Entry Version:
Citation:
Pancreapedia: Exocrine Pancreas Knowledge Base, DOI: 10.3998/panc.2012.16
| Attachment | Size |
|---|---|
| 352.31 KB |
Gene smbol: BECN1
Other Names: ATG6, Vps30
1. General Function
Beclin 1/ATG6/Vps30 (UniProtKB/Swiss-Prot Q14457) is a 450 amino-acids length protein, with three domains; BH3 (aminoacid 114 to 123), Coiled Coil domain (CCD; aminoacid 144 to 269) and the Evolutionarily Conserved Domain (ECD; aminoacid 244 to 337). BH3 proteins are part of the Bcl-2 family; they are pro-apoptotic damage sensors that play an important role in protecting against cancer (1). The BH3-only domain of Beclin 1 can interact with Bcl-2 and Bcl-XL (2;3). Both cellular and viral Bcl-2 (vBcl-2), or more specifically ER-targeted Bcl-2, inhibit Beclin 1-dependent autophagy by interfering with the Beclin 1-PtdIns 3-kinase interaction (PI3K) and the Beclin 1-associated PI3K activity (3,4). The interaction between Bcl-2 and Beclin 1 is greatly reduced upon starvation, which suggests that the dissociation of Bcl-2 from Beclin1 is important for activating autophagy. We demonstrated that, VMP1 (Vacuole Membrane Protein 1) displaces Bcl-2 from Beclin 1, partitioning Beclin 1 to the autophagic pathway (Molejon et al., Sci Rep. 2012; In press).
The ECD is essential for Beclin 1 mediated autophagy and to inhibit tumorigenesis. Beclin 1 also contains a short leucine-rich amino acid sequence that is responsible for its efficient nuclear export signal (NES) (5). Mutations of the Beclin 1 NES interfere with its ability to promote nutrient deprivation-induced autophagy and suppress tumorigenesis. PI3KC3/Vps34 interacts with the ECD and CCD domains, while the activating molecule in Beclin 1-regulated autophagy (Ambra1)/UV radiation resistance-associated gene (UVRAG)/Atg14L interact with the CCD domain. The CCD, a universal oligomerization domain, mediates Beclin 1 self-interaction and dimer formation in vivo and in vitro. The amino terminus binds less effectively than the CCD to contribute to Beclin 1 self-assembly.
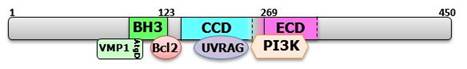
Figure 1. Schematic representations of domains of human Beclin 1 and the Bcl-2-Beclin 1-PtdIns 3-kinase-UVRAG multiprotein complex. Beclin 1 has a BH3 domain (amino acids 114-123), a central coiled-coil domain (CCD, amino acids 144-269) and an evolutionarily conserved domain (ECD, amino acids 244-337). VMP1 and Bcl-2 interacts with the BH3 domain of Beclin 1, UVRAG with the CCD, and the class III PtdIns 3-kinase with the ECD and CCD.
Beclin 1, through its BH3 domain, binds to VMP1-carboxyl-terminus domain (named: Autophagy related domain (AtgD)) promoting autophagy (6; Molejon, Sci Rep. 2012; In press). This interaction promotes Beclin1-hVps34 complex, which is responsible for autophagic vesicle nucleation in mammals (7,8). Also, Beclin1-BH3 interacts with Bcl-2 and this interaction leads to inhibition of autophagy by interfering with the formation and activity of the autophagy promoter complex, Beclin 1-hVps34 (4). Mutations impairing VMP1-Beclin1 interaction lead to inhibition of the autophagic process in pancreatic human cells (Molejon et al., Sci Rep. 2012; In press).
a. Beclin 1 is a mammalian tumor suppressor
The autophagy function of Atg6/Beclin 1 is highly conserved throughout eukaryotic evolution. In addition to Beclin 1’s role in autophagy, genetic knockdown or knockout studies of beclin 1 have demonstrated its role as a tumor suppresor gene. In humans, monoallelic deletions of beclin 1 are frequently observed in sporadic breast, ovarian and prostate carcinoma (9).
Bcl-2 is an anti-apoptotic protein (10), and Beclin 1 was first identified as a Bcl-2-interacting protein in a yeast two-hybrid screen (8,11). The beclin 1 gene is monoallelically deleted in up to 75% of ovarian, 50% of breast and 40% of prostate cancers (12). Decreased expression of Beclin1 is also observed in other types of cancers including human brain tumors (13) and cervical cell carcinoma (14). Expression of Beclin 1 in human MCF7 breast carcinoma cells promotes autophagy and inhibits in vitro clonigenicity and tumorigenesis in nude mice (7). Beclin 1 is a haploinsufficient tumor suppressor gene in mice (15, 16), sharing 98% identity with human beclin 1. Homozygous beclin 1−/− mice die in early embryogenesis, and heterozygous beclin 1+/− mice have reduced autophagy levels and increased incidence of spontaneous tumors, which, together with the in vitro data, establishes a clear role for autophagy, and Beclin 1, in tumor suppression.
Beclin 1 levels appear to be one of the critical factors that affect the induction of autophagy. As indicated above, beclin 1 is haploinsufficient, and various cancer cells show decreased levels of Beclin 1 (17). The anti-cancer drug tamoxifen may work in part by increasing expression of this protein (18). More evidence also supports the importance of Beclin 1 levels in autophagy regulation. For example, ceramide is thought to play a role in apoptosis (19, 20). Altered levels of Beclin 1 are also seen in situations other than cancer that involve autophagy; high levels of Beclin 1 are associated with neurons at the site of traumatic brain injury (21), whereas inhibition of Beclin 1 expression protects against cell death due to ischemia/reperfusion (22). Finally, a recent elegant work showed that Akt-mediated phosphorylation of Beclin 1 functions in autophagy inhibition and oncogénesis (23).
b. Induction of autophagy by Beclin 1- PI3K Complex
Although more Beclin 1 binding proteins and complexes are being identified in mammals, each of the individual complexes seems to recycle various elements derived from other cellular processes. Beclin 1 (Atg6/Vps30), PI3KC3/Vps34 and Vps15 have been predicted to regulate autophagy in a similar manner to yeast. Phosphatidylinositol 3-phosphate (PI3P) is essential for canonical autophagosome formation. Vacuolar protein sorting protein 34 (Vps34) and class III PI3K (hVps34) produce PI3P for autophagy in yeast and mammals, respectively. In yeast, Vps34 forms two distinct protein complexes: Complex I consist of Atg14, Atg6/Vps30, Vps15, and Vps34, whereas complex II is composed of Vps38, Atg6/Vps30, Vps15, and Vps34 (8). Only complex I functions in autophagy, whereas complex II is required for vacuolar protein sorting. The two PtdIns 3-kinase complexes contain three common subunits: the PtdIns 3-kinase enzyme Vps34, the regulatory protein Vps15 and Atg6/Vps30. In addition, each complex has a specific factor, Atg14 for complex I and Vps38 for complex II. Atg14 directs complex I to the phagophore assembly site, also termed the pre-autophagosomal structure (PAS). An autophagosome formation-specific PI3K complex has also been identified in mammals recently. It consists of Atg14L (also known as Atg14 and Barkor), Beclin 1 (Atg6 homolog), hVps15, and hVps34 (24-26). Mammals have at least two other stable hVps34 complexes, which play roles in endosomal transport and autophagosome-lysosome fusion by including UVRAG (UV irradiation resistance-associated gene) and Rubicon (Run domain protein as Beclin 1 interacting and cysteine-rich containing) as subunits instead of Atg14L (one complex contains UVRAG alone, whereas the other contains both UVRAG and the negative regulator Rubicon) (25; 26). Mammalian cells also possess other Beclin 1 binding proteins including Bcl-2, AMBRA1, Bif-1 (also known as Endophilin B1), and the pancreatitis associated transmembrane protein VMP1 (6,27; Molejon et al., Sci Rep. 2012; In press). In addition to PI3K, phosphatidylinositol 3-phosphatases including myotubularin-related phosphatase 3 (MTMR3) and Jumpy (MTMR14) are implicated in autophagy (28, 29). They negatively regulate autophagosome formation and autophagosome size. Therefore, the balance between PI3K and phosphatidylinositol 3- phosphatase determines autophagy initiation mediating local PI3P levels.
c. Inhibition of autophagy by Beclin 1–Bcl-2/Bcl-XL complexes
Mutations of either the BH3-only domain within Beclin 1, or the BH3 receptor domain within Bcl-2 or Bcl-XL, disrupted the Beclin 1–Bcl-2 complex, resulting in the stimulation of autophagy. Bcl-2 or Bcl-XL reduces the pro-autophagic activity of Beclin 1 (2, 4). Interestingly, ER-localized Bcl-2, but not mitochondrial-localized Bcl-2, inhibits autophagy (30), which is consistent with the older notion that ER-associated class III PI3K activity may be crucial in the nucleation of autophagosome formation.
Different mechanisms have been described to regulate dissociation of Beclin 1 and Bcl-2/Bcl-XL during autophagy in mammalian cells. These include: (A) competitive displacement of the Beclin 1 BH3 domain by other Bcl-2 family proteins. The interaction between Beclin 1 and the anti-apoptotic proteins is inhibited by tBid, Bad and BNIP3, but not by Bax and Bak (30). Moreover, the pro-apoptotic BH3-only proteins such as BNIP3, Bad, Noxa, Puma, BimEL and Bik all induce autophagy (31). (B) JNK1 or ERK-mediated phosphorylation of Bcl-2 (32; 33) or DAPK-mediated phosphorylation of Beclin 1 (34). (C) Competitive displacement of Bcl-2 by other Beclin 1-binding proteins such as HMGB1, UVRAG, Atg14L/Barkor or VMP1; this likely initiates a program of heightened anti-apoptotic state, promotes autophagy, and ultimately protects the cell during cell stress. (D) NAF-1 (nutrient-deprivation autophagy factor-1) dysfunction. NAF-1 is a component of the inositol-1,4,5 trisphosphate (PI3P) receptor complex. NAF-1 binds Bcl-2 and this interaction is independent of a BH3 domain, but depends at least in part on its redox sensitive CDGSH iron/ sulphur-binding domain (36). ROS are important signaling molecules that initiate autophagy. (E) Beclin 1 self-interaction: Beclin 1 can form large homo-oligomers, which may provide a platform for further protein–protein interactions and displacement of Bcl-2 or Bcl-XL.
2. Beclin 1 in Pancreas
Beclin 1 is a VMP1 partner in pancreatic cell autophagy. VMP1-triggered autophagy is induced in vivo during experimental acute pancreatitis, where it is associated with Beclin1 and LC3 in the membrane of autophagosomes (6). We have reported experimental data indicating that VMP1 interacts with Beclin 1. We found the interaction of VMP1 with endogenous Beclin 1 in VMP1-expressing cells and the interaction of both endogenous proteins in rapamycin-induced autophagic cells. VMP1-Beclin 1 direct interaction was confirmed using recombinant peptides. Moreover, VMP1-Atg domain has proved to be essential for VMP1-induced autophagy, because the VMP1-defective mutant lacking its c-terminus domain -VMP1ΔAtgD- expression failed to induce LC3 recruitment (6). Our data have also shown the co-localization of transient Beclin 1 and LC3 in VMP1-induced vesicles, and this triple co-localization was abolished when cells were transfected with the defective mutant VMP1ΔAtgD. On the other hand, VMP1 expression failed to induce autophagy when it was expressed in low-Beclin 1, MCF7 cells. These findings suggest that VMP1-induced autophagy probably involves the interaction with Beclin 1. Experimental animal models have shown VMP1-Beclin 1 colocalization. VMP1-EGFP co-localized with endogenous Beclin 1 in pancreas from transgenic mice, and both endogenous proteins co-localized in tissue undergoing pancreatitis-induced autophagy (6).
3. Tools for study of Beclin 1
a. cDNA clones
Two human Beclin 1 clones are available from OriGene (Cat. #SC117750 and #SC324265). Also OriGene offer a GFP-tagged clone (Cat #RG201629), a Myc-DDK-tagged clone (Cat.#RC201629). None of these have been checked by us.

Figure 2. AR42J rat acinar cell (A); 293T Human Embryonic Kidney (B); and HeLa human cervical cancer cell line were transfected with the CFP-Beclin 1 expression plasmid. Confocal microscope image, inverted LSM Olympus FV1000, Buenos Aires, Argentina.
b. Antibodies
Rabbit antibody raised against Beclin 1 have been used to identify Beclin 1 by western blotting, IP, immunofluorescence (Sigma Aldrich Cat. B6186). We used this antibody for western blot, IP and inmunofluorescence at a dilution of 1:100-1:500. We also used anti-Beclin 1 (Santa Cruz Biotechnology, Inc. Cat. sc-11427) with the SNAP i.d. system for western blot at a dilution of 1/500.
c. Viral Vectors
The human BECN1 cDNA was cloned into the pReceiver-Lv105 lentivirus vector (GeneCopoeia Cat. LP-M0768-Lv105-0200-S) (39).
d. Mouse Models
Beclin 1 is a novel Bcl-2-homology (BH)-3 domain only protein. The embryonic phenotype of Beclin 1 null mice is even more severe than that of other autophagy gene-deficient mice, which die in early embryonic development (E7.5 or earlier) with defects in proamniotic canal closure (37). Heterozygous beclin 1+/− mice have reduced autophagy activity and increased incidence of spontaneous tumors.
4. References
- Karst AM, Li G. BH3-only proteins in tumorigenesis and malignant melanoma. Cell Mol Life Sci. 2007; 64:318–330. PMID: 17192811
- Maiuri MC, Le Toumelin G, Criollo A, et al. Functional and physical interaction between Bcl-XL and a BH3-like domain in Beclin-1. EMBO J. 2007; 26:2527–2539. PMID: 17446862
- Oberstein A, Jeffrey PD, Shi Y. Crystal structure of the Bcl-XL-Beclin 1 peptide complex: Beclin 1 is a novel BH3-only protein. J Biol Chem. 2007; 282:13123–13132. PMID: 17337444
- Pattingre S, Tassa A, Qu X, et al. Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell. 2005; 122: 927–939. PMID: 16179260
- Liang XH, Yu J, Brown K, Levine B. Beclin 1 contains a leucine-rich nuclear export signal that is required for its autophagy and tumor suppressor function. Cancer Res. 2001; 61: 3443–3449. PMID: 11309306
- Ropolo A, Grasso D, Pardo R, Sacchetti ML, Archange C, Lo Re A, Seux M, Nowak J, Gonzalez CD, Iovanna JL, and Vaccaro MI. The pancreatitis-induced vacuole membrane protein 1 triggers autophagy in mammalian cells. J Biol Chem. 2007; 282: 37124-37133. PMID: 17940279
- Liang XH, Jackson S, Seaman M, et al. Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature. 1999; 402: 672–676. PMID: 10604474
- Kihara A, Kabeya Y, Ohsumi Y, Yoshimori T. Beclin-phosphatidylinositol 3-kinase complex functions at the trans-Golgi network. EMBO Rep. 2001; 2:330–335. PMID: 11306555
- Levine B, Klionsky DJ. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell. 2004; 6:463–477. PMID: 15068787
- Walensky LD. BCL-2 in the crosshairs: tipping the balance of life and death. Cell Death Differ. 2006; 13: 1339–1350. PMID: 16763614
- Liang XH, Kleeman LK, Jian HH, Gordon G, Goldamn JE, Berry G, Herman B, Levine B. Protection against Fatal Sindbis Virus Encephalitis by Beclin,a Novel Bcl-2-Interacting Protein. J Virol. 1998; 72: 8586–8596. PMID: 9765397
- Aita VM, Liang XH, Murty VVVS, et al. Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics. 1999; 59: 59–65. PMID: 10395800
- Miracco C, Cosci E, Oliveri G, et al. Protein and mRNA expression of autophagy gene Beclin 1 in human brain tumours. Int J Oncol. 2007; 30:429–436. PMID: 17203225
- Wang ZH, Xu L, Duan ZL, et al. Beclin 1-mediated macroautophagy involves regulation of caspase-9 expression in cervical cancer HeLa cells. Gynecol Oncol. 2007. PMID: 17617446
- Qu X, Yu J, Bhagat G, et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest. 2003; 112:1809–1820. PMID: 14638851
- Yue Z, Jin S, Yang C, Levine AJ, Heintz N. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci USA. 2003; 100: 15077–15082. PMID: 14657337
- Miracco C, Cosci E, Oliveri G, et al. Protein and mRNA expression of autophagy gene Beclin 1 in human brain tumours. Int J Oncol. 2007; 30:429–436. PMID: 17203225
- Furuya N, Liang XH, Levine B. Autophagy and cancer. In: Klionsky DJ. ed. Autophagy. Georgetown, TX: Landes Bioscience. 2004.
- Levade T, Malagarie-Cazenave S, Gouaze V, et al. Ceramide in apoptosis: a revisited role. Neurochem Res. 2002; 27:601–607. PMID: 12374195
- Pettus BJ, Chalfant CE, Hannun YA. Ceramide in apoptosis: an overview and current perspectives. Biochim Biophys Acta. 2002; 1585:114–125. PMID: 12531544
- Diskin T, Tal-Or P, Erlich S, et al. Closed head injury induces upregulation of Beclin 1 at the cortical site of injury. J Neurotrauma. 2005; 22:750–762. PMID: 16004578
- Valentim L, Laurence KM, Townsend PA, et al. Urocortin inhibits Beclin1-mediated autophagic cell death in cardiac myocytes exposed to ischaemia/reperfusion injury. J Mol Cell Cardiol. 2006; 40:846–852. PMID: 16697404
- Wang RC, Wei Y, An Z, Zou Z, Xiao G, Bhagat G, White M, Reichelt J, Levine B. Akt-Mediated Regulation of Autophagy and Tumorigenesis Through Beclin 1 Phosphorylation. Science. 2012 Oct 25. [Epub ahead of print] PMID: 23112296
- Itakura E, Kishi C, Inoue K, Mizushima N. Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell. 2008; 19:5360–5372. PMID: 18843052
- Matsunaga K, Saitoh T, Tabata K, Omori H, Satoh T, Kurotori N, et al. Two Beclin 1-binding proteins, Atg14L and Rubicon, reciprocally regulate autophagy at different stages. Nat Cell Biol. 2009; 11: 385–396. PMID: 19270696
- Zhong Y, Wang QJ, Li X, Yan Y, Backer JM, Chait BT, et al. Distinct regulation of autophagic activity by Atg14L and Rubicon associated with Beclin 1-phosphatidylinositol-3-kinase complex. Nat Cell Biol. 2009; 11: 468–476. PMID: 19270693
- Funderburk SF, Wang QJ, Yue Z. The Beclin 1-VPS34 complex – at the crossroads of autophagy and beyond. Trends in Cell Biology. 2010; 20: 355–362. PMID: 20356743
- Taguchi-Atarashi N, Hamasaki M, Matsunaga K, Omori H, Ktistakis NT, Yoshimori T, Noda T. Modulation of local PtdIns3P levels by the PI phosphatase MTMR3 regulates constitutive autophagy. Traffic. 2010; 4:468-78. PMID: 20059746
- Vergne I, Roberts E, Elmaoued RA, Tosch V, Delgado MA, Proikas-Cezanne T, Laporte J, Deretic V. Control of autophagy initiation by phosphoinositide 3-phosphatase Jumpy. EMBO J. 2009; 28(15): 2244-58. PMID: 19590496
- Petiot A, Ogier-Denis E, Blommaart EF, Meijer AJ, Codogno P. Distinct classes of phosphatidylinositol 3'-kinases are involved in signaling pathways that control macroautophagy in HT-29 cells. J Biol Chem. 2000; 275:992–998. PMID: 10625637
- Obara K, Sekito T, Ohsumi Y. Assortment of phosphatidylinositol 3-kinase complexes – Atg14p directs association of complex I to the pre-autophagosomal structure in Saccharomyces cerevisiae. Mol Biol Cell. 2006; 17:1527–1539. PMID: 16421251
- Sinha S, Levine B. The autophagy effector Beclin 1: a novel BH3-only protein. Oncogene. 2008; 27. PMID: 19641499
- Wei Y, Pattingre S, Sinha S, Bassik M, Levine B. JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol Cell. 2008; 30:678–688. PMID: 18570871
- Tang D, Kang R, Livesey KM, Cheh CW, Farkas A, Loughran P, et al. Endogenous HMGB1 regulates autophagy. J Cell Biol. 2010; 190:881–892. PMID: 20819940
- Zalckvar E, Berissi H, Mizrachy L, Idelchuk Y, Koren I, Eisenstein M, et al. DAP-kinase-mediated phosphorylation on the BH3 domain of beclin 1 promotes dissociation of beclin 1 from Bcl-XL and induction of autophagy. EMBO Rep. 2009; 10:285–292. PMID: 19180116
- Chen CH, Wang WJ, Kuo JC, Tsai HC, Lin JR, Chang ZF, et al. Bidirectional signals transduced by DAPK-ERK interaction promote the apoptotic effect of DAPK. Embo J. 2005; 24: 294–304. PMID: 15616583
- Chang NC, Nguyen M, Germain M, Shore GC. Antagonism of Beclin 1-dependent autophagy by BCL-2 at the endoplasmic reticulum requires NAF-1. Embo J. 2010; 29:606–618. PMID: 20010695
- Yue Z, Jin S, Yang C, Levine AJ and Heintz N. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci. 2003; 100(25): 15077–15082. PMID: 14657337
- Liang XH, Kleeman LK, Jiang HH, Gordon G, Goldman JE, Berry G, Herman and Levine B. Protection against Fatal Sindbis Virus Encephalitis by Beclin, a Novel Bcl-2-Interacting Protein. Journal of Virology. 1998; 72(11): 8586–8596. PMID: 9765397


