2013. PMID: 23622125.
- Yang K, Ding YX, and Chin WC. K+-induced ion-exchanges trigger trypsin activation in pancreas acinar zymogen granules. Arch Biochem Biophys 459: 256-263, 2007. PMID: 17270141.
- Yang YD, Cho H, Koo JY, Tak MH, Cho Y, Shim WS, Park SP, Lee J, Lee B, Kim BM, Raouf R, Shin YK, and Oh U. TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature 455: 1210-1215, 2008. PMID: 18724360.
- Yasui M. pH regulated anion permeability of aquaporin-6. Handb Exp Pharmacol 299-308, 2009. PMID: 19096784.
- Yasui M, Hazama A, Kwon TH, Nielsen S, Guggino WB, and Agre P. Rapid gating and anion permeability of an intracellular aquaporin. Nature 402: 184-187, 1999. PMID: 10647010.
- Yasui M, Kwon TH, Knepper MA, Nielsen S, and Agre P. Aquaporin-6: An intracellular vesicle water channel protein in renal epithelia. Proc Natl Acad Sci U S A 96: 5808-5813, 1999. PMID: 10318966.
- Yoo SH. Role of secretory granules in inositol 1,4,5-trisphosphate-dependent Ca2+ signaling: from phytoplankton to mammals. Cell Calcium 50: 175-183, 2011. PMID: 21176957.
- Yoo SH, and Albanesi JP. Inositol 1,4,5-trisphosphate-triggered Ca2+ release from bovine adrenal medullary secretory vesicles. J Biol Chem 265: 13446-13448, 1990. PMID: 2166028.
- Yule DI, Ernst SA, Ohnishi H, and Wojcikiewicz RJ. Evidence that zymogen granules are not a physiologically relevant calcium pool. Defining the distribution of inositol 1,4,5-trisphosphate receptors in pancreatic acinar cells. J Biol Chem 272: 9093-9098, 1997. PMID: 9083036.
- Zhao H, and Muallem S. Na+, K+, and Cl- transport in resting pancreatic acinar cells. J Gen Physiol 106: 1225-1242, 1995. PMID: 8786358.
- Zheng D, Feeney GP, Handy RD, Hogstrand C, and Kille P. Uptake epithelia behave in a cell-centric and not systems homeostatic manner in response to zinc depletion and supplementation. Metallomics 6: 154-165, 2014. PMID: 24301558.
- Zhong XZ, Cao Q, Sun X, and Dong XP. Activation of lysosomal P2X4 by ATP transported into lysosomes via VNUT/SLC17A9 using V-ATPase generated voltage gradient as the driving force. J Physiol 594: 4253-4266, 2016. PMID: 27477609.